Q: The decomposition of N205 is first-order in N205. If the half-life for the reaction is 18.6 min, how…
A: Since we know that, for different order of reactions, the relationship between the concentration of…
Q: What is the overall order of the reaction if NO2 is a first order reactant and H2 is second order…
A: Order of reactions refers to the power dependence of rate on the concentration of all reactants.…
Q: H2O2(aq) → H0(1) + ½ O2(g) is first order in H2O2 with a rate constant of 1.10×10-3 min-!. If the…
A: We have to find the concentration of H2O2 after passing of 2057 min
Q: 9. At a given temperature, a first-order reaction has a rate constant of 2.5× 10–3 s–1.How long will…
A:
Q: What is the half-life of a first-order reaction with a rate constant of 7.40×10−4 s−1?
A: Given that, Rate constant for a first order reaction K = 7.40×10-4 s-1 half life time period for…
Q: If a reaction is first order with a rate constant of 0.0450 s¯1, how much time is required for 65%…
A: A reaction is define as first order is the rate constant depend upon only one reactant there may be…
Q: The decomposition of XY is second order in XY and has a rate constant of 7.00×10−3 L mol−1 s−1 at a…
A:
Q: Cyclobutane decomposes to ethylene according to the reaction below: C4H8(g) 2C2H4(g) ---> Given that…
A: The given reaction: C4H8 (g) → 2 C2H4 (g)is a first order…
Q: From the reaction data below, determine whether the reaction is first order or second order and…
A: Plot of 1/[reactant] vs time
Q: The thermal decomposition of sulfuryl chloride to sulfur dioxide and chlorine SO,C½(g) 4 so2(g) +…
A: Consider the given thermal decomposition reaction of sulfuryl chloride is as follows; SO2Cl2 g →∆…
Q: The gas phase decomposition of nitrosyl chloride at 400 K NOCI(g)- →NO(g) + ½ Cl2(g) is second order…
A:
Q: The decomposition of N2O5 in solution in carbon tetrachloride is a first-order reaction. 2N2O5 →…
A:
Q: The rate at which a certain drug is eliminated by the body follows first-order kinetics, with a half…
A: The half-life of the first-order reaction does not depend on the initial concentration of the…
Q: (Q90) A second order reaction has an initial concentration of 1.46 M. If the half-life of the…
A: Given: Initial concentration = 1.46 M The half-life of the reaction = 32.8 minutes
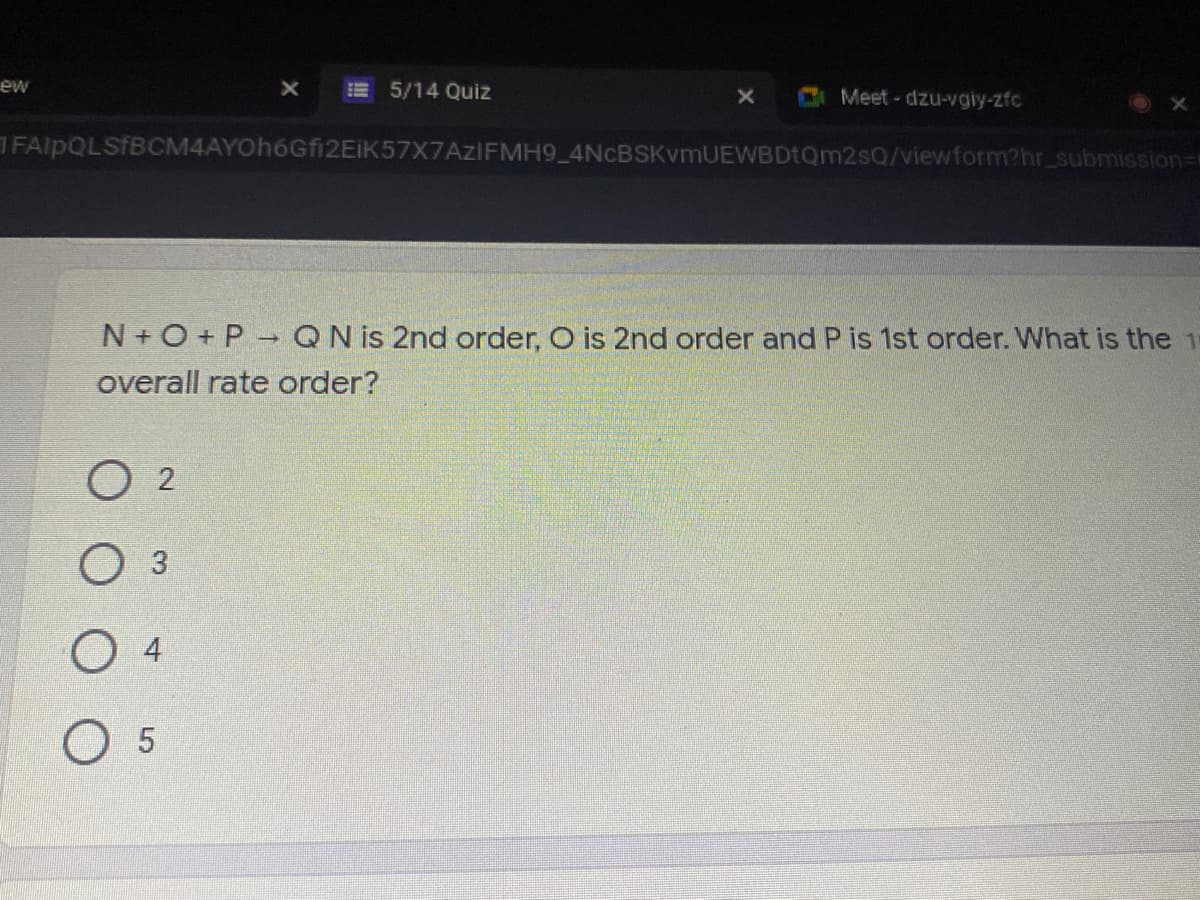
Q: For the reaction 2A+B+2C→D+2E, the reaction is first order in A, zero order in B, and first order in…
A: Answer: The overall reaction order is 2.
Q: Consider the following reaction A B. If the reaction is thought to be ZEROTH order, which of the…
A:
Q: 2. In the presence of excess thiocyanate ion, SCN-, the following reaction is first order in…
A:
Q: A new element is discovered. It decomposes at rate = k[X]^3 If the half life is 4 hours and the…
A: Given If the half life is 4 hours and the concentration is 16 micromolar, what is the rate constant…
Q: 3. The decomposition of a herbicide A in a water at 20° C is a 1st-order reaction with half-life of…
A: Given:Half-life = 2 months.Initial concentration No = 1.6×10-6 MFinal concentration N = 1×10-7 M
Q: In a study of the gas phase decomposition of nitrosyl chloride at 400 K NOCI(g)NO(g) + ½ Cl2(g) the…
A:
Q: The reaction, A2(g) + BC(g) ---> AB(g) +AC(g), is found to be second order in A2 and sero order in…
A: The rate law is an expression showing the relationship of the reaction rate to the concentrations of…
Q: 2) The reaction 2H, + NO ® H,O + 1/2 N, is first order in H, and second order in NO. The overall…
A:
Q: The following reaction is first order in N2O5N2O5: N2O5(g)→NO3(g)+NO2(g)N2O5(g)→NO3(g)+NO2(g) The…
A: What would the rate of the reaction if the reaction were zero order?
Q: rate constant of a first-order process
A: First-order reaction depicts that the rate of reaction is directly proportional to the concentration…
Q: In a study of the gas phase decomposition of nitrosyl chloride at 400 K NOCI(g) NO(g) + ½ Cl2(g) the…
A:
Q: I experimentally determined that the following reaction was second order with respect to NO2. From…
A: Here I am providing an image that will guide you how to deal with this type of problem
Q: Americium-241 is used in smoke detectors. It has a first-order rate constant for radioactive decay…
A: Since the reaction is first order, then the relationship between rate constant and half-life period…
Q: Americium-241 is used in smoke detectors. It has a first-order rate constant for radioactive decay…
A: Given: First order rate constant K=1.6×10-3 yr-1 Rate constant for the decay of iodine k=0.011…
Q: 4) Hydrogen peroxide decays into water and oxygen in a second-order process, H202(aq) > H20(1) + 1/2…
A: Given Initial concentration = 0.100 M Concentration at time t = 0.0425 M Time = 320 seconds
Q: . A reaction that is second-order in one reactant has a rate constant of 2.2 x 10-2 L/(mol • ). If…
A: Given : Rate constant of reaction = 2.2 X 10-2 M-1s-1. Initial concentration of reactant = 0.360 M…
Q: In a study of the decomposition of nitrous oxide at 565 °C N20(g)N2(g) + ½ 02(g) the concentration…
A:
Q: (9, The decomposition of a certain chemical X is found to be second order with respect to [X] and…
A: For Second Order Reaction: k t = 1[A]t-1[A]0 where, k = rate constant = 5.8 × 10-4 M-1 s-1 t = time…
Q: In the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III)…
A:
Q: the half life of a 1st order reaction is found to be 28.25 min. what is the value of the rate…
A: The reaction rates follow direct relationship with the reactant's species involved in first-order…
Q: The decomposition of N2O5 in solution in carbon tetrachloride is a first-order reaction. 2N2O5 →…
A: Given the first-order reaction: 2N2O5 → 4NO2 + O2 The integrated-rate law equation for studying…
Q: Americium-241 is used in smoke detectors. It has a first-order rate constant for radioactive decay…
A: Since it is a first order reaction, then the relationship between rate constant and half-life period…
Q: The decomposition of nitrogen dioxide, 2 NO2(g) 2 NO(g) + O2(g) is second order with a rate constant…
A: Second order rate constant K= 1/t . x /(a-x) Half life t1/2 = 1/a K
Q: In the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III)…
A: As we can see here, SCN- is in excess, this is a type of pseudo 1st order reaction in which one of…
Q: You determine through experimentation that the reaction A⟶P is first order with respect to A. If…
A: Half-life of a first-order reaction = 0.693 / rate constant
Q: NH4NCO(aq)(NH2)2CO(aq) is second order in NH4NCO. In one experiment, when the initial…
A: The integrated rate law for a second-order reaction is 1[A]t-1[A]0 = kt Where [A]t = concentration…
Q: The reaction C2H5Cl →C2H4 + HCl is first order in C2H5Cl. The rate constant is 1.60 x 10-6/s. In an…
A:
Q: The decomposition of N,O5 to NO, and O, is first order, with a rate constant of 4.80 x 1o4/s at…
A: Since the reaction is given first order Hence we can say ln(A0 / A) = Kt where A0 = initial…
Q: The rate constant for the first-order conversion of A to B is 3.33 hr-1. How much time will be…
A: Given K(rate constant) = 3.33/hr Time required for reaching 75% of original value=?
Q: The decomposition of aqueous hydrogen peroxide (H2O2) to gaseous oxygen and water is a 1st order…
A: t1/2 = 0.693/ K
Q: If a reaction is first order with a rate constant of 0.0450 s⁻¹, how much time is required for 45%…
A: We have the first order reaction, with rate constant of 0.0450 S-1 . We have to predict the time…
Q: The thermal decomposition of sulfuryl chloride to sulfur dioxide and chlorine SOCl(g) SO(g) + Cl(g)…
A: The thermal decomposition of sulfuryl chloride takes place into sulfur dioxide and chlorine as…
Q: In a particular experiment, it was found that the concentration of N,0, in liquid bromine varied…
A:
Q: The half-life of a first-order reaction is found to be 28.25 min.What is the value of the rate…
A:
Q: What is the half life of a picloram if upon plotting the experimental data it is linear and 1st…
A: Rate constant k = 1.7 × 10-2 / day We need to find half life.
