Q: (а) (b) (c) .CI (d) (е)
A: Name of bicyclo compounds are special. Using bicyclo prefix A
Q: Determine which C of the double bond is more substituted C(1) or C(2) for each of the following…
A: The alkene containing a large number of alkyl groups connected to the double bonded carbon atom is…
Q: Drawing the Products of the Chlorination of an Alkane Draw all the constitutional isomers formed by…
A: The isomers are as given below:
Q: Give the systematic (IUPAC) names of the following alkenes. (a) CH2=CH-CH,-CH(CH3)2 (b)…
A: IUPAC rules: Assign the numbering from functional group on parent hydro carbon. write the…
Q: Name of the following Alkenes :
A: Structure of the given molecule:
Q: Give IUPAC name for CH3 | H3C-CH2-C-CH2-C-H…
A: The compound given is
Q: Alkenes have the general formula ____. Select one: A. CnH2n–2 B. CnH2n+2 C. CnH2n D. CnH2n–4
A: Alkenes have the general formula CnH2n hence the answer is option C.
Q: Give the systematic name of the alkene, indicating cis or trans configuration. H. CH3 H. ČH3…
A: Rules for naming alkene is as follows: First, select the continuous longest carbon chain. Numbering…
Q: 1) Name the alkane shov below
A: To find: The name of the alkane.
Q: CH3 CI CH3CH2CHCH;CCH3 (b) CH3 How much is the percentage yield of the chloroalkane in (b). (Hint:…
A:
Q: Give the systematic (IUPAC) names of the following alkenes. (d) CH—С—CH—CH—CH> CH3 (e) (f) (g) CH3
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: General formula of alkenes is O CnH2n O Cn-2Hn O CnH2n+2 O CnH2n-2
A: Alkenes are hydrocarbons that contain carbon carbon double bond former homologous series of Carbon…
Q: C. sd. Name each of the following cyclic hydrocarbons: b. a. CH3 d. f. e. CH3 CH3 H3C
A: Using IUPAC namenclature in GOC.
Q: . CH(CUSCH = CH2 2.
A: [1.] 3,3-dichloroprop-1-ene [2.] 4-bromo-2,5-dimethylhept-2-ene
Q: Classify each alcohol as primary, secondary, or tertiary
A: The given compound:
Q: Which of the following molecules is unsaturated? А. СзН8 В. СНЗОН С. С 5Н10 D. CH4 Hydrocarbons with…
A: The answer with explaination is shown below in detail -
Q: Provide the proper IUPAC name for the alkene shown below (indicate R or S). CH3 "CH,CH3
A:
Q: Give the systematic (IUPAC) names of the following alkenes. CH2“C“CH¬CH“CH2
A: IUPAC nomenclature in organic chemistry In chemistry, IUPAC nomenclature is the official set of…
Q: H3C H2/Pd H3C CH2CH3 ? CH3
A: Given reaction,
Q: Name the following alkene or alkyne: H3C-CEC-CH-CH2-CH2-CH3 ČH-CH3 ČH3
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: name of the following alkene is CH(CH3)=CH-CH(CH3)(CH2CH3)
A: Alkenes are the type of unsaturated hydrocarbons that consist of one or more double bonds between…
