Q: Part 1 of 2 The pressure changes from 760 torr to 1 atm, and the temperature changes from 37 °C to…
A:
Q: Cyclohexane derivatives exist primarily in the most stable of the available chair conformations.…
A: Given question related to writing the most stable of the available chair conformations of the given…
Q: Because a qualitative analysis may often consist of a rather long series of manipulations,…
A: To draw a properly labelled flow chart for the qualitative analysis of Group I cations.
Q: What quantity of moles of H.SO are required to completely react with 7,20 mol of Al according to the…
A: Given reaction is:To find:Moles of H2SO4 :
Q: At 500 K, the reaction below has an equilibrium constant of 3.2 x 100, and the following…
A: Equilibrium constant(Kc) is the ratio of molar concentration of products placed at numerator with…
Q: Nitrogen and hydrogen react to form ammonia, like this: N₂(g) + 3H₂(g) → 2NH₂ (g) Suppose a mixture…
A:
Q: Absorbance / % 8 100 m hm W
A: Given an infrared spectrum of unkown molecule.Determine the functional group present in molecule.
Q: Choose the two Newman chair conformations of ethylcyclohexane: Н Н Et- 5 S □ H Н I Н. T Et Et H EtH…
A: We have to choose the correct option.
Q: what is the chemical formula of fired cooking oil
A: Fried cooking oil does not possess a distinct chemical formula because it is a blend of…
Q: Consider the reaction: C(s) + O₂(g) CO₂(g) Write the equilibrium constant for this reaction in terms…
A: For a reversible reaction, after some point in time the rate of the forward reaction becomes equal…
Q: Question At equilibrium for the reaction in aqueous medium below, the concentrations of A, B, and…
A: At equilibrium,The concentration of A = 0.0032M The concentration of B = 0.0086M The concentration…
Q: You are standardizing a solution of KOH which is approximately 0.4 M. You fill a 50 mL buret with…
A: An acid-base titration is illustrated in the preceding example. It is a titration used to…
Q: 1. A sample of 0.7 M C was placed into a container that produced the following reaction. If the Kc…
A:
Q: An analytical chemist measures the amount of Elements E, and E₂ in four samples of an unknown…
A: Given:X = 10.0 gSampleMass of E1Mass of E2112.2 g13.8 g212.5 g14.4 g36.1 g6.9 g411.7 g13.3 gFind out…
Q: Answer the questions in the table below about this molecule: 10 i HO What is this molecule's…
A: Here, we need to give the chemical formula of a given chemical structure. The chemical formula of a…
Q: You mixed 1.72 g of (L)-limonene ([α] = ±124°) with 4.28 g of (+)-methyl lactate ([α] = ±8.23°) in…
A: Given that:The mass of (L)-limonene = 1.72 gThe specific rotation by (L)-limonene = -124o…
Q: The freezing point of benzene, C6H6, is 5.50 °C at 1 atmosphere. A nonvolatile, nonelectrolyte that…
A: The freezing point of pure benzene = The mass of benzene = 252.1 g = 0.2521 kgThe mass of DDT =…
Q: Part 2 of 3 Check H₂CCH₂CH₂-C-CH₂CH₂CH₂CH, -C-CH₂CH₂CH.C CH₂CH₂ H CH, CH₂CH₂CH, H,C-C-C-H HCH₂CH, X…
A: According to IUPAC nomenclature :Select the longest continuous chain of carbon atoms called as…
Q: 1. Which of the following amines is the weakest base? A. a B. N-CH3 CH3 C. D. -N(CH3)2
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: The freezing point of ethanol, CH3CH₂OH, is -117.300 °C at 1 atmosphere. K(ethanol) = 1.99 °C/m In a…
A: Given:Mass of solute = 10.78 gMass of solvent ethanol = 294.9 gThe solution began to freeze at…
Q: Phosphorus pentachloride decomposes according to the chemical equation PC1, (g) PC13 (g) + Cl₂(g) Ke…
A: Answer:For any given reaction, value of equilibrium constant KC is equal to the ratio of molar…
Q: Provide a synthetic sequence for each of the following transformations. More than one step is…
A: Given are organic reactions. We can prepare given product by performing various Electrophilic…
Q: Hi! I need some help with this question! Consider the following oxidation-reduction reaction:…
A: Given a chemical reaction, answermultiple questions based on this equation.