Q: If a reaction is first order with a rate constant of 0.0450 s⁻¹, how much time is required for 65%…
A: Given : First order rate constant = 0.0450 s⁻¹ how much time is required for 65% of the initial…
Step by step
Solved in 2 steps

- Instantaneous rates for the reaction of hydroxide ion with Cv+ can be determined from the slope of the curve in Figure 11.3 at various concentrations. They are (1) At 4.0 105 mol/L, rate = 12.3 107 mol L1 s1 (2) At 3.0 105 mol/L, rate = 9.25 107 mol L1 s1 (3) At 2.0 105 mol/L, rate = 6.16 107 mol L1 s1 (4) At 1.5 105 mol/L, rate = 4.60 107 mol L1 s1 (5) At 1.0 105 mol/L, rate = 3.09 107 mol L1 s1 (a) What is the relationship between the rates in (1) and (3)? Between (2) and (4)? Between (3) and (5)? (b) What is the relationship between the concentrations in each of these cases? (c) Is the rate of the reaction proportional to the concentration of Cv+? Explain your answer.The first-order rate constant, k1, for the decomposition of ampicillin at pH 5.8 and 35?C is k1 = 2 × 10-7 sec-1. The solubility of ampicillin is 1.1 g/100 mL. If it is desired to prepare a suspension of the drug containing 2.5 g/100 mL, calculate the zero-order rate constant, k0, and the shelf-life, that is, the time in days required for the drug to decompose to 90% of its original concentration (at 35?C) in solution. Note: 100 mL = 1 deciliter = 1 dL.Combination # [IO3-]0 [H+]0 [I-]0 average dt(sec) Initial rate, M s-1 kR (including units) 1 0.010 0.00002 0.10 18.1 0.00055 2 0.020 0.00002 0.10 9.1 0.0022 3 0.010 0.00002 0.20 5.1 0.0020 4 0.010 0.00002 0.10 5.1 0.0020 1. Determine the orders x,y and z 2. Calculatethe rate constant, kR and average kR for combimnations 1-4 3. Write the experimenally determined rate law Rate =kR[IO3-]x [I-]y [H+]z
- Using the data in the table, determine the rate constant of the reaction and select the appropriate units. A+2B⟶C+D Trial [?] (?) [?] (?) Rate (M/s) 1 0.360 0.290 0.0144 2 0.360 0.580 0.0144 3 0.720 0.290 0.0576 k=A + 3B + 2C --> D + 2E Determine the rate law and rate constant using the experimental data below [A] (M) [B] (M) [C] (M) Rate (M/sec) Exp. 1 0.100 5.00 x 10-4 1.00 x 10-2 0.137 Exp. 2 0.100 1.00 x 10-3 1.00 x 10-2 0.268 Exp. 3 0.200 1.00 x 10-3 1.00 x 10-2 0.542 Exp. 4 0.400 1.00 x 10-3 2.00 x 10-2 1.084Using the data in the table, determine the rate constant of the reaction and select the appropriate units. A+2B⟶C+D Trial [?] (?) [?] (?) Rate (M/s) 1 0.340 0.210 0.0204 2 0.340 0.420 0.0204 3 0.680 0.210 0.0816 ?=
- what is the activation energy? rate = r = k [I-]^1 [S2O82-]^1 Mixtures: Temperatures (celsius): Initial Rates: 1 21.9 6.45 * 10-4 2 38.9 3.54 * 10-4 3 2.4 4.35 * 10-4The equilibrium NH3(aq) + H2O(l) ↔NH4+(aq) + OH−(aq) at 25 °C is subjected to a temperature jump which slightly increases the concentration of NH4+(aq) and OH−(aq). The measured relaxation time is 7.61 ns. The equilibrium constant for the system is 1.78 × 10−5 at 25 °C, and the equilibrium concentration of NH3(aq) is 0.15 mol dm−3. (a) Calculate the rate constant for the forward step. kf = _____________. Just value in 3 sig. fig., normal or exponential format, e.g. type in 1.16E6 meaning 1.16 x 106, must use capital E here. Choose a unit in the next question, must be in one of those.The rate constant for the fi rst-order decomposition of N2O5 in the reaction 2 N2 O5(g) → 4 NO2(g) + O2(g) with v = kr[N2O5] is kr = 3.38 x 10-5 s-1 at 25 oC. What is the ha lf- life of N2O5? What w ill be the total pressure, init ial ly 78.4 kPa for the pure N2O5 vapour, (a) 5.0 s, (b) 5.0 min after init iation of the reaction?
- Given the following data, determine the rate law and calculate K Experiment [NO] (M) [Cl2] (M) Rate (M/s) 1 0.0300 0.0100 3.4 x 10-4 2 0.0150 0.0100 8.5 x 10-5 3 0.0150 0.0400 3.4 x 10-4 the units on K are M-2s-1. You should enter the answer without units to 2 sig figs.Given: 2NO2 --> 2NO + O2 [NO2]o (M) Rate (M/s) 0.01 7.1 x 10-5 0.02 28.1 x 10-5 a) Give the rate law and overall order. b) What is [NO2] at 125 s if [NO2]o = 0.015 M?Use this data to determine the value of the rate constant. Expt 1 [Cl2] [Br2] Rate [ClBr]/min 1 0.011 0.025 0.0028 2 0.033 0.025 0.0252 3 0.033 0.100 0.0252 4 0.011 0.050 0.0028 Question 3 options: 0.056 / min 23 / min 30 / min 1.1 / min 925 / min 102 / min 2.5 / min 5.1 / min 463 /min 44.8 / min 10.2 / min



