Name: period: Light Fequency and wavelength Equations: C = Af Units: c: m/s Speed of Light [and all Electromagnetic Spectrum Waves] (c) = 3.0 x 10* m/s Frequency: Hz (1/s) Wavelength (A): meters PROBLEMS: SHOW YOUR WORK AND LABEL YOUR ANSWERS! 1. What is the frequency of green light, which has a wavelength of 4.9 x 10'm? 2. Green light has a frequency of 6.01 x 10“ Hz. What is the wavelength? 3. Calculate the wavelength of radiation with a frequency of 8.0 x 10“ Hz. 4. What is the wavelength of light with a frequency of 7.66 x 10" Hz? 5. A helium laser emits light with a wavelength of 633 nm. What is the frequency of the light? [Hint: First, convert nanometers(nm) into meters by multiplying by 10'] 6. What is the wavelength of X-rays having a frequency of 4.80 x 10" Hz?
Name: period: Light Fequency and wavelength Equations: C = Af Units: c: m/s Speed of Light [and all Electromagnetic Spectrum Waves] (c) = 3.0 x 10* m/s Frequency: Hz (1/s) Wavelength (A): meters PROBLEMS: SHOW YOUR WORK AND LABEL YOUR ANSWERS! 1. What is the frequency of green light, which has a wavelength of 4.9 x 10'm? 2. Green light has a frequency of 6.01 x 10“ Hz. What is the wavelength? 3. Calculate the wavelength of radiation with a frequency of 8.0 x 10“ Hz. 4. What is the wavelength of light with a frequency of 7.66 x 10" Hz? 5. A helium laser emits light with a wavelength of 633 nm. What is the frequency of the light? [Hint: First, convert nanometers(nm) into meters by multiplying by 10'] 6. What is the wavelength of X-rays having a frequency of 4.80 x 10" Hz?
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter6: The Structure Of Atoms
Section6.1: Electromagnetic Radiation
Problem 6.1CYU: (a) Which color in the visible spectrum has the highest frequency? Which has the lowest frequency?...
Related questions
Question
100%
Yeah i dont know how to do any of these
![Name:
period:
Light Fequency and wavelength
Equations: C= Af Units:
Speed of Light [and all Electromagnetic Spectrum Waves] (c) = 3.0 x 10' m/s
C: m/s
Frequency: Hz (1/s) Wavelength (A): meters
PROBLEMS: SHOW YOUR WORK AND LABEL YOUR ANSWERS!
1. What is the frequency of green light, which has a wavelength of 4.9 x 10°m?
2. Green light has a frequency of 6.01 x 10“ Hz. What is the wavelength?
3. Calculate the wavelength of radiation with a frequency of 8.0 x 10“ Hz.
4. What is the wavelength of light with a frequency of 7.66 x 10" Hz?
5. A helium laser emits light with a wavelength of 633 nm. What is the frequency of the light?
[Hint: First, convert nanometers(nm) into meters by multiplying by 10']
6. What is the wavelength of X-rays having a frequency of 4.80 x 10" Hz?
7. A popular radio station broadcasts with a frequency of 94.7MHZ.
What is the wavelength of the broadcast? ( IMHZ=10ʻHZ)
8. Violet light has a wavelength of 4.10 x 10 m. What is the frequency?](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F564c30f2-3cd4-4889-b68e-28588d1c2533%2F59135dda-ed0f-4c20-a59e-89435e8108a8%2Fkmrv3rd_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Name:
period:
Light Fequency and wavelength
Equations: C= Af Units:
Speed of Light [and all Electromagnetic Spectrum Waves] (c) = 3.0 x 10' m/s
C: m/s
Frequency: Hz (1/s) Wavelength (A): meters
PROBLEMS: SHOW YOUR WORK AND LABEL YOUR ANSWERS!
