Q: 125g of a certain seasoning and are told that it contains 96.0 g of salt. what is the percentage of…
A:
Q: The compound shown here is classified as what type of organic compound? HỌC-CH, COCH,CH A) ether B)…
A: Different functional groups in organic chemistry are, Note: Here, R or R' is a alkyl group. Aldehyde…
Q: Of the following, a) melting Ob) sublimation c) boiling is an exothermic process. d) None of these…
A: Answer: Mentioned all the processes are physical processes. Physical processes can be divided into…
Q: give the retrosynthetic analysis which has an aromatic starting compound that gives this product and…
A:
Q: A sample containing 12c, 160, and an unknown isotope is analyzed in a mass spectrometer. As in the…
A:
Q: a: Sketch within a cubic unit cell the following crystallographic directions: [121], [201], [2 1 3),…
A: The crystallographic direction within a cubic unit call can draw by converting the Miller indices…
Q: Is O₂ being created or destroyed by the chemical reaction? If O₂ is being created or destroyed, what…
A: From the information of pressure of a gas vs time we can determine rate of a reaction. We can…
Q: me failoning is an untelanced reaction Use the oxidation number method to balance. PbOx(s) + Sb(a) +…
A: The unbalanced redox reaction is: PbO2(s) + Sb(s) + NaOH(aq) → PbO(s) + NaSbO2(aq) + H2O(l)
Q: (4) CuSO4 is electrolyzed for 10 minutes with 5.1 A current, calculate mass (g) of Cu deposited? B)…
A: The Faraday's first law of electrolysis states that, the amount of compound deposited at electrode…
Q: Calculate the binding energy per nucleon (in J) for 7Be and Be. The atomic masses are 7.016930 u for…
A: Given the two isotopes of beryllium and there atomic mass as well and we are asked to calculate the…
Q: Calculate the enthalpy change for the following reaction, __C₂H4(9) + F₂(g) → _CF4(9) + HF(g) using…
A: Recall the given reaction, _ C2H4 g + _ F2 g → _ CF4 g + _ HF g∆Hf° C2H4 =…
Q: 1. Use the polygon rule to draw the MO energy diagram. 2. Is it aromatic, antiaromatic, or…
A:
Q: What is the ppm concentration of 370g of HCN gas inside 123,490g of air? A. 3000 ppm B. 1500 ppm…
A:
Q: please provide real life applications for following chemical reaction. as well please provide a few…
A: INTRODUCTION: The real life application of the chemical reaction means the use of products that we…
Q: CHALLENGE PROBLEM: Unfortunately, right before the trip you come down with a case of the…
A: Given, Person's body weight = 128 lbs Eyedropper intake = 10 drops per 15 lb body
Q: By definition, atoms are electrically neutral. Is it possible for atoms of two different elements…
A: Atom consist of protons, neutrons and electrons.Protons and neutrons are present in nucleus of an…
Q: Lactated Ringer’s/5% Dextrose solution contains: 6 g/L of Sodium Chloride (NaCl MW 58.5) 3.1 g/L of…
A: Each KCl forma 1 potassium ion, K+. Hence mEq/mL of potassium ion is the same as mEq/mL of KCl…
Q: Convert 2.01 kL into liters
A: These are the following conversion formula: 1kg = 1000 g 1 L = 1000 ml 1 kL = 1000 L
Q: oft انسار Eto Nat
A: Michael addition reaction: alpha , beta unsaturated esters undergo addition reaction when treated…
Q: IV- Fill the final answer: (1) In certain electrolysis, 0.156g of Zn is deposited in cell containing…
A: Electrolysis is proceed by the redox reaction. The redox reaction are defined as the reaction in…
Q: Both OF2 and CS2 molecules contain polar bonds. Explain why one of these molecules is polar and the…
A: A bond formed between two atoms with a difference in electronegativity is called as polar bond. If…
Q: puzzle cube has a volume of 187 mL what is the volume of the cube in in^3
A: Since, 1cm3= 1ml 1 inch = 2.54 cm then,
Q: An electrolytic cell contans 50.0 mL of a 0.152 M solution of FeCl3. A current of 0.775 A is passed…
A:
Q: What are all the main reaction mechanisms in organic chemistry?
A: Every reaction is a conversion of one compound (reactant) to another (product). An equation…
Q: A rock contains 0.697 mg of 206pb for every 1.000 mg of 238 U present. Assuming that no lead was…
A: Given, Mass of 206Pb = 0.697 mg Mass of 238U = [A]0 = 1.000 mg Half life period for 238U = t1/2 =…
Q: The interfacial energy γoil-water of an oil – water interface at 25 oC is 53 mJ/m2. The surface…
A: The ability of one liquid to spread over the other liquid is known using spreading coefficient. For…
Q: a) b) c) d) e) 1) g) h) i) D k) 1) n) MeO it with Jul 8.0 MeO 1.00 8.0 1 L 7.8 I 7.8 ľ K FAK 1 7.6…
A: 1HNMR Spectra given above have signals between 6 to 8 ppm taht is aromatic region .therefore, by…
Q: Match the spectrum with the correct compound and explain
A: Since you have asked multiple questions, we will solve first question for you. For remaining…
Q: You cut six segments of string that are supposedly the same length. The strings you cut were suppose…
A:
Q: The number 37.84637 rounded to 5 significant figures is When 12.9 is subtracted from 64.657, the…
A: we have to complete the given statements
Q: Select the compound with the higher boiling point. HF or HCL
A: Boiling point: The temperature at which the vapor pressure of a liquid becomes equal to the…
Q: 3. Formulate the potential product(s), starting material or reagents of each of the following…
A: We have to write the regents , starting material or reactants in the given reaction sequence.
Q: Give the names of the polymers in the IUPAC system H(-O-CH-O-CO-CHCO-)OH
A:
Q: Give the names of the polymers in the IUPAC system CH2CH(OCO-CH3) (O-ph-OCO-(CH2);CO)
A:
Q: How many significant figures would this calculation have if it were completed: (9.04-8.23 +21.954…
A:
Q: A vibrational energy level of of a diatomic molecule AB has an energy of 1.25x10-19 J, what is its…
A: Given -> Vibrational energy (En) = 1.25 × 10-19 joule Zero point energy (E0) = 2.5 × 10-20 joule…
Q: lase. g/mol
A: Molarity of solution is defined as number of moles of solute dissolved per liter of solution.
Q: 2. Some possible sources of experimental error are listed below. Tell whether the error would make…
A: 1.No effect 2.too high 3.too high 4.too low
Q: The occupation probability of state / with an energy & is given by: exp(-ɛ¡/kµT) q p(ε₁) = where k…
A: Partition function is sum of an exponential function of all the possible energy of the system.
Q: Which of the following aqueous solutions would have the highest boiling point? 1.0 m NaNO, 1.0 m…
A:
Q: Love the following information to answer the next question. The base lithium propandate…
A: Given → Concentration of lithium propanoate = 0.25 mol/L
Q: WRITE THE STRUCTURAL FORMULA OF THE FOLLOWING: 6. 2-METHYL OCTADECANE 7. ICOSANE 8. 4-ETHYL…
A:
Q: 1-chloro-1-methylcyclopropane + H20 1-methylcyclopropane + HCI
A:
Q: ropose a chemical equation related to the reaction. Kremil-S: AlH5MgO5 Vinegar: CH₃COOH
A: Chemical Equation : Symbols are used in chemical equations to denote things like the direction of a…
Q: What is the pressure (in atm) at the critical point for this substance? a) 31.1 Ob) 73.0 Oc) 5.11…
A: Critical point : Critical point of a pure substance is defined as a point at which liquid and gas…
Q: ey tend to gain electrons when forming ions. ey tend to be poor conductors. ey tend to lose…
A: Metal and non metals can be differentiated on the basis of physical and chemical properties.
Q: Givens: 1 mile 5,280 ft 1 inch = 2.54 cm 3 feet 1 yard 454 g = 1lb 946 mL- 1 qt 4 qt 1 gal 12 inches…
A: Concept based on Dimensional analysis.
Q: Krypton consists of several radioactive isotopes, some of which are listed in the following table.…
A:
Q: If the following redox reaction Cd + 2 Ti³+2 Ti²+ + Cd²+ is found to occur spontaneously, identify…
A: The redox reaction is: Cd + 2Ti3+ → 2Ti2+ + Cd2+
Q: The ionization constant of acetic acid (0.05 M) is 1.74x105. Answer 2 and 3 questions: (2) Calculate…
A: Given , Concentration of acetic acid (CH3COOH ) = 0.05 M Ionisation constant =1.74×10-5
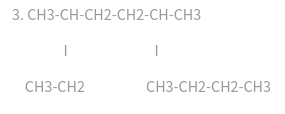
Name each compound according to the IUPAC system:
Step by step
Solved in 3 steps with 1 images


