Q: Bromine has two abundant isotopes, 79Br (51%) and 81Br (49%) . Chlorine has two abundant isotopes,…
A: A question based on mass spectrum that is to be accomplished.
Q: 7. Calculate the boiling point of an aqueous solution that contains 20.0 mass % ethylene glycol…
A: Boiling point elevation is defined as the increase in the boiling point of a solvent upon the…
Q: Consider the substituted cycloalkene shown. Name substituents using branched alkyl group common…
A:
Q: Use the M+1 signal in the mass spectrum to approximate the number of carbon atoms in the compound.
A: The M+1 peak is caused by the presence of the 13C isotope in the molecule. The proportion of carbon…
Q: What stretching frequencies (in cm-¹) are present in the structure below? 1715 O 1745 1800 CI
A: The stretching frequency of a molecule depends on the neighbour attached to it. When a…
Q: What are the steps in diluting high concentration stock solutions into lower concentration…
A: The formula for dilution is C1V1 = C2V2. Where, C1 is the concentration of the starting solution, V1…
Q: 1. Solve the following queries. Make sure you show full procedure using conversion factors if…
A:
Q: 5. A solution of propylene glycol (antifreeze) in the cars radiator has a freezing point of -20°C.…
A:
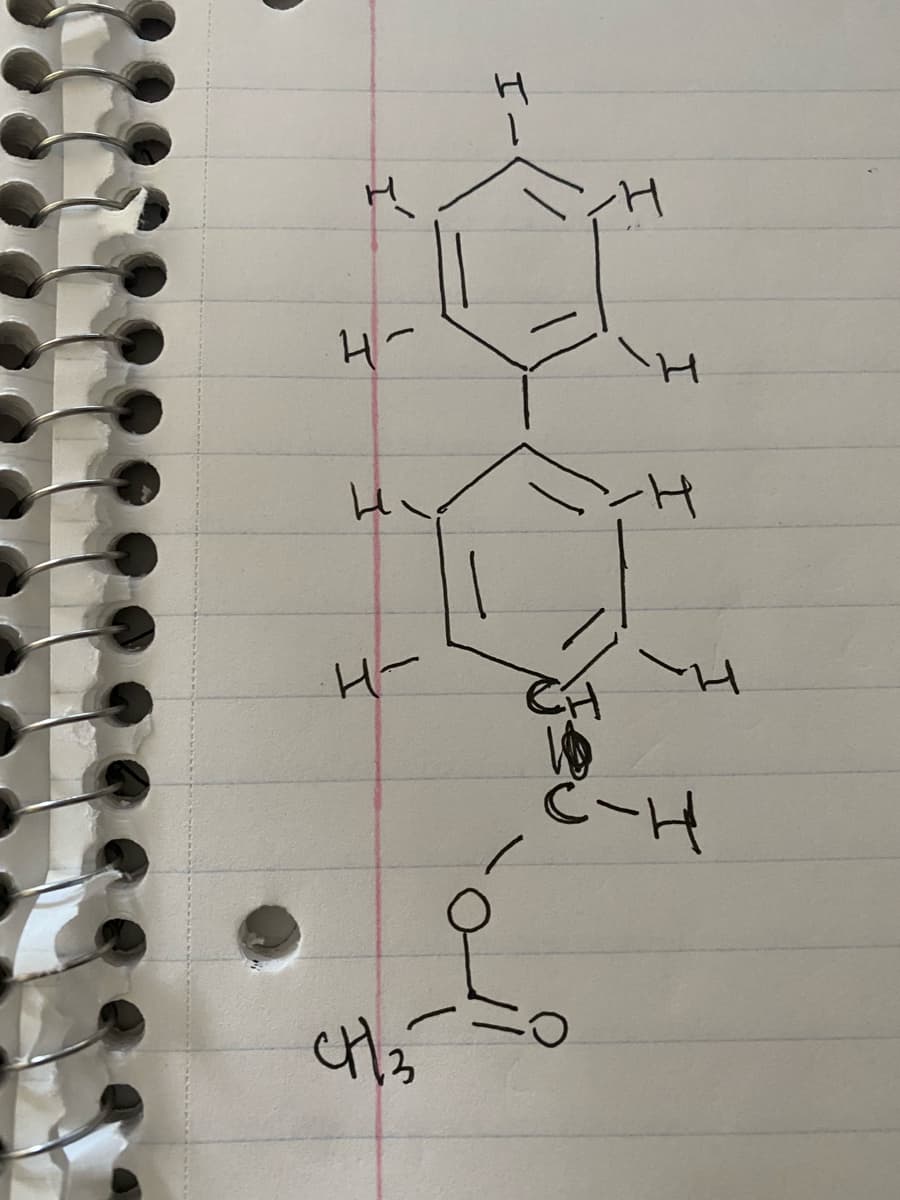
Q: Give the structure that corresponds to the following molecular formula and ¹H NMR spectrum:: CH₁60:8…
A: 1H-nmr spectra provides the hydrogen environment of the unknown compound. The splitting of nmr…
Q: Wine goes bad soon after opening because the ethanol (CH3CH₂OH) in it reacts with oxygen gas (O₂)…
A: Given, mass of oxygen = 9.3 g To calculate : mass of ethanol consumed in the reaction
Q: . Determine the formula, name, and percent composition for the cobalt(II) chloride hydrate given the…
A: Hi there! as there are multiple questions posted. we are solving first question. If you need…
Q: H₂N OH OH OH NH₂ NH₂ H₂N flip OH NH₂ OH OH none of these 0 NH₂ X Note: Only check the boxes of…