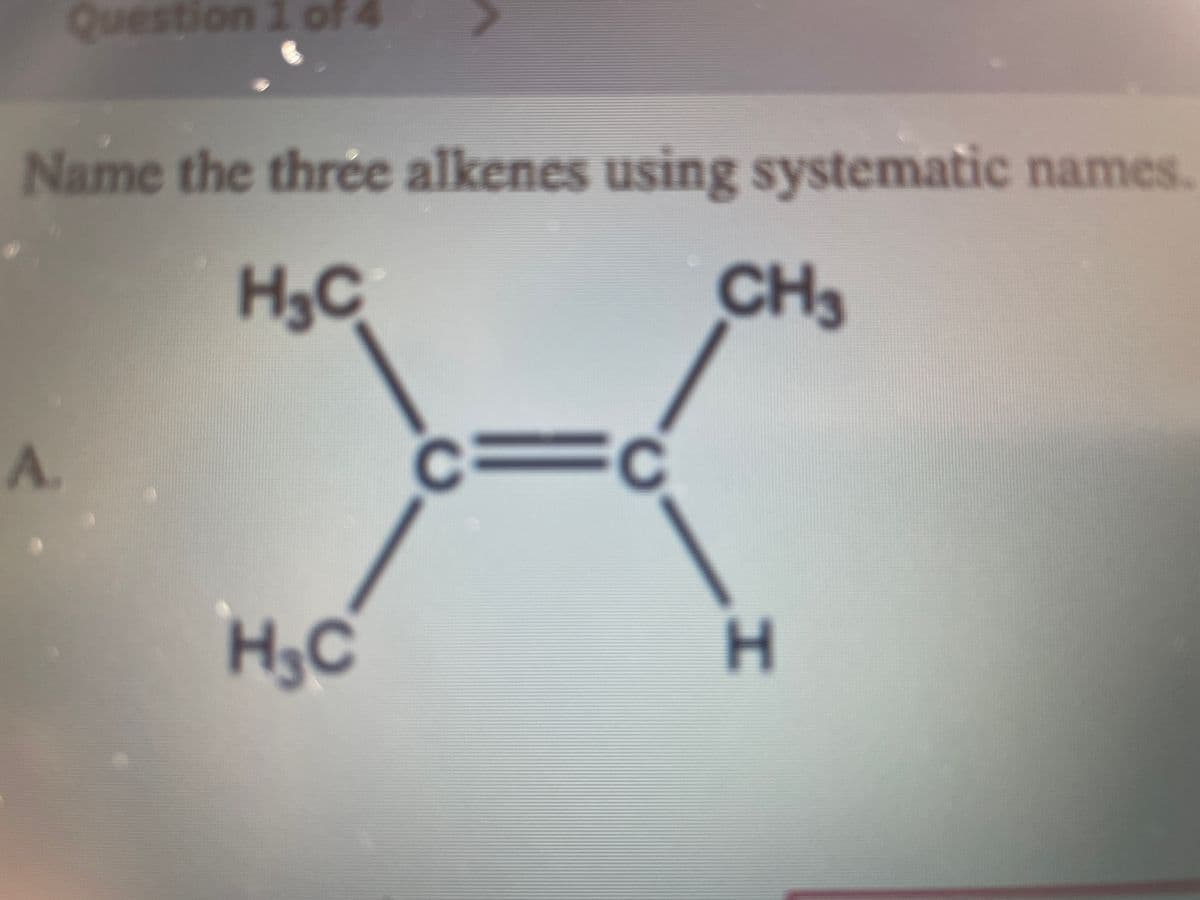
Q: Name the three alkenes using systematic names. H3C CH3 А. H. H3C The name of compound A is: CH2 H2…
A:
Q: The IUPAC name of this disubstituted cyclohexane is trans-1-ethyl-2-methylcyclohexane CH3 CH2CH3…
A:
Q: Draw the structure of the product expected in the following Sonogashira reactions:
A: Note: As per the Q&A guidelines, I am answering only the first three subparts of a question. So…
Q: 3. Give the IUPAC name for each alkene. a. b. Br OCH3 d. С.
A: To write the name of each structure, use the below rules. Identify the parent carbon chain in each…
Q: The IUPAC name for the following alkane is: CH3 CH3 CH-CHCH-CH-CH3
A: Rule:- 1) select the longest continues carbon chain as perent chain 2) number the parent chain by…
Q: 1. Fill in the boxes with the missing reagents or major organic products for the alkene reactions.…
A: Here some organic reactions are given, we have to write the products and also appropriate reagents…
Q: What is the IUPAC name of this alkane? CH 3 CH 3 - C-CH 2- CH CH 3 C H3 C H2 - CH3
A: We have to tell which option is correct
Q: Which molecular formula corresponds to an alkene? A) CSH14 B) C6H14 C) C2H4 D) C1H24 E) C:H6
A:
Q: Name the three alkenes using systematic names. H3C CH3 А. c=c H3C The name of compound A is: CH2 H2…
A: → Rule for Nomenclature of organic compound :-1) Select the longest chain of C-C bond and it is not…
Q: What is the IUPAC name of the following alkanes?
A: IUPAC nomenclature Identify the longest carbon chain. Identify the functional groups present. Give…
Q: The angle of the -C-C-H bond. Br H. C=C CI F 109.5 120° 180° 90° This bicyclic alkanes share only…
A:
Q: Name each alkene. c. CH2=CH-CH-CH,-CH2--CH; CH;-CH CH CH; d. CH;-CH-CH=C-CH; CH2-CH,
A: Given compounds are alkenes. The IUPAC name of alkenes are written by giving “ene” as the suffix.…
Q: CI NH2
A: The systematic structure determination of organic compound is given by IUPAC. The naming of organic…
Q: 2. Write balanced equations for the combustion of the following compounds Decane cyclohexane…
A:
Q: CH3CH=CHCH=CH2
A: There are 5 carbons in the chain labelled from 1 to 5. Hence the parent alkane is pentane there are…
Q: What is the molecular formula of the following alkene? CI o CH14CI o CH13CI o CH15CI o CH11CI
A: The given alkene structure is,
Q: What is the name of this alkene? CH3CH2=CH2
A: Rules for naming the alkene is as follows; Select the continuous longest carbon chain. In this…
Q: Br CH3 Common Name а. OH NO 2 Br b. Common Name c. H3C-CH-CH-CH2-C-NH-CH2-CH3 IUPAC_
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Name the alkene. CH2 H2 C. CH3 H3C H2 name:
A: To solve this problem we have to write the name of the given alkene .
