Q: A sample of solid aluminum is heated with an electrical coil. If 164 Joules of energy are added to a...
A: Specific heat capacity is the amount of heat in Joules is required by the substance to raise its tem...
Q: Using IR Spectroscopy to Determine the Types of Bonds in a Compound What types of bonds are responsi...
A: For compound A, two important picks, above the 1500 cm-1, are observed, ~3000 cm-1 and ~1700 cm-1 wh...
Q: Draw a stepwise mechanism for the reaction of an alcohol with an isocyanate to form a urethane.
A: The given reaction is,
Q: Consider an octahedral complex MA3B3. How many geometricisomers are expected for this compound? Will...
A: Isomerism-molecules with same molecular formula but different structural formula called as isomers a...
Q: A mixture of A, B, and C was added to a separatory funnel containing CH2Cl2 and 10% aqueous NaOH sol...
A: SOLUTION: Step 1: A separatory funnel is used in liquid-liquid extraction to separate the mixture of...
Q: Naporsyn 0.375 gm is ordered for a patient. Naporsyn is supplied in 250 mg scored tablets. How many ...
A: Since Naporsyn is supplied in 250 mg scored tablets hence it means that in each tablet, there is 250...
Q: The p subshell contains 3 orbitals that have the same energy and shape, but different orientations i...
A: The term quantum numbers refer to a set of numerical values which can give the ideas about the shell...
Q: specify the hybridization at the atoms labeled a-d.For each atom enter one of the following: sp3, ds...
A: The hybridization of the atoms in the first structure can be listed as follows-
Q: What is ∆G° for a redox reaction where 6 moles of electrons are transferred and E° =-2.25 V? (F = 96...
A:
Q: A voltaic cell is based on the two standard half-reactionsCd2+(aq) + 2 e----->Cd(s)Sn2+(aq) + 2 e...
A: Redox reaction- the reaction in which oxidation and reduction takes place simultaneously called as r...
Q: Which of the following has the largest dipole moment? (A) CH3-CEN (B) NF3 CN C=C NC H (С) CHз-CH2NH2...
A: Dipole moment is the measure of the polarity between two atoms that are bonded to each other. It is ...
Q: Draw the structure of C in the following reaction scheme, and show how C can be converted to D by a ...
A:
Q: 50.0 mL of 6.0 M acetic acid reacts with 1.55 g magnesium. what volume of hydrogen gas will be produ...
A: Stoichiometry, the essential technique widely utilized for obtaining product's amount by relating th...
Q: Explain why the pKa of the COOH group of glycine is much lower than the pKa of the COOH of acetic ac...
A: According to the given data the pKa of the -COOH group of glycine is less than pKa of -COOH group of...
Q: Assuming that you start with a 0.0150 M solution of pyridine, set-up a RICE table where x is the cha...
A: This problem is related to equilibrium. Given data: Kb for pyridine =1.70 x 10-9. Kb is the base dis...
Q: During the electrolysis of molten NaI, what reaction occurs at the anode? A) I⁻(l) + e⁻ → I₂⁻(...
A:
Q: Chemistry Question
A: Density of water = 1 g/mL Thus mass of water = 58.6 g Molar mass of CaCl2 = 110.98 g/mol Calculation...
Q: Iron has a density of 7.86 g/cm3 and crystallizes in a body-centered cubic structure at room tempera...
A: In body centered cubic unit cell, the number of atom (Z) is 2
Q: What steps are needed to convert but-1-ene (CH3CH2CH=CH2) to octane [CH3(CH2)6CH3] using a coupling ...
A: Species containing copper-carbon bonds are known as organocuprate reagents. These reagents act as nu...
Q: In the lab, a student needs to determine the van't Hoff factor for a deicer in water. Using the same...
A: The depression in freezing point is given by depression in freezing point = i X Kf X m where i = V...
Q: ABS, a widely produced copolymer used in crash helmets, small appliances, and toys, is formed from t...
A: A copolymer of acrylonitrile, buta-1,3-diene and styrene is called ABS (Acrylonitrile butadiene styr...
Q: Explain this result: Acetic acid (CH3COOH), labeled at its OH oxygen with the uncommon 18O isotope (...
A:
Q: Complete the hydrolysis reaction of the following salts and classify the solution as acidic/basic/ne...
A: The hydrolysis reaction of the salts has to be completed. The solution acidic, basic or neutral has ...
Q: Draw a structure for each of the following: a. m-chloromethylbenzene c. o-nitroaniline ...
A: a. The structure of m-chloromethylbenzene is drawn below.