A: As we are given that the molecule is flipped by 180°.When we flip the Molecule , it rotate by the…
Q: Assuming octahedral coordination, draw an energy level diagram and fill with electrons. Do this both…
A:
Q: A mixture of 0.05087 mol of C₂H4, 0.08092 mol of N₂, 0.06764 mol of NH3, and 0.0 848 mol of C6H6 is…
A: First , calculate the number of moles of each gas at equilibrium and then their sum, that is total…
Q: Predict the formula for a compound made from X2+ and Y²-. Insert subscripts as needed. formula: XY
A:
Q: ОН
A: The naming of an organic compound can be done with help of rules of international union of pure and…
Q: d calculate RF for the foll.
A: Paper chromatography in analytical chemistry is a technique we have used to identify and separate…
Q: Differentiate reversible and irreversible denaturation. Give 1 example to each denaturation process.
A: A question based on general chemistry that is to be accomplished.
Q: Shown below is the major resonance structure for a molecule. Draw the second best resonance…
A: Given molecule: We have to draw the second-best resonance structure of the molecule.
Q: How do I solve this? The boiling point elevation constant for water is 0.52 C/m. A 1.34 m aqueous…
A: Colligative properties: The colligative properties are those properties of solutions that depend on…
Q: In an aqueous solution of a certain acid the acid is 28.% dissociated and the pH is 1.99. Calculate…
A:
Q: Use the following vapor pressure data to answer the questions: A B Liquid C7H16 C₂H5Br Vapor…
A: Given:
Q: Complete combustion of 4.50 g of a hydrocarbon produced 14.4 g of CO2 and 5.15 g of H2O. What is the…
A: Mass of hydrocarbon = 4.50gm Mass of CO2= 14.4 gm Mass of H2O = 5.15 gm Find empirical formula of…
Q: 2) The graph of weight vs. volume for copper is shown below. What is the volume of a piece of copper…
A: y axis is mass in g and x axis is volume in ml. density = massvolume slope = density
Q: 3. You are designing a hollow fiber unit. The fiber diameter is 800 µm and their length is 30 cm.…
A: Please find the below attachment.