1. What is the frequency of green light, which has a wavelength of 4.9 x 10°m?
2. Green light has a frequency of 6.01 x 10“ Hz. What is the wavelength?
3. Calculate the wavelength of radiation with a frequency of 8.0 x 10“ Hz.
4. What is the wavelength of light with a frequency of 7.66 x 10" Hz?
5. A helium laser emits light with a wavelength of 633 nm. What is the frequency of the light?
[Hint: First, convert nanometers(nm) into meters by multiplying by 10']
6. What is the wavelength of X-rays having a frequency of 4.80 x 10" Hz?
7. A popular radio station broadcasts with a frequency of 94.7MHZ.
What is the wavelength of the broadcast? ( IMHZ=10ʻHZ)
8. Violet light has a wavelength of 4.10 x 10 m. What is the frequency?
Expert Solution
Step 1
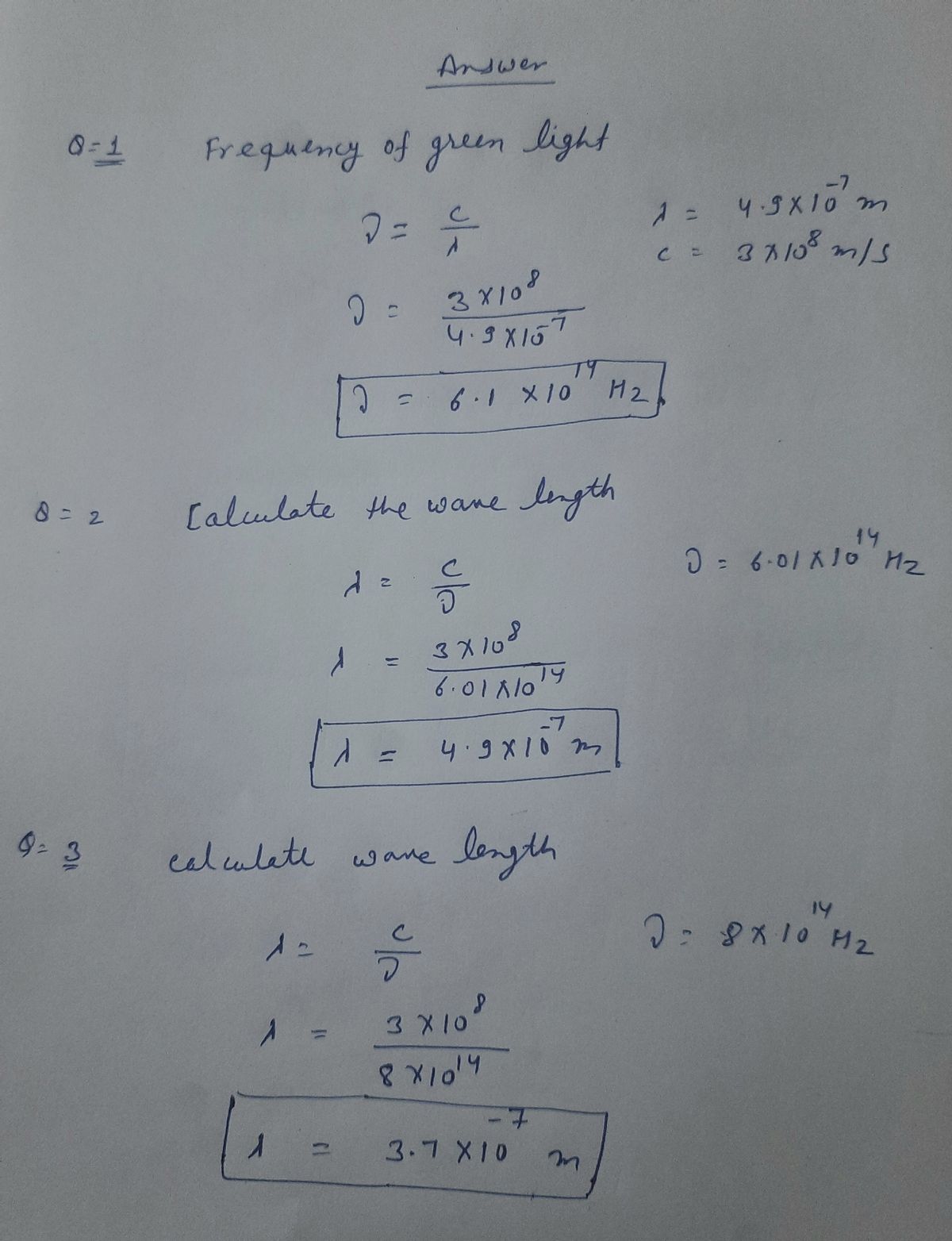
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning