Q: good solvent f
A:
Q: Explain in detail the experimental procedure
A:
Q: Why is it important to do extraction with solvent more number of time than just once?
A: Solvent extraction is the liquid-liquid extraction wherein the compounds are separated based on…
Q: Draw/illustrate the chemical reaction involved in Seliwanoff’s test
A:
Q: What is the melting point of crude and pure acetanilide?
A:
Q: If a lab partner near your work area ingests a dangerous chemical, the correct action on your part…
A: While working in a lab there are various precautions that need to be taken care of. In a lab, there…
Q: 45. In the process of .methanol salicylic acid will produce methyl salicylate." lodoform test…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is retrosynthetic analysis ?
A: In the field of organic chemistry, the most important part is to produce an organic compound. The…
Q: When glucose is used as a positive control on rollers test what evidence indicates the presence of…
A: The presence of Aldehyde group in any substrate can be identified by Tollen's Test.
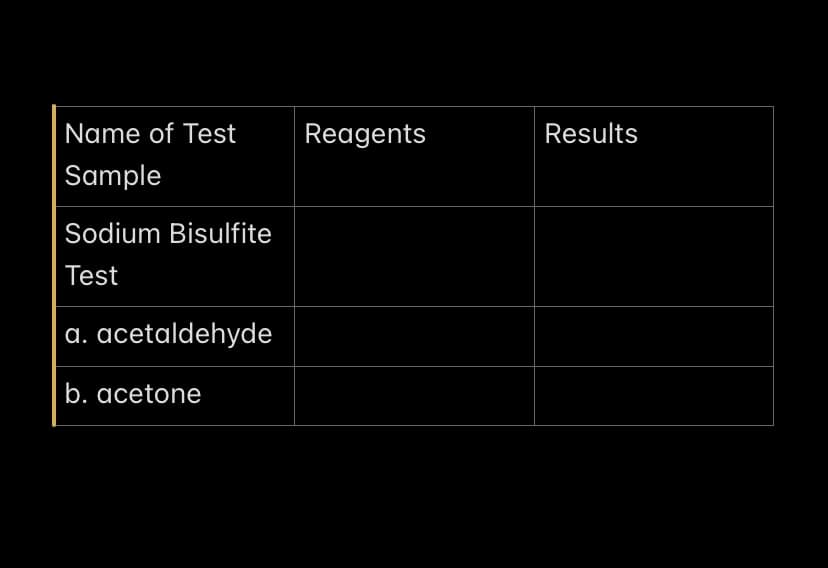
Q: Acetone will give no reaction with sodium bisulfite. Yellow crystals are formed in the iodoform test…
A: Carbon centre in acetone is electrophilic and it can give nucleophilic addition reaction.
Q: each of your extractions uses methylene chloride as the organic solvent. will your organic layer be…
A: Separatory funnel helps in separating the two immiscible solvents. The two layers are formed by…
Q: what is chromyl chloride test ?
A: The test is used to identify the presence of chloride in the sample.
Q: Draw an appropriate structure for (S)-a-Cyanovaleric Acid Upload Choose a File
A: In this question, we want to draw a (S)-a-Cyanovaleric Acid. You can see details Solution below…
Q: Compare and contrast three terms in terms of: a. materials used how the tests are performed b.…
A:
Q: In lab when creating aspirin, why was the use of chilled water necessary to decompose acetic…
A: In lab when creating aspirin, why the use of chilled water was necessary to decompose acetic…
Q: Identify It is used to remove the traces of water that may be present in the organic layer after…
A: An organic layer may be washed with brine ( which is a saturated solution of NaCl ). The purpose of…
Q: Test for acidity, sugar, and alcohol content in alcoholic and non-alcoholic beverages lab report
A: A question based on qualitative analysis, which is to be accomplished.
Q: Ampicillin How was it made using a new synthetic approach? What was the scale of production?
A: Ampicillin is penicillin with a 2-amino-2-phenylacetamido group substituent at position 6 of the…
Q: desribe some important properties of Gels.
A: Gels are defined as a substantially dilute cross-linked system, which exhibits no flow when in the…
Q: What is the purpose of chromic acid test? What is the evidence of positive result?
A:
Q: What is the difference between Benedict's and Barfoed's test?
A: Chemical reagent can be used to distinguish between two compounds and it is also used for…
Q: How do you remove Prussian blue stain?
A: Prussian blue is a dark blue dye that is produced on the oxidation of ferrous ferrocyanide salts.…
Q: Discuss the extraction method for Lemon oil
A: The distillation plant is arranged first Thisconsists of a distillation flask, a basket heater, a…
Q: Introduction about rice husk ash
A: Rice husks are the hard protective coverings of rice grains which are separated from the grains…
Q: Name of Name of Chemical Functional Negative test result/ Positive test result/color color of test…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Write 3 ways that can be used to get rid of emulsion.
