Name Positive ion Negative ion Formula K+ Co32- K2CO3 Potassium carbonate Sodium nitrate NANO3 Calcium bicarbonate Ca(HCO3)2 Chromium(III) hydroxide Cr(OH)3 Lithium phosphate LIЗРО4 Potassium sulfate K2S04
Name Positive ion Negative ion Formula K+ Co32- K2CO3 Potassium carbonate Sodium nitrate NANO3 Calcium bicarbonate Ca(HCO3)2 Chromium(III) hydroxide Cr(OH)3 Lithium phosphate LIЗРО4 Potassium sulfate K2S04
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter15: Molecular Luminescence Spectrometry
Section: Chapter Questions
Problem 15.12QAP: Equations for the chemiluminescence determination of SO2 are given on page 383. Derive an expression...
Related questions
Question
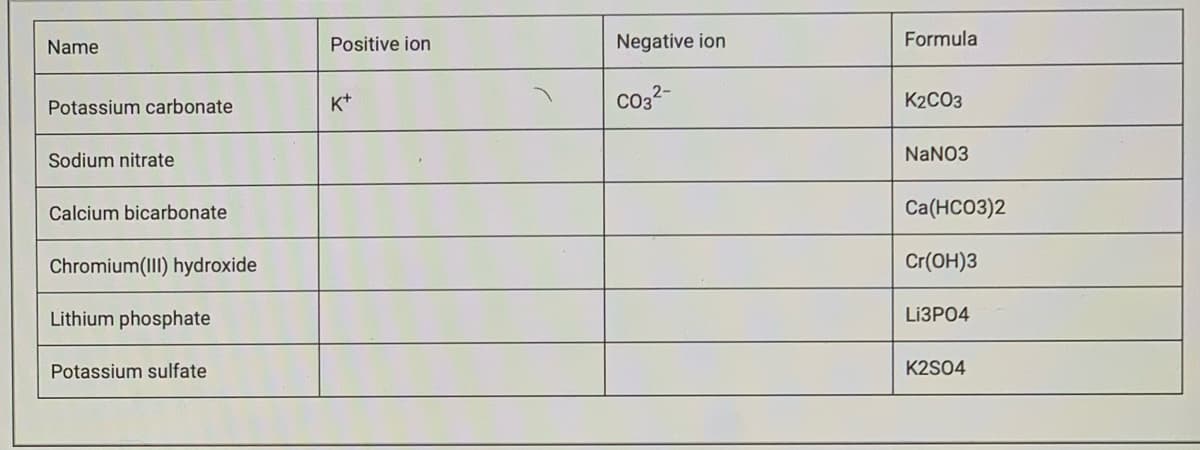
Transcribed Image Text:Name
Positive ion
Negative ion
Formula
K+
Co32-
K2CO3
Potassium carbonate
Sodium nitrate
NANO3
Calcium bicarbonate
Ca(HCO3)2
Chromium(III) hydroxide
Cr(OH)3
Lithium phosphate
LIЗРО4
Potassium sulfate
K2S04
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
