Q: 100- 90 80 Золотовуразрушена Мадурда аградулатуратнових 70- 60 50 40- 30 3500 3000 2500 2000 про…
A: The question is based on the concept of IR spectroscopy. We need to analyse the spectra and assign…
Q: ¡Draw the condensed or line-angle structural formula for the ester formed in each of the following…
A: To draw the product of the given organic reaction mechanism.
Q: Write the basic equilibrium equation for HS⁻. Be sure to include the proper phases for all species…
A:
Q: Given AH = -19.4 kJ/mol and AS° = +92.3 J/mol K, determine AG° for the phase change CO₂(g) → CO₂(aq)…
A: The question is based on the concept of chemical thermodynamics. we need to calculate free energy…
Q: 2. Draw the structures and explain why CH3CH₂O and CH3CO₂ are good nucleophiles but CH3SO3, water,…
A: Nucleophile is an electron of rich species due to lone pair of electron or negative charge on that…
Q: In the reaction, H₂PO4+HASO4²- involved in the equilibrium? Select one: O a. H₂PO4, HPO4²- O b.…
A: According to Bronsted-Lowry acid base theory, an acid is proton donor and base is a proton acceptor.
Q: 1. Calculate AG in two different ways for the following dimerization at 0°C at standard pressure for…
A:
Q: Propose the reaction product (major) for the following reaction. OH но T DMSO, DCC Et N, relfux
A:
Q: What are responsible for the high electrical conductivity of metals? delocalized positive ions…
A: Higher the electronegativity higher the polarity of bonds. electronegativity of given elements is…
Q: What is K for a reaction if AGº =-282.0 kJ/mol at 25 °C (or 298 K)? (R = 8.314 J/mol · K)
A:
Q: What is the product of each of the following reactions? ▾ Part A ОН ОН 2. HCI
A: We have been provided with reactants if a chemical reaction and we have to find out it's product.…
Q: 출 6.51 1 7.253 6.582 6.580 6.579 6.577 6.575 6.462 6.461 6.459 6.457 4.578 2.263 2.261 2.260
A: The given spectra is H1-NMR. This spectroscopy is used to identify the position of equivalents and…
Q: Starch and glycogen are both polymers of which sugar? O a. a-D-glucose O b. fructose O C. lactose O…
A:
Q: Determine the pH of a solution of aspirin (acetylsalicylic acid, HC,H,O4) by constructing an ICE…
A: Given: Mass of aspirin = 652 mg Molar mass of aspirin = 180 g/mol Volume of solution = 237 mL
Q: Fill in the missing information: Symbol Atomic # Mass Number # Protons # Electrons #Neutrons Charge…
A: Since, For the given notation of the element, Number of proton = Atomic number Number of neutron =…
Q: At 560 K, AG° = -21.5 kJ/mol for the reaction A (g) + 2 B (g) → 3C (g). If the partial pressures of…
A:
Q: Question 1 11.08 g of liquid methanol (CH3OH) is burned in excess oxygen gas according to the…
A:
Q: CH₂ 2-acetyl pyridine Basic condition Nitrobenzaldehdye O₂N- Addition product OH ON Condensation…
A:
Q: 3 of stion us page Which is the stronger base if the equilibrium lies to the right? CH₂ OH O a. Il O…
A:
Q: Explain the purpose of the (1) Dowex-50X2-100 ion exchange resin and (2) K2CO3 in the Fischer…
A: In the Fischer esterification experiment, both Dowex-50X2-100 ion exchange resin and K2CO3 serve…
Q: Suppose that a catalyst lowers the activation barrier of a reaction from 124 kJ mol−1kJ mol−1 to 53…
A: The question is based on the concept of chemical kinetics. we need to calculate the rate factor…
Q: The compounds listed have very similar molar masses. Which has the strongest intermolecular forces?…
A: In polar molecules containing F, O, N, strongest intermolecular forces is Hydrogen bonding.
Q: Complete the balanced molecular reaction for the following weak acid with a strong base. Be sure to…
A: Since, Balanced reaction means that both side number of atom present in equal number. Thus,
Q: =O + H₂- Pt
A:
Q: Part A Which of these compounds is a secondary alcohol? ►View Available Hint(s) нс-он нс- нс OH…
A:
Q: what is the Zeff value experienced by a phosphorus valence electron. Assume that the atom is in its…
A: The electrons which are present in the outermost shell of any atom is called valence electrons. The…
Q: Determine the pH of a solution by constructing a BCA table, constructing an ICE table, writing the…
A: Answer: When acid and base solutions are mixed, they react with each-other to form salt and water…
Q: Draw the skeletal (line-bond) structure of 4,4-dimethylhex-2-ene.
A: Line bond structure, also known as line-angle formula, is a way of representing the molecular…
Q: Draw the major products of this reaction. Ignore stereochemistry. Ignore inorganic byproducts. one…
A: When an alkene reacts with ozone in presence of Zn, HOAc, the double bond of the alkene will cleave…
Q: Consider the reaction: O ICI(g) + Cl₂(g) → ICl3(s). The AG (f) of ICI(g) is -5.5 kJ/mol and the…
A:
Q: In the titration of 230.0 mL of 0.4000 M HONH₂ with 0.2000 M HBr, how many mL of HBr are required to…
A:
Q: Propose an efficient synthesis for the following transformation: 8-6 The transformation above can be…
A:
Q: Please give me answers in 5min I will give you like sure
A: In the reactant part Acid anhydride is given. It a functional group consisting of two acyl groups…
Q: ) Is this more likely to occur at low analyte concentrations or high analyte concentrations? Explain…
A: Stray light (spectral purity) gives potentially rise to incorrect absorbance measurements.
Q: The primary structure of a protein is given by what? O a. The number of different functional groups…
A: There are four different types of structure of proteins i.e, primary, secondary, tertiary and…
Q: Calculate the amount of heat needed to met 77,0 g of sold ethanol (CH,CH,OH) and bring it to a…
A:
Q: Draw the structure of a. 1,3,5-trimethylphenol b. 4-ethyl-2,6-difluorotoluene
A: In the given question, we have to draw the structure of given compounds. The Rules are as follows:-…
Q: A base is found to have a Kb = 6.38 at 25 °C. What is the pKa (to two decimal places) of the…
A: Kb =6.38 at 25 0C We need to calculate the pKa of the conjugate acid.
Q: Why was a plastic coffee cup used as a reaction vessel? What error might have been introduced into…
A: The question is based on the concept of calorimetry . we need to explain the reasoning for using…
Q: At what temperature, in °C, is a certain reaction at equilibrium if AH = +86.7 kJ/mol and AS =…
A: Enthalpy change (∆H) = +86.7 kJ/mol Entropy change (∆S) = +170.2 J/mol.K = +(170.2/1000) kJ/mol.K =…
Q: 4 A short RNA segment is depicted below. Identify the circled functional group on the 5' end of the…
A: 1. Phosphate hence answer a 2. Nucleotide hence answer d
Q: A solution is prepared by adding 0.27 mol of lead(II) nitrate, Pb(NO3)2, to 0.932 L of water. If the…
A: The question is based on the concept of chemical equilibrium and salt hydrolysis. we need to…
Q: Water at 65°F and 20 psia is mixed steadily with saturated water vapor at 20 psia in an insulated…
A: Water temperature: T_cold = 65°F Water pressure: P = 20 psia Saturated water vapor pressure:…
Q: Calculate the pH for each of the cases in the titration of 35.0 mL of 0.240 M KOH(aq) with 0.240 M…
A:
Q: ter the solubility expression for Mg3(PO4)₂(s). sp =
A: The solubility product constant (Ksp) is a equilibrium constant for a solid substance dissolved in…
Q: Predict the product for the following reaction sequence. Br Mg/ether (A) 2,2-dimethylpropanoic acid…
A:
Q: Determine the functionality of the following monomer CH2 –NH2 | CH₂ || HOOC–CH2–CH2–C–CH2−C=CH, in a…
A: In this monomer, there are two functional groups present: carboxylic acid (-COOH) and amine (-NH2).
Q: Continue with the last question, if the conjugate system has 8 double bond, Calculate the energy…
A:
Q: Good conductor only when melted ionic crystal O covalent network crystal Ometallic crystal molecular…
A: Electric conductor :- Those substances which allows flow of current through it.
Q: In the laboratory you are given the task of separating Pb²+ and Cu²+ ions in aqueous solution. For…
A: Check the solubility rules for a compound to check whether the compound will formed precipitate or…
Draw the starting materials and mechanism arrows(s) to give the product shown.
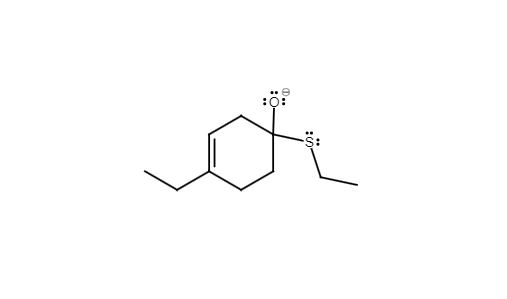
Step by step
Solved in 3 steps with 2 images