Q: Question: Is the Wurtz reaction still used today? Discuss why or why not. Discuss some industrial…
A: In this question we are going to know about Wurtz reaction, whether the reaction still used today?…
Q: Calculate the volume in milliliters of a 2.5 mol/L silver nitrate solution that contains 50.0 g of…
A:
Q: Step 6: Determine Polarity Is the central atom surrounded by identical electron groups? Circle one:…
A: Here, the central atom is Sulphur, and Hydrogen atom is side atom
Q: A) The vapor pressure of diethyl ether (ether) is 463.57 mm Hg at 25 °C. A nonvolatile,…
A: Here multipart question given A and B
Q: (Intermediate) Reactant OD CX Q H₂B-H Apply Mechanism Hint Solution 6 remaining step(s) can be…
A:
Q: A solution is prepared by mixing 2.77 g of AlCl3 and 22.55 g of water . Calculate the freezing…
A: Given - Mass of AlCl3 = 2.77 g Mass of water = 22.55 g
Q: What will be the major organic product from the dehydration of 2-propanol in the presence of a…
A: Alcohol gives dehydration reactions when they react with a strong acid like H2SO4 or H3PO4 to form…
Q: Give a clear handwritten answer...no need of detailed answer ...please give answer all sub parts
A: "To get remaining sub-parts solved please repost the complete question and mention the sub-parts to…
Q: Molecule NH3 H₂S NI3 CS₂ CC14 110⁰ OF2 SO₂ • Bond Angle(s) 110⁰ trigonal Pyramidal bent ≈1100 ~110⁰…
A: NH3 - 107° - Trigonal pyramidal - polar H2S - 92° - Bent - weakly polar CCl4 - 109.5° -…
Q: In each of the following compounds, a particular atom is indicated with an ar- row. Determine…
A: The atom which are electron deficient are electrophilic in Nature while which have excess electron…
Q: Part I: Hydrocarbon Structures and Names. INSTRUCTIONS: For each row in the table, you are given one…
A: The rules of IUPAC nomenclature are (i) Longest chain rule: The chain containing the principal…
Q: solvent BP = 101.85 OC Table 13.6 Molal Boiling Point Elevation and Freezing Point Depression…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Be sure to answer all parts. Chlorine oxide (CIO), which plays an important role in the depletion of…
A:
Q: 2. Calculate each of the following: d. Mass % of Mn in potassium permanganate
A: We have given the compound potassium permanganate. Formula of potassium permanganate is KMnO4 .…
Q: Molecular Shape of NH3 1 central
A: •Here given molecule is NH3 which is known as ammonia. •Here we are discussing its molecular shape,…
Q: Calculate the number of formula units of calcium phosphate produced when 19.50 grams of calcium…
A: The mass of Calsium chloride reacted with iron (II) phosphate is 19.50 grams We have to Calculate…
Q: What is the product of this?
A: Diels-Alder reaction: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a…
Q: Write the skeletal structure of Ethanol. What is the major intermolecular forever found in this…
A:
Q: In biological cells, the energy released by the oxidation of foods is stored in adenosine…
A: Given=> pH= 7 Temperature(T) = 310 K ∆rH= -20 kJ/mol ∆rG= -31 kJ/mol •Here we have to…
Q: What would the wet gummy bear look like? Draw a microscopic view in the box to the right. Use filled
A: Osmosis: It is defined as the spontaneous movement of solvent molecules through a selectively…
Q: What orbital designation a. n = 3 b. n = 3 would we give to the following sets of quantum numbers?…
A:
Q: Which of the structures have a major peak above 3000 cm-¹? (Select all that apply). OH
A:
Q: Calculate each of the following: a. Mass % of N in dinitrogen trioxide b. Mass % of I in…
A:
Q: Select the correct molecular geometry and bond angles around the carbocation: trigonal pyramidal,…
A: Carbocation is formed by heterolytic bond cleavage.we have to determine the geometry and bond angle…
Q: How will you utilize and apply your learnings in Organic Chemistry? Establish its linkage or…
A: Utilisation and application of learnings in organic chemistry and its linkage to veterinary medicine…
Step by step
Solved in 2 steps with 2 images