A:
Q: limiting reagent
A:
Q: How to draw molecule.
A:
Q: equation of alcohol (ethyl, isopropyl, n-butyl, and tertiary butyl) that give a positive result in…
A: Alcohols containing -CH(OH)CH3 groups and ketones containing -(C=O)CH3 groups give iodoform…
Q: will happen to Acetone, formaldehyde, and benzaldehyde in Fehling's Test? PLEASE PUT REASON OR…
A: The situation of Acetone, formaldehyde, and benzaldehyde in Fehling's Test has to be explained
Q: Give three ways to make sugar dissolve Taster.
A: A solute get dissolved in a solvent if either it dissociates in it or shows special type of…
Q: What is Kemmerer water sampler
A: Kemmerer water sampler :- It is a device that makes it possible to obtain a sample of water from…
Q: which compound is used to remove the permanent hardness of water. draw the structure of that…
A: Permanent hardness of water is due to the presence of sulphates, chlorides, nitrates of magnesium…
Q: How do you remove substances that Give The Same Reaction As Sugar With Nylander’s Test?
A: Solution: The reaction is sugar with Nylander's test is shown below: A black precipitate of metallic…
Q: Discuss the extraction method for Rose oil
A: Rose oil is the essential oil extracted from the petals of various types of rose. Rose ottos are…
Q: What happen to the glowing wood splint when it is inserted inside the test tube?
A: Given, Glowing wood splint is inserted into a test tube Required,…
Q: True or False Aspirin is an acetylation product of m- hydroxybenzoic acid :Select one
A: Acetylation reaction are organic reaction where acetyl functional group (CH3CO) is incorporated in…
Q: Lab question oxidation of aromatic side chain what substance produces the brown/ black spot on the…
A: Given : Oxidation of aromatic side chain. To find : Substance that produce the brown/black spot on…
Q: What are the benefits of howard formula compared to ficher projection formula
A: Introduction : Howarth projections are common way to write the structural formula of sugars in…
Q: Pertinent General Reactions Lucas test: Ferric chloride test:
A: The Lucas test in alcohols is a test to differentiate between primary, secondary, and tertiary…
Q: acetone астА methylacetate
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Give an IUPAC or common name for attached compound.
A: In the IUPAC naming of ester, the alkyl group coming from alcohol act as a substituent while the…
Q: what odor does ethyl alcohol produces in iodoform test?
A: What odor does ethyl alcohol produces in the iodoform test?
Q: Which statement is true? Statement 1: Given equal total volume of organic solvent, single…
A: In this question, we want to select the correct statement. You can see details explanation below.…
Q: Observation or results of the reaction in Lucas test
A: Lucas test is used to distinguish different degree of alcohol such as 1 ° 2° 3° . With the help…
Q: Explain which hydrolyzed component will be positive in the Br2 water test. (Refer to image)
A: Alkenes can be defined as the organic compounds that contain carbon and hydrogen atoms in a…
Q: How does alcohol, water and vinegar remove juice stain
A: In this question, we have to explain the juice stain removing reason by alcohol, water and vinegar.…
Step by step
Solved in 2 steps

- 1. Samples that should not be stored should be prepared/analyzed as soon as possible, especially for measurement…a. hardnessb. Nitritec. Conductivityd. Phenol 2. The following are general sample treatments in pesticide analysis, except…a. Extractionb. Cleaningc. Concentrationd. FilteringYou finish doing an experiemnt with Benzoin. These are the results you get:Boiling start point: 137 ceciusBoiling end point: 130 ceciusVile while empty (with cap on): 15.348Vile with crude prod (with cap on: 15.748 Your starting weight: 1.00115.748 - 15.348 = 0.400 / 1.001 = 0.3996 or 39.96% YieldAnswer the following questions with this information:Initial Mass of impure sample:Mass of recrystallized benzoin:Percent reccovery of Benzoin:Melting pot range of purified benzoin:Literature melting point for benzoin:How to assay expectorant KI in details please
- Determine the percentage Fe in a sample of limonite from the following data:Sample : 0.5000g ; KMnO4 used = 50.00 ml ; 1.000ml of KMnO4 is equivalent 0.005317 g Fe,FeSO4 used = 6.00 ml; 1.000ml FeSO4 is equivalent 0.009200 g FeO ( ans 44.59 %)Indicate solution and the process of proving.Mean, standard deviation, Coefficient variable, and 95% confidence limits of HCl molarity’s * 0.08883M * 0.08743M * 0.08790M

