Q: 2) Calculate the molarity of a solution made by dissolving 12.5 grams of Na₂CrO4 in enough water to…
A: The number of moles of solute present in 1 L of solution is called as molarity(M) Or…
Q: The enzyme that catalyzes the following reaction belongs to which class? is-ef O Lyase Isomerase…
A: Enzymes are bio catalysts which catalyse reactions in biological systems. They are highly specific…
Q: 14. Which of the following processes are spontaneous or non-spontaneous? Insert (S) for spontaneous…
A: The process that takes place on its own accord without any external influence is called a…
Q: How many stereoisomers does the following structural formula have? Enter the numerical value. goth…
A: The question is about the possible number of stereoisomers of the given structure.The total number…
Q: calculate the following: 1. volume (in mL) of 355mg of ortho-dichlorobenzene 2. mass (in g) of…
A: To calculate the volume and mass in the given scenarios, we'll use the relationships between mass,…
Q: A student neglected to add sodium carbonate when extracting the tea leaves with hot water. Yet a…
A: As the coffee contains acidis tannin in it and hence it is adviced to add a base like sodium…
Q: Acetylene and oxygen react to form carbon dioxide and water, like this: 2C₂H₂(g)+50₂(g)…
A: Answer:According to Le Chatalier's principle, on changing any parameter of the system that is in…
Q: The electron configuration 4s2 3d¹0 4p² belongs to which of the following? a Ga+ ion a Ge²-ion a P+…
A: Given,The electron configurations are: 4s2 3d10 4p2The electron configuration : 4s2 3d10 4p1
Q: 8. Cyclopentadienone cannot be obtained as its own compound. Any attempts to synthesize it has…
A:
Q: Calculate the force of attraction between a K+ and an O2- ion the centers of which are separated by…
A: Coulomb's law is an expression used to calculate the force of attraction or repulsion between two…
Q: The boiling point of water H₂O is 100.0 °C at 1 atmosphere. A nonvolatile, nonelectrolyte that…
A:
Q: 2. Spiropentane has unusual strain and hybridization. The structure of spiropentane has been…
A: Because of its unique structure, spiropentane is an intriguing chemical with a high degree of…
Q: Consider the reaction: 2 NO₂(g) + Cl₂(g) = 2 NO₂Cl(9) If 2.70 atm of NO,Cl(g) is placed in an empty…
A: The equilibrium reaction is as follows:The initial pressure of = 2.70 atmThe equilibrium pressure…
Q: complete the table Procedure Preheat the hotplate stirrer with an aluminum block to 45–50 °C (do…
A: To complete the table for the given reduction of 4-bromonitrobenzene to 4-bromoaniline.Given…
Q: b. Draw the arrow pushing mechanism for the polycondensation of the following two monomers and the…
A:
Q: te the compression factor, Z for hydrogen sulfide gas at 50.0 bar and 373 K. The van der Waals…
A: Compressibility factor (Z) is a measure of deviation in the behaviour of the properties of the gas…
Q: A diprotic acid, H₂A, has Ka, = 3.5 x 105 and Ka₂ = 9.2 × 10-1⁰. What is the concentration of the…
A:
Q: Write the mass-balance expressions for a solution that is a. 0.30 M in HCN. M = b. 0.29 M in CH3NH2.…
A: Mass-balance equations relate the equilibrium concentrations of various species in a solution to one…
Q: Br e. Draw the cis and trans isomer of the molecule above. f. Draw the chair form of the cis isomer…
A: The cis and trans isomers are called geometric isomers. If both the substituents are present on same…
Q: How do you Identify the unknown substance using the H NMR and CNMR Spectrums
A: Nuclear magnetic resonance (NMR) spectroscopy is a powerful method for identifying unknown…
Q: Calculate the pH of a 0.20 M Na₂CO solution. For CO², Kb₁ = 2.1 x 1 104 and Kb₂ = 2.2 x 10⁹. 3
A: The question is based on the concept of pH of the solution. It is defined as a negative logarithm of…
Q: which is NOT appropriate for lab and why not? chemical resistant goggles, eyeglasses, or safety…
A: In a laboratory work everyone should wear personal protective equipment (PPE) for safety purpose…
Q: Determine the empirical and molecular formulas of each of the following substances. Styrene, a…
A: Empirical formula :-The formula which shows only relative number of atoms of different elements…
Q: The specific heat of a certain type of metal is 0.128 J/(g⋅°C). What is the final temperature if 305…
A:
Q: Calculate the difference in the populations of the two nuclear spin states of H nuclei in a magnetic…
A: Upon the application of an external magnetic field, two nuclear spin states are generated in the…
Q: Suppose 10.0 g of Compound A are consumed in a rection with excess 10.0 g of Compound B that…
A: To solve this problem, Law of Conservation of mass is required. According to this law, Mass of…
Q: 9. Provided is target molecule E and the corresponding synthetic equivalents from a proposed…
A: Given is retrosynthetic analysis of organic compound.
Q: 2. A tablet of Vitamin C weighed 0.306 g and required 15.70 mL of 0.0300 M KIO, solution for…
A: Answer:To measure the ascorbic acid in a vitamin-C tablet it is titrated with Iodate ion in acidic…
Q: What is the mass percentage of Na in Na₂S? Provide an answer to two decimal places.
A:
Q: Which of the following is a conformer of trans-1,2-diethylcyclohexane? 도봉분명 II IV A land IⅡ ① B. I,…
A:
Q: 段階的に解決し、 人工知能を使用せず、 優れた仕事を行います ご支援ありがとうございました SOLVE STEP BY STEP IN DIGITAL FORMAT DON'T USE AI |…
A: Details
Q: complete the following calculations: 1. moles of 0.475 g of benzoic acid 2. moles of 1.25 g of…
A: complete the following calculations: 1) moles of 0.475 g of benzoic acid 2) moles of 1.25 g of…
Q: gas and water (H₂O) vapor. Suppose you have 3.0 mol of C₂H₂ and 2.0 Acetylene (C₂H₂) gas and oxygen…
A: The balanced equation involved:
Q: part E
A: The cis and trans isomers are called geometric isomers. If both the substituents are present on same…
Q: MISSED THIS? Watch KCV 17.5; Read Section 17.5. You can click on the Review link to access the…
A: The solubility product is a measure of the extent of the solubility of a sparingly soluble compound.…
Q: I am trying to find the concentration of glucose after a 1/50 dilution (quenching) in mM. The…
A: Dilution increases the average distance between solvent molecules. Dipole-dipole quenching results…
Q: is first order with respect to A and B. When the initial concentrations are [A] = 1.75 × 10−2 M and…
A: Concentration of A = 1.75×10^-2 M Concentration of B= 2.51×10^-3 M Rate = 2.93×10^-4 M We are going…
Q: 2. A student mixed 50.00 mL of 1.00 M HCl at 20.5 °C with 50.00 mL of 1.00 M NH3 at 20.5 °C. After…
A: The given case involves the neutralization reaction between NH3 and HCl. The balanced reaction…
Q: How would you prepare 250 mL of a 6.0 M NaNO3 solution from a 15 M NaNO3 solution
A:
Q: Consider the titration of 30.0 mL of 0.311 M weak base B (Kb = 1.3 x 10-10) with 0.150 M HI. What is…
A:
Q: For the reaction below, Kc = 1.10 × 10⁻⁴. Note Kc is sometimes called K. What is the equilibrium…
A:
Q: Part A Give typed full explanation A 25.0 mL sample of 0.150 M hydrofluoric acid is rated with…
A: Given,Molarity of hydrofluoric acid ( HF) = 0.150 MVolume of HF = 25.0 oCMolarity of NaOH = 0.150…
Q: Use the data below to determine the rate law of the following reaction A + 2B -> C + D Experiment…
A: The rate of the reaction is directly proportional to the concentration of the reactants. So when the…
Q: From left to right, Identify the hybridization of the C atoms in the following structure.…
A: Steric number = number of lone pair of electrons on the central atom + number of bonds pair to the…
Q: 8. A chem 335 student was tasked with synthesizing a Diels-Alder adduct of maleic anhydride and a…
A: Given question related to the determination of the percent composition of a diene by using gas…
Q: + 17. What is/are the best reagent(s) used for converting following epoxide to the product below? A)…
A:
Q: What is the relationship between the two structures shown? H H CI Br Br CI and same compound…
A: Given are two newman projections.Same compounds have same molecular as well as structural…
Q: HCI, H₂O then basic work-up H₂N. .H
A:
Q: The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an…
A: Answer:Here: are the rate constants at temperatures T1 and T2 respectively.
Q: OH ; 1 Br → Name: 1 OMe COCI Name:1 Name: ooo Name Name:
A: Name the substituents before the parent chain just as with other alkanes. Start the name of the…
Give the major organic product of the reaction.
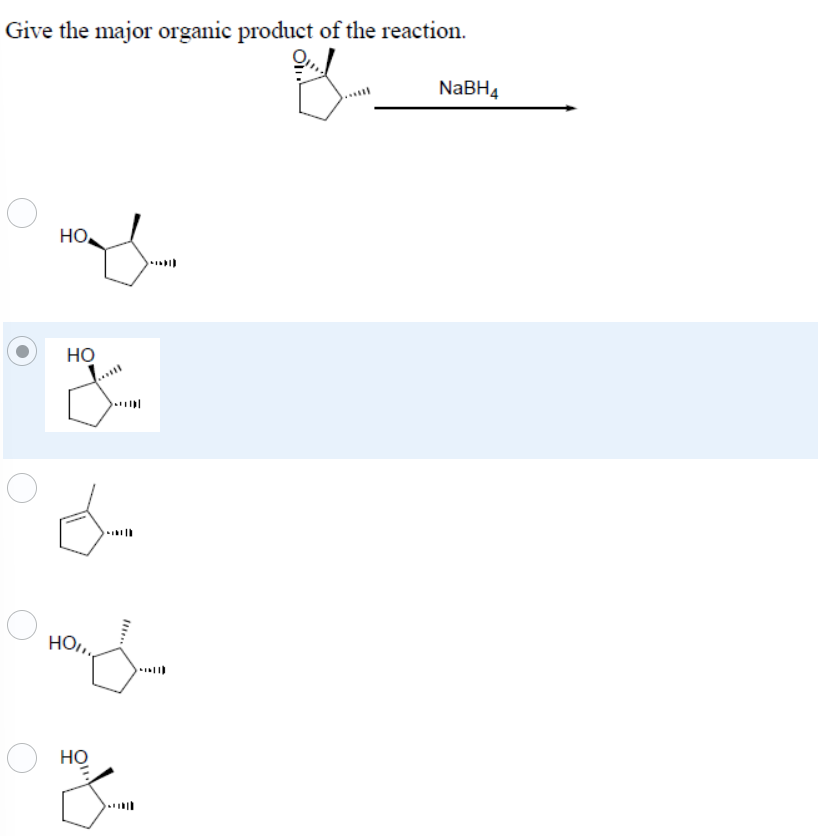
Step by step
Solved in 3 steps with 2 images

- Draw a structural formula for the major organic product of each reaction and specify the most likely mechanism by which each is formed. (g) CH3CH2ONa++CH2=CHCH2ClethanolOn reaction with acid, 4-pyrone is protonated on the carbonyl-group oxygen to give astable cationic product. Using resonance structures and the HĂ¼ckel 4n + 2 rule,explain why the protonated product is so stablegive a primary organic product(s) for each reaction shown