Q: Philippine Toxic and Hazardous Wastes Act places limits on the quantities of toxic substances that…
A: For the maximum allowable concentration of hexavalent chromium (Cr+6) in mol/L of sewage water, The…
Q: For the following reaction, 6.92 grams of carbon (graphite) are allowed to react with 9.86 grams of…
A: Given,The reaction:carbon (s) + oxygen (g) carbon dioxide (g)mass of carbon = 6.92 gmass of oxygen…
Q: Phosphorus pentachloride decomposes according to the chemical equation PC1, (g) =PC13 (g) + Cl₂(g)…
A: The concentration of PCl5 is:The given reaction is:
Q: [Fe(CN)6]³- + Ferricyanide ion (yellow) HO OH OH L-Ascorbic acid OH '0 [Fe(CN)6] Ferrocyanide ion…
A: To determine the balanced equation for the redox reaction:
Q: At 455 K, this reaction has a Ke value of 0.0390. 2 X(g) + 2 Y(g) = Z(g) Calculate Kp at 455 K. Note…
A: The relationship between Kp and Kc for a reaction: Here, R = the universal gas constant = T =…
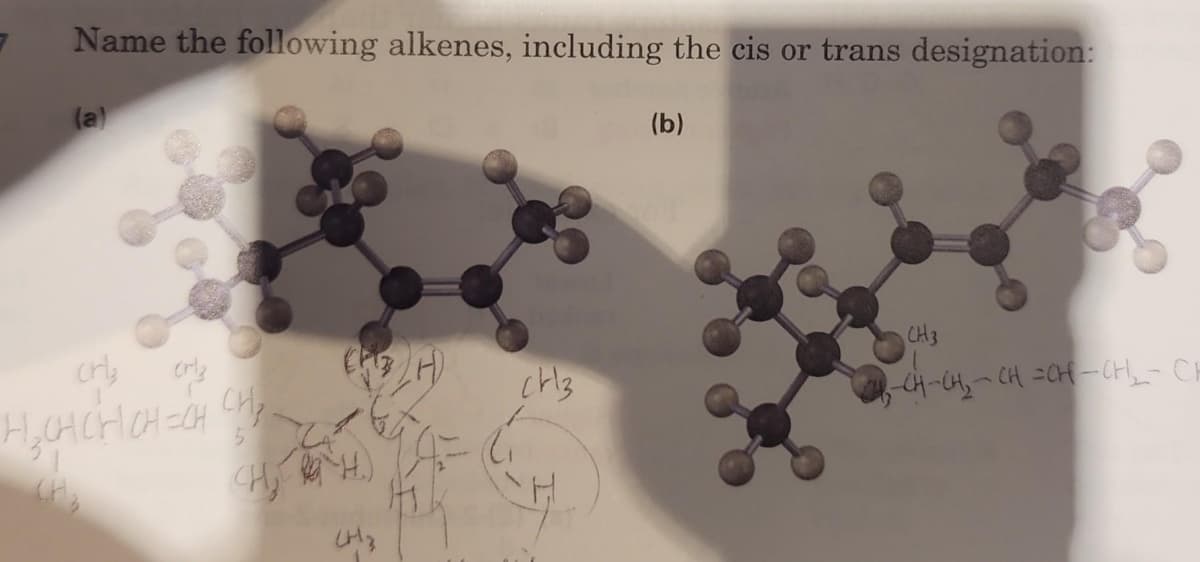
Q: Write an acceptable IUPAC name for the compound below. Include 'cis' and trans' as part of the name…
A: 1). Select the parent chain with the longest possible continuous carbon chain with a maximum number…
Q: Balancing net ionic redox reactions in acidic solution ... when getting to the step where you…
A: In a redox reaction, the reduction reaction of one chemical species and the oxidation reaction of…
Q: Calculate the density of neon gas (in g/L) at 480 mm Hg and 54.3 °C.
A: Ideal gas equation, PV = nRT = P =Where D = Density = M = molar mass R = gas constant = 0.0821…
Q: Draw the mechanism for the formation of compound B. CH3 *C N. F F 0: + Br CH
A: Given is organic reaction.Let's see mechanism for given reaction is next step.
