Q: The Kp for Ag,CrO, is 1.2 × 10 1². a) Complete writing the balanced equilibrium equation for the dis...
A: Ksp is the solubility product of a sparingly soluble salt in an aqueous medium.
Q: A mixture contains Mg(NO3)2 and KBr. A 1.85 g sample of this mixture is dissolved in water and an ex...
A: Given, The mass of mixture of Mg(NO3)2 and KBr = 1.85 g. This mixture is dissolved in water and an ...
Q: Consider the insoluble compound aluminum phosphate , AIPO4 . The aluminum ion also forms a complex w...
A: Kf= 7.7 * 1033 K = ? We have to balanced ionic equation and tell about state of the components And ...
Q: As a chemist, your role is to interpret the structures from quantum chemical calculations based on y...
A: Thus, compounds with an O—O bond length of ≈1.4–1.5 Å and νOO between ≈800 and 930 cm−1 are designat...
Q: volume
A:
Q: Draw the product of the reaction where methoxide behaves as a base. Br. OCH3 + H. H
A:
Q: What are the solvents used in the fabrication of organic electronic devices today? In your words, wh...
A: The solution of the question is given below:
Q: Which of the following statements is incorrect regarding IR spectroscopy? Question 24 options: ...
A:
Q: 1. Fill in the diagram (with "high" or "low") to show how intermolecular forces influence the volati...
A: Vapor pressure, volatility and boiling point are the property of a liquid based on the strength of i...
Q: ᴄᴀʟᴄᴜʟᴀᴛᴇ ᴛʜᴇ ᴅᴇɴꜱɪᴛʏ ᴏꜰ ᴘᴜʀᴇ ᴍᴇᴛᴀʟꜱ, ᴇxᴘʀᴇꜱꜱ ʏᴏᴜʀ ᴀɴꜱᴡᴇʀ ɪɴ ᴋɢ/ᴍ3 ᴀ. Iron @ ꜰʙᴄ ᴀɴᴅ ꜰᴄᴄ ʙ. ᴄopper (...
A: The solution of the question is given below:
Q: Complete the following reactions: || C-0-CH,CH, +NAOH- a. b. CH;(CH,),-C-0-CHCH,+H,O CH3
A: Answer of the question given below,
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side pro...
A: Aldehydes are the organic compounds which contains the aldehyde group (-CHO). The reaction of aldehy...
Q: Explain how you would draw XeF4 and determine its shape, hybridization, polarity and intermolecular ...
A: The molecule given is Xenon tetrafluoride XeF4.
Q: The analysis of a product of combustion on dry basis, when C6H18 was burned with atmospheric air, is...
A: Solution - According to the question - Given - CO2=12%, CO=1.75% O2=3.01% N2=83.24% Assume Product...
Q: At 25 °C, what is the hydroxide ion concentration, [OH¯], in an aqueous solution with a hydrogen ion...
A: [OH-] = 4.54 x 10-8 M
Q: Proton NMR wllen 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0. 0.5 ppm 33.2 Integration Values 48.4 33.3 48.7
A: The correct answer about NMR spectrum is given below
Q: A welder’s acetylene tank has a volume of 75.0 L. It is stored at a temperature of 23.24 °C and has ...
A:
Q: A common ketone starting material is shown below. Predict the major product for each reaction based ...
A: Given that , An aldehyde reacts with different reagents and the respective products are as follows.
Q: Esterase hydrolyzes ester bonds. For simplicity, only the amino acids whose side-chains interact wit...
A: Ans. Yes. Yes, the esterase will most likely bind to the substrate when returned to the pre-treatmen...
Q: Considering IR spectroscopy, which of the following statements is incorrect? Question 20 options:...
A: IR spectroscopy is used to determine the functional groups present in compound.. Rule: molecule cont...
Q: HO H2SO4
A: In elimination reaction a species leave the substrate and double or triple bond is formed.
Q: e Reaction Quotient, Q, is equal to
A: Given, Molarity of KOH = 4.82×10-4 M Volume of KOH = 12 mL Molarity of AgNO3 = 8.44×10-4 M Volume o...
Q: (a) Initial molar concentration of Fe" after mixing: [Fe"]. = Initial molar concentration of SCN" af...
A: Fe3+(aq) + SCN- (aq) ⇔FeSCN2+ ...
Q: Determine the enthalpy of the following reaction: CH3CH=CH2 + 4.50=0 - ---> 30-С-О + ЗН-О-Н using th...
A: Bond bond energy is the energy required to break the bonds between two elements
Q: 9. Consider the following molecule of propanone: a. Use your understanding of VSEPR structures to ex...
A: Carbon Hybridization - Carbon is a vital and common chemical element that is required for organic li...
Q: (14) Which of the following compounds would have the largest absorption in its UV-Vis spectrum? (a) ...
A:
Q: 21. Which of the following compounds is not capable of hydrogen bonding? H. a. H. H FF H. с. H. H. d...
A: Hydrogen bonding: The hydrogen atom attached to the electronegative atom like fluorine, oxygen, or n...
Q: Complete this diagram showing the type of phase change in matter by using the direction the arrows a...
A:
Q: Draw a galvanic represented by the following line notation and calculate the standard cell potential...
A:
Q: A total of 665.19 Joules of heat are observed as 49.79g of lead is heated from 12.0°C to 78.12°C. Fr...
A:
Q: A technical machinist is asked to build a cubical steel tank that will hold 455 L of water. Calculat...
A:
Q: 2. are the percent compositions of the flue (dry basis) if the fuel is burned stoichiometrically? As...
A: The solution of the question is given below: The information given is: C = 83 % H2 = 7% O = 5% S = ...
Q: ST5G.2 - Estimate the solubility of CaF2(aq) [in units of PICOmolar] in the presence of 0.701 molar ...
A: Here we have to determine the solubility of CaF2 in picomolar in presencee of 0.701M of AlF3. Dta Ks...
Q: Consider the following equilibrium. A(g) + B(g) = C(g) + D(s) AH > 0 What effect would each of the f...
A: Given reaction is : A (g) + B (g) <--------------> C (g) + D (s) , ΔH > 0 What effect woul...
Q: 8) A 0.185 M solution of a weak acid has a pH of 2.95. Calculate the acid ionization constant (Ka) f...
A: A 0.185 M solution of a weak acid (HA) has a pH of 2.95. Calculate the acid ionization constant Ka f...
Q: Which of the following bonds will show the peak at highest wavenumberin the IR spectrum of the follo...
A: Wavenumber is defined as the reciprocal of wavelength, and wavenumbers of infrared radiation are typ...
Q: Dehydrating Cyclohexanol Pre lab Q's A. Which the following is true? I both constitutional isbiners ...
A: Dehydration means removal of water. In dehydration reaction of alcohol, the -OH group is first proto...
Q: Identify the suitable reagents for the following: H,O
A:
Q: What type of reaction accompanied by heat liberations? Select one: a. non-spntaneous b. spontaneou...
A: A spontaneous reaction is reaction that occurs without the input of energy. A non spontaneous react...
Q: The equilibrium constant, Kc, for the following reaction is 1.29×10-2 at 600 K. COCI2(g) Co(g) + Cl2...
A: Equilibrium constant Kc = 1.29 * 10-2 Initial concentration of COCl2 = 0.364 M We need to find equi...
Q: Consider this equilibrium reaction at 400 K. Br, (g) + Cl, (g) = 2 BRCI(g) K. = 7.0 = 0.00415 M, [Br...
A:
Q: Use the graph below to answer the following questions. 2. What is the vapor pressure of CHCI3 at 50°...
A: 2) Vapour pressure of CHCl3 at 50∘C = 70 kPa 3) Boiling point of water when external presssure is 30...
Q: Consider the following system at equilibrium where AH° = -10.4 kJ, and Kc = 55.6, at 698 K. H2(9) + ...
A:
Q: 3. A solution that may contain any of the group III cations. Treatment of the solution with CIO in a...
A: Group 3 cation involve Cr3+ , Al3+ , Ni2+ and Fe3+
Q: Reset Help pH neutral Since the anion of PbBry is in acidic solution this compound is in more solubl...
A:
Q: Consider the insoluble compound iron(II) hydroxide , Fe(0H)2 · The iron(II) ion also forms a complex...
A: Fe(OH)2 is an insoluble compound. Iron (II) forms complex with CN- ions. In the presence of CN- ions...
Q: What volume (in mL) of 0.2850 M HBr is required to neutralize 50.00 mL of 0.7000 M KOH?
A:
Q: A supersaturated solution of CH3COO-Na+ will crystallize spontaneously. Predict the signs of AH,AS,A...
A: A reaction can be predicted based on the sign of change in enthalpy, entropy, and Gibb's free energy...
Q: 131 For the radioactive isotope 15'I, the time required for 50% disintegration is 8 days. The time r...
A:
Q: What is the product of the following reaction? HF C14H20
A: Friedel-Crafts Alkylation refers to the replacement of an aromatic proton with an alkyl group. This ...
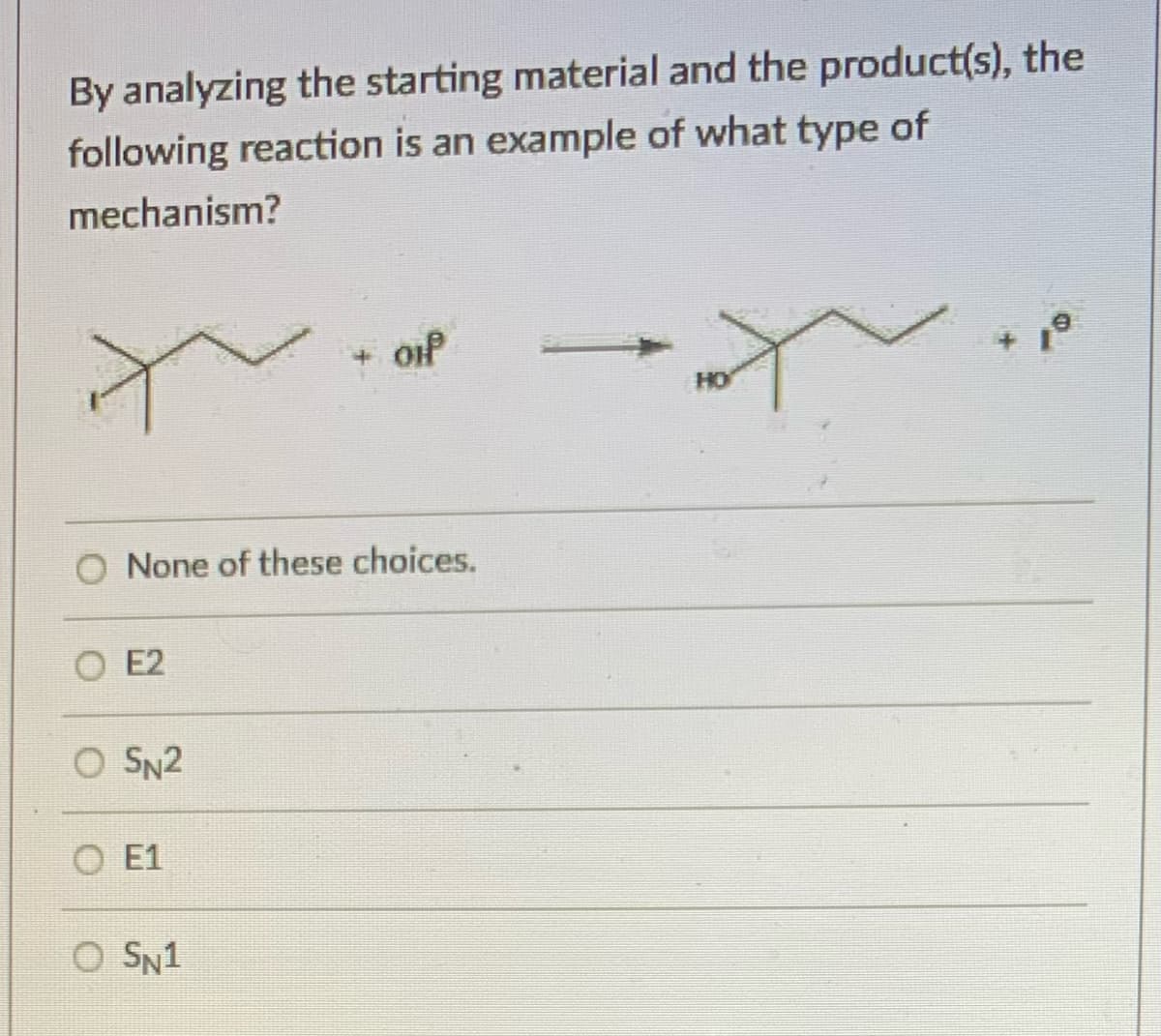
Step by step
Solved in 2 steps

- When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor productsTaking into account anti periplanar geometry, predict the major E2 product formed from each starting material.Draw the organic products formed when cyclopentene is treated with each reagent. With some reagents, no reactionoccurs.a. H2 + Pd-Cb. H2 + Lindlar catalystc. Na, NH3d. CH3CO3He. [1] CH3CO3H; [2] H2O, HO–f. [1] OsO4 + NMO; [2] NaHSO3, H2Og. KMnO4, H2O, HO–h. [1] LiAlH4; [2] H2Oi. [1] O3; [2] CH3SCH3j. (CH3)3COOH, Ti[OCH(CH3)2]4, (–)-DETk. mCPBAl. Product in (k); then [1] LiAlH4; [2] H2O
- Show how to make these deuterium-labeled compounds, using CD3MgBr and D2O as your sources of deuterium, and anynon-deuterated starting materials you wish.(a) CH3CH(OD)CD3Which substituent is the strongest deactivator to EAS reactions? -NO2 -Cl -CH3 -OHThe reaction H2C=CHCH2Br + NaN3 followed by reaction with LiAlH4 yields CH3CH2CH2NH2 H2C=CHCH2NH2 H2C=CHCH2N3 none of the above
- Which of the following substituents acts as a moderate activator and ortho/para director? -SO3H -Br -NHCOR -COOH -CHOWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutane
- Which of the synthetic procedures shown in Image 31 would carry out the following transformation? A. b B. c C. a D. dDraw the ALL of the E2 organic product formed when the structure shown below undergoes dehydrohalogenation in alcoholic KOH with heat. Which of the products is formed the most?Draw a structural formula for the product of each SN2 reaction. Where configuration of starting material is given, show the configuration of the product, (CH3)3N + CH3I in the presence of acetone = ?