Q: Propose a mechanism for the formation of fructose-1,6-bisphosphate from dihydroxyacetone phosphate a...
A: Given: Formation of fructose-1,6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-p...
Q: Classify each transformation as substitution, elimination, or addition.
A: Substitution reaction is a reaction in which an atom or a group of atom is replaced by another atom ...
Q: How can 1H NMR distinguish between the compounds in each of the following pairs?
A: NOTE: Since, we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the ques...
Q: What amide(s) will form each of the following amines on treatment with LiAlH4?
A: LiAlH4 is the strong reducing agent. It will reduce the carbonyl group present in amide into CH2 gro...
Q: Choose from the options A-E and explain briefly by illustrations the chemistry behind each answer. P...
A: Since you have posted multiple questions, and have not specified which question you want us to solve...
Q: Please write a balanced equation and indicate the state. HCl(aq) + H2O (l)
A: Since HCl is a strong acid and H2O is neutral compound hence H2O will act as a base and will take pr...
Q: In the sketch below, the red spheres represent protonsand the gray spheres represent neutrons. (a) W...
A: EXPLANATION part a) The give figure in the question clearly depicts that the first reactant nuclei c...
Q: Aspirin is an anti-inflammatory agent because it inhibits the conversion of arachidonic acid to pros...
A:
Q: What is the concentration of H+ ions in a 2.40 M solution of HNO3?
A: Here the given acid is nitric acid (HNO3). It is a strong acid in general. For string acids, the aci...
Q: How is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules...
A: Enantiomers: When two isomers having mirror images but are non-superimposable to each other. Diaster...
Q: I need help what goes in each box
A: Since 1 cm = 107 nm Hence distance in cm X 107 = distance in nm
Q: For each of the following substances, determine the ideal van't Hoff factor when dissolved in water....
A: Van't Hoff factor is basically the number of ions produced by the 1 formula unit of electrolyte salt...
Q: When buta-1,3-diene (CH2=CH−CH=CH2) is treated with HBr, two constitutional isomers are formed, CH3C...
A: Electrophilic addition reaction follows Markovnikov’s rule. According to Markovnikov’s rule, the pos...
Q: Calculate the change in S when one mole of water is heated from 263 to 283 K given the molar capacit...
A:
Q: Methanol, CH3OH(g), is a possible automobile fuel. The alcohol produces energy in a combustion react...
A:
Q: Draw a stepwise mechanism for the following reaction.
A:
Q: Ninhydrin reacts with an amino acid to form a purple-colored compound. Propose a mechanism to accoun...
A: Given,
Q: Classify each amine in the following compounds as 1°, 2°, or 3°.
A: The amines are classified as 1°, 2°, and 3°. If nitrogen atom is been attached to single substituent...
Q: Label the hybridization for each non-hydrogen atom and fill in all lone pairs of electrons for the s...
A:
Q: The molar mass of unknown molecular compound is determined using freezing point depression. The subs...
A: Kf = molal depression constant
Q: When 1 mole of CO(g) reacts with Cl2(g) to form COCl2(g) according to the following equation, 108 kJ...
A: Exothermic reaction-the reaction in which heat is given out or released called as exothermic reactio...
Q: Draw the products of each reaction, and state whether the reaction is faster or slower than a simila...
A:
Q: By taking into account electronegativity differences, draw the products formed by heterolysis of the...
A: Cleavage of bond- In organic chemistry cleavage of bond can be classified as-homolytic and heteroly...
Q: Determine the molality and molarity of a sodium chloride solution prepared by adding 37.60 g of soli...
A: A) Molality is defined as the moles of solute present per Kg of solvent => molality = moles of s...
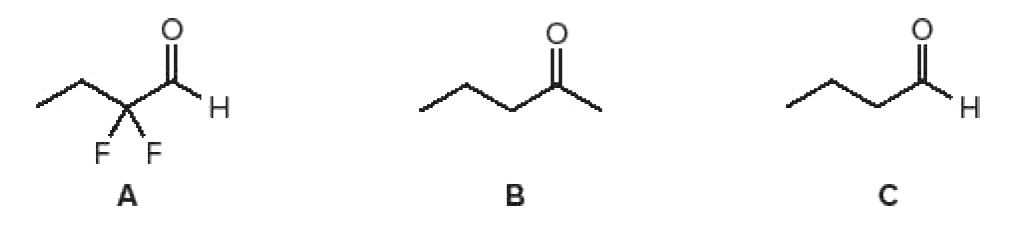
Rank the following carbonyl compounds in order of increasing percentage of hydrate present at equilibrium.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