Q: Which of these five-carbon alcohols would you expect to be most water soluble? OH ха В 00 A OH C OH…
A: A type of organic compound that is composed of at least one hydroxyl functional group covalently…
Q: 1. Design a synthesis to prepare the following alcohol. You must use a Grignard reagent as one of…
A: The starting material for preparation of 1-phenylpentan-3-ol is a 3-phenylpropanal and…
Q: A sample of an unknown compound is vaporized at 110. °C. The gas produced has a volume of 1370. mL…
A:
Q: Part 2 out of 5 Draw the missing resonance contributor. CH₂CH₂CH.CH=CHCH₂ CH₂CH₂CH₂CH=CH-CH₂ + Br Br…
A: Given is organic reaction.Given is nucleophilic substitution reaction.In first step we get…
Q: (d) Show how you would make the following transformation: HC CCH₂ CH₂CH₂-
A: The given transformation is .We have to carry out the above transformation.The types of reactions…
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.22 M…
A: A buffer solution is a water solvent-based solution that necessarily contains a mixture of a weak…
Q: Draw the products of the three step reaction sequence shown below. Ignore inorganic byproducts. If…
A: LiAlH4 is a reducing agent, It reduces the nitro (NO2) group to the amine (NH2) group. Amine reacts…
Q: 3. You discovered a new diprotic acid (acid X). You want to characterize the pka's of acid X, so you…
A: A diprotic acid can be described as an acid in which a pair of potential protons are released in an…
Q: Prontosil is an antibacterial sulfa drug. Provide the intermediates, reagents, and product complete…
A: Prontosil is an antibacterial sulfa drug. Given is prontosil synthesis reaction.
Q: Consider an ideal gas mixture of 0.50 g H2 and 4.00 g N2 in a 1.00 L flask at 25 oC. (a) Calculate…
A:
Q: 3. Draw the dash-wedge structure for the following molecules. C₂H₂ C₂H, H C₂H₂ H Br H H CH₂ H
A: To draw dash-wedge structure from Newman representation, first fix the side, from you are viewing…
Q: Consider the following reaction at equilibrium: A (g) 2B (g) Note: Reference the Fundamental…
A: The given equilibrium reaction is .The given table is shown below.We have to complete the…
Q: Enter the number for the mechanism arrows for the 1st step. H. H 1 5 34 H 'Н CI-CI-FeCl3 7 8 9 10 11…
A: The reaction between benzene and FeCl3 is a Lewis acid-catalyzed alkylation reaction. In…
Q: When plotting a graph that measures pH levels of a weak acid at increasing volume of sodium…
A: Given:A graph that measures pH levels of a weak acid at increasing volume of sodium hydroxide.…
Q: Part A A particular balloon can be stretched to a maximum surface area of 1257 cm². The balloon is…
A: Given problemA particular balloon can be stretched to a maximum surface area of 1257 cm². The…
Q: The equilibrium constant, Ke, for the following reaction is 0.0952 at 350. K. CH4 (9) + CCl4 (9)…
A: The value of the equilibrium constant, Kc for the given reaction is 0.0952.The moles of CH4 and CCl4…
Q: Fill in the blanks: Give the name of the organic families shown in the image A A= B= 3 OH B -do-d- C
A: Organic producers rely on natural substances and physical, mechanical, or biologically based farming…
Q: Question 1: draw the curved arrow mechanism for the diels alder cycloadditions shown below (step 1).…
A: According to Alder's endo rule, groups present in outside at diene, groups present in endo at…
Step by step
Solved in 4 steps with 4 images

- Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.Name the alkene below.Use ONLY E/Z designators to indicate stereochemistry.H2C=C=CHCH3Draw and name all of the structural isomers possiblefor an alkyl halide with no branches and the molecularformula C 5 H 10 B r 2
- a. Draw the structures and give the common and systematic names for alkynes with molecular formula C7H12. Ignore stereosiomers. (Hint: There are 14.) b. How many would there be if stereoisomers are included?Assign the following alkene molecules as being cis or trans, E or Z isomers.Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.) Neither E,Z isomerism nor chirality
- Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.)E,Z isomerism but not chiralityPropose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12 c. A diene (dialkene), C5H8 d. A keto alkene, C5H8OOChem HELP... Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form. a) Br2, CCl4 b) Br2, H2O c) OsO4, then aqueous NaHSO3 d) ICl e) mCPBA, then H3O+, H20 f) O3, then Me2S (conformational structure not required) g) BH3:THF, then H2O2, HO- h) D2, Pt i) Hg(OAc)2 in THF-H2O, then NaBH4, HO- j) BD3 :THF, then CH3CO2T
- Use the molecular formula C8H15Br to illustrate thefollowing isomers:1.1.1 positional isomers 1.1.2 (-)-gauche en syn-periplanêre rotamere 1.1.3 E- en Z-alkene 1.1.4 enantiomere (gee ook beskrywers)Please draw the line structures of all of the C8H17+ carbocations that have the carbon-skeleton below. And circle the most stable carbocation of those drawn.Which of the structures below is (4R)-4ethyl-2,4- dimethylcyclohexan-1-one?

