Multiple Attempts Not allowed. This test can only be taken once. Force Completion This test can be saved and resumed later. Your answers are saved automatically. Question Completion Status: 14 A Moving to another question will save this response. Question 1 Record your response as a numerical response only. Question 1 of 10 >>>> 1 points Save Answer In general, reaction rates double when the temperature is increased by 10° C. The temperature of a reaction is increased by 30° C. The rate of the reaction will increase by a factor of: A Moving to another question will save this response. Question 1 of 10 >>>> K
Multiple Attempts Not allowed. This test can only be taken once. Force Completion This test can be saved and resumed later. Your answers are saved automatically. Question Completion Status: 14 A Moving to another question will save this response. Question 1 Record your response as a numerical response only. Question 1 of 10 >>>> 1 points Save Answer In general, reaction rates double when the temperature is increased by 10° C. The temperature of a reaction is increased by 30° C. The rate of the reaction will increase by a factor of: A Moving to another question will save this response. Question 1 of 10 >>>> K
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter7: Reaction Rates And Chemical Equilibrium
Section: Chapter Questions
Problem 7.74P
Related questions
Question
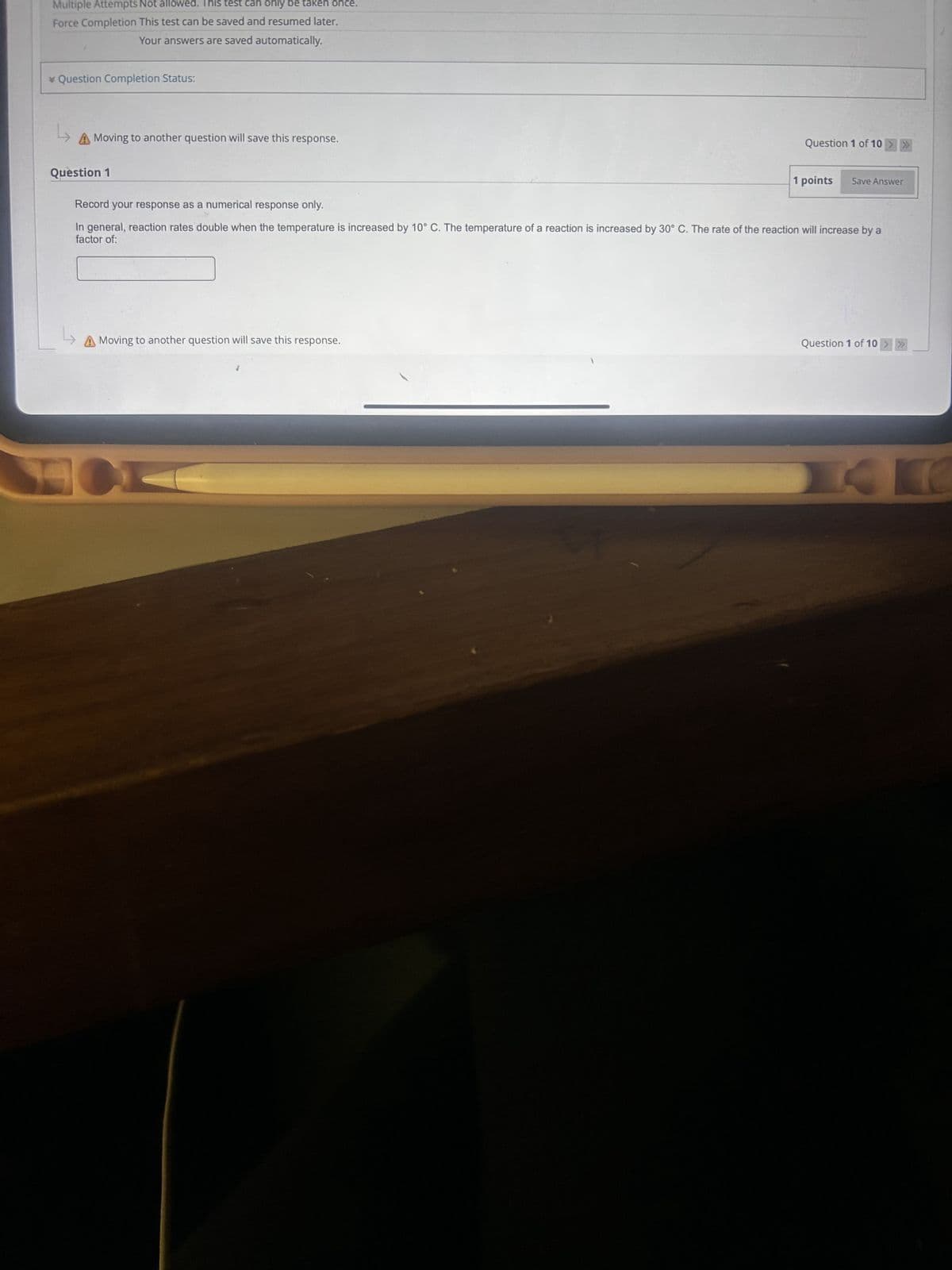
Transcribed Image Text:Multiple Attempts Not allowed. This test can only be taken once.
Force Completion This test can be saved and resumed later.
Your answers are saved automatically.
Question Completion Status:
14
A Moving to another question will save this response.
Question 1
Record your response as a numerical response only.
Question 1 of 10 >>>>
1 points
Save Answer
In general, reaction rates double when the temperature is increased by 10° C. The temperature of a reaction is increased by 30° C. The rate of the reaction will increase by a
factor of:
A Moving to another question will save this response.
Question 1 of 10 >>>>
K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER