Q: State the number of neutrons in an atom of each of the following isotopes. (a) hydrogen-1 (b)…
A: Introduction: The atoms of the same element which have the same number of the proton but vary in…
Q: Indicate if each of the following statements is true or false:a. The proton is a negatively charged…
A: a. The proton is a negatively charged particle. The given statement is False. The protons are…
Q: Symbol Protons Neutrons Electrons Mass # Charge 8. 10 8. 8. 18 9. 10 -1 10 10 10 00
A: Atomic number is equal to no. Of protons No. Of protons are equal to no. Of electrons in neutral…
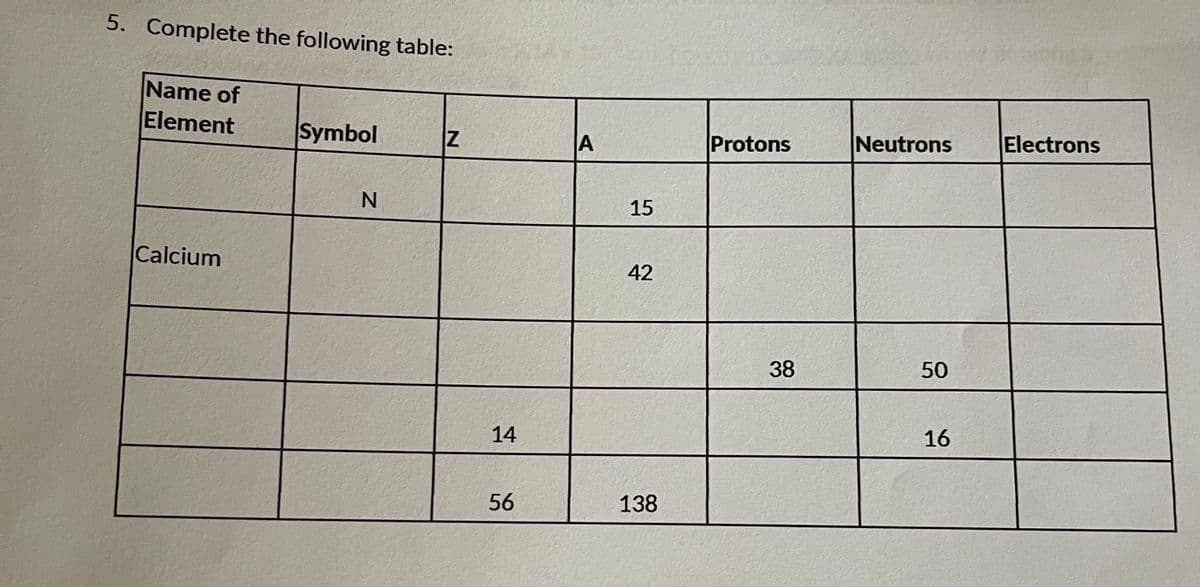
Q: Fill in the missing boxes in the chart below: Nuclear Symbol Neutrons Atomic Number Electrons Mass…
A: An atom is the smallest particle that is capable of independent existence. When an atom loses…
Q: AVhat is the symbol for an ion with: a. 77 protons, 75 electrons, and 116 neutrons b. 117 protons,…
A: a) Ir2+ b) Ts5+
Q: MCQ 20: The number of atoms in the same element has the same number of A. protons B. electrons C.…
A: ATOM The atom is the smallest unit of a chemical element and is the basic unit of matter which…
Q: SUBATOMIC PARTICLES AND ISOTOPES Complete Symbol Atomic Mass Number of Number of Number of Element…
A:
Q: Using your periodic table as a resource, complete the following table for each of the following…
A: A neutral atom contains same no. of protons and electrons. The total no. of protons or electrons…
Q: 2. The symbol °e is a symbol used for a(n) - A. Proton B. Neutron C. Beta Particle D. Gamma Ray
A: NOTE- Since you've posted multiple questions, we're solving the first question for you. If you want…
Q: For each atom described, list the number of electrons, protons and neutrons. a. Neutral Ne 1910 b.…
A:
Q: anion Carbon has an atomic number of 6. The molecule shown is a/an of carbon. isotope cation ion (c)…
A: Given atomic number of C = 6 In Carbon -14 6 protons 8 neutrons The molecule shown is an isotope…
Q: MCQ 4: The atomic number is the number of A. protons (p) B. electrons (e) C. neutrons (n") D.…
A: In an atom, a nucleus contains protons and neutrons in which former is positively charged and latter…
Q: QUESTION 5 Fill in the blanks: Atomic # Element Symbol Mass # number of neutrons number of electrons…
A: atomic mass = no. of proton + no. of neutron (in atom) In a neutral atom , no. of proton =no. of…
Q: Identifying and Labelling Isotopes Complete this table - Fill in the missing information Isotope…
A: Atomic number (Z) :- The number of protons present inside the nucleus of an atom is known as atomic…
Q: Element Atomic Symbol Symbol Be 121 Sb 51 Atomic Mass Number Number 8 Protons Neutrons Electrons 51…
A: -> Atomic number = number of proton = number of electron (for element) -> Atomic mass = proton…
Q: A neutral isotope has 45 protons and 36 electrons identify the element symbol of this isotope in…
A: Isotopes of an element have same number of protons with different number of neutrons in the nucleus.…
Q: Determine the PROTONS, ELECTRONS and NEUTRONS in the following isotopes: 33P 240U p* = p* = e = e =…
A: p+ = atomic number e- = p+ ( if no charge on atom no= atomic mass - atomic number
Q: the correct symbol for the atom or ion with 11 protons, 12 neutrons, and 11 electrons.
A:
Q: Fill in the table. Element Number of Protons Number of Neutrons Number of Electrons Mass Number…
A: Fill in the blanks--
Q: 22 D. Isotopes Nuclear Symbol Protons Neutrons Electrons Ca 20 20 20 24 20 "Ca 20 38
A:
Q: Fill in the following table: Elemental symbol Atomic Number Mass Number # protons 59 27 # neutrons #…
A: Mass number = Number of protons + Number of neutrons Since an atom is electrically neutral Number of…
Q: Write the nuclear symbol for the neutral atom shown. Nucleus nuclear symbol: 4 protons 5 neutrons 4…
A: Given Number of proton = 4 Number of neutron = 5 Number of electron = 4 Nuclear symbol = ?
Q: For each chemical symbol, determine the number of protons and electrons an atom of the element…
A: Element: It is purest form of atom which can't be fragmented into further small spices. Combination…
Q: 9.For each of the following species determine the number of protons, electrons and neutrons: • 'He 2…
A: The number of protons, neutrons, and electrons can be determined as follows- The number of protons…
Q: Complete the sentences with the correct number of each subatomic particle for the indicated atom,…
A: Given here, 57Fe (Iron-57) and Na+ (Sodium cation).we are asked to calculate the number of protons,…
Q: The table below lists the subatomic particles for three atoms: 1, 2, and 3. Which statement is…
A: Isotopes have same number of protons but different number of neutrons. Mass number = number of…
Q: The correct number of protons, neutrons, and electrons for 35Cl-1 is: a. 17 protons, 17…
A: The mass number represent the total number of proton and neutrons in an atomic nucleus , which is…
Q: Complete the following table: Symbol #of Protons #of Electrons # of Neutrons "He 20N 2 27A
A: 1) The ion given is 4He+. Since the atomic number of He is 2. Hence number of protons = atomic…
Q: C. The Atom Name of Element Symbol of Element Atomic Mass Protons Neutrons Electrons Number Number…
A: To calculate the number of electrons, protons, neutrons we need to know the following formula:…
Q: 1. Nuclear Symbol Protons Neutrons Electrons 107 47A 109 47Ag
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: See if you can complete the following table using only what is on this page. Hint: Two of the…
A: The equations which we will be using are 1) Atomic number = number of electrons in neutral atom 2)…
Q: How many protons, neutrons and electrons are there in a neutral atom of the isotope with the nuclear…
A: Atomic number (Z) :- The number of protons present inside the nucleus of an atom is known as its…
Q: many protons, neutrons, and electrons are in the following isotope of copper:"Cu? rotons eutrons…
A: Proton -29 Electron -29
Q: The mass number of an element that has 5 protons, 5 electrons, and 6 neutrons is 11 10 16
A: Mass number = no. of protons + no. of neutrons So, according to question we have , no. of protons =…
Q: Determine the number of protons and the number of neutronsin each isotope.a. 14/7N b. 23/11Na c.…
A: Concept Introduction: An element consists of same type of atoms. An atom is composed of certain…
Q: Identify the element with each atomic number, and give the number of protons and electrons in the…
A: The atomic number (Z) of any atom is the number of protons present in the nucleus of that atom. The…
Q: An atom has 18 neutrons, 15 protons and 16 electrons. What is the correct representation for this…
A: Atomic mass is calculated as the sum of protons and neutrons present inside the nucleus of the atom.…
Q: Element Atomic Atomic Mass Neutrons Electrons Protons Symbol Symbol Number | Number 79BY 35 32 74 Ge…
A: Atomic symbol for any elements is generally represented as, Where, A…
Q: Fill in the nine gaps in this table. Element symbol B. Protons 6. Neutrons 10 Electrons 18 Mass…
A:
Q: See if you can complete the following table using only what is on this page. Hint: Two of the…
A: Atomic number (Z) is the number of protons in an atom. Mass number (A)is the combination of protons…
Q: O An ion with 13 protons, 14 neutrons and charge of 3+ has an atomic number of: A 13 B 14 C 17 D 27.
A: To determine the atomic number.
Q: See if you can complete the following table using only what is on this page. Hint: Two of the…
A: An atom is composed of protons, electrons, and neutrons. The number of protons will be equal to the…
Q: For each of the isotopes as shown, determine the numbers of protons, neutrons, and electrons in a…
A: Atomic Number and Mass Number: Atomic number of an element (Z) = Total number of protons present in…
Q: Element Number of Mass Number of Number of symbol protons number electrons neutrons Са 20 42 20 20…
A: Number of protons present in element = number of electrons Mass number of elemnt = number of protons…
Q: See if you can complete the following table using only what is on this page. Element Symbol Nuclear…
A: Answers to all the blanks in the table is given below.
Q: (a) 63Cu Protons Neutrons Electrons
A: Given :- (a) 63Cu (b) 32S To determine ; total protons,electrons and neutrons in above cases
Q: this page. Element Nuclear Atomic Symbol Mass Number Number Symbol Protons Neutrons Electrons 107 47…
A: Given, The nuclear symbol = 10747Ag The mass number of Fe (Iron) = 59 The no. of elctrons Fe (Iron)…
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

- Refer to the nomenclature flowchart (Figure 3.11) to name each compound.a. KClO3 b. I2O5 c. PbSO4whwn naming a compound do ignore the di,tertri and the iso and juat look at the alphebets of the names, like dimethyl and isopropyl;As what type of molecules would you classify ceramide? Explain your reasoning