Q: the general molecular formula for a cyclic alkene must be CnH2n False O true
A:
Q: 9. Give a systematic name for each of the following alkanes (a) (b) (c) (d) CH, (e) (1) CH,
A: (a) The given compound is, The systematic name for the above compound is…
Q: 2. Give the IUPAC names of the following alkenes and cycloalkenes. CH2 a. CH,=CH-CH,–CH(CH;)2 b.…
A: " Since you have posted a question with multiple sub-parts , we will solve first three sub-parts for…
Q: Draw a structural formula for an alkene with the indicated molecular formula that gives the compound…
A: Structural formula of alkene can be:
Q: Which alkenes exist as pairs of cis,trans isomers? For each that does, draw the trans isomer. Q.)…
A: The pairs of cis,trans isomers of given alkene can be drawn as Q.) CH3CH=CHBr
Q: General formula of alkenes is a. CnH2n-2 b. CnH2n c. Cn-2Hn d. CnH2n+2
A: Alkene is the unsaturated hydrocarbon having carbon carbon double bond.
Q: ORGANIC CHEMISTRY IUPAC NAME FOR CH3-CH2-CH=CH-CH3
A: The functional group in the above compound is double bond, i.e alkene Since the length of the chain…
Q: what is the most stable alkene?
A: 1) A more substituted alkene is more stable alkene. 2) Conjugated double bonds are more stable than…
Q: Which alkenes exist as pairs of cis,trans isomers? For each that does, draw the trans isomer. Q.)…
A: For cis and trans isomerism, the two substituents on each C should be different. For example: A and…
Q: Name the three alkenes using systematic names. H3C CH3 A. H3C The name of compound A is: CH2 B. CH3…
A:
The name of the compound A is :
can u please explain thanks
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- kindly name the following compound. Topic is benzenes, alkenes, alkynesFor Alkenes, there are four columns, (name/condensed/molecular/sketch), help fill in the following sections for the following 3.Please help. This one question has four different parts. A,B,C is to name each of the following disubstituted benzenes and the last one is name the alkane. Please help with all parts of the question.
- I do not understand how I solve to fugure out the names of these substitutde hydrocarbons.What is the IUPAC name of the compound shown here? (view attatchement) **FIRST TIME YOU ANSWERE THIS WAS WRONG, please help with correct answer. The answer you gave me before was 1,4-dimethylcyclobut-1-ene but is was WRONG . This is what the paragraph also said next to the question "Alkanes are called saturated hydrocarbons because each carbon atom has the maximum number of hydrogen atoms. In contrast, alkenes and alkynes are unsaturated because they contain double or triple bonds that reduce the number of hydrogen atoms in the compound. When naming unsaturated hydrocarbons, a suffix reflects the type of multiple bond in the compound: ene is used for alkenes (double bond), and yne is used for alkynes (triple bond). For cyclic alkenes and alkynes, the ring is numbered such that the double or triple bond is between the first two carbon atoms. For straight-chain molecules, the numbering starts at one end of the chain, so the location of the double or triple bond must be…the subject is naming alkanes. I need to understand when to name something by di, tert... for example, tert-butyl
- Hi, here's my question. Write the condensed structural formulas for two alkenes and one alkyne that all have the molecular formula C6H10.question 3. How many distinct structural (constitutional) isomers exist for C₈H₁₈ that have heptane as their root name (longest carbon chain)?question 57 Draw the structure of the following compound: trans-1-bromo-2-chlorocyclohexane
- Based on the HYDROCARBONS TABLE below, answer the following questions accordingly. All hydrocarbons are clear, colorless liquids at room temperature. Which hydrocarbon ignites with the most luminous flame? - Which hydrocarbon is aromatic? - Which hydrocarbon is actively unsaturated? - Which hydrocarbon is aliphatic saturated? - CHOICES: a.Hydrocarbon A b.Hydrocarbon B c.Hydrocarbon C2-Propylbut-1-ene is an incorrect name under the IUPAC system. What is the correct name for this compound? Hint: Drawthe structure based on the incorrect name and rename it.2,2-dimethyl-6ethylheptane why is incorrect name

