2. Give the IUPAC names of the following alkenes and cycloalkenes. CH2 a. CH,=CH-CH,–CH(CH;)2 b. CH2=CH–CH2-CH=CH2 f. CH;(CH,);-C-CH,CH3 C. CH2 h. g.
2. Give the IUPAC names of the following alkenes and cycloalkenes. CH2 a. CH,=CH-CH,–CH(CH;)2 b. CH2=CH–CH2-CH=CH2 f. CH;(CH,);-C-CH,CH3 C. CH2 h. g.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterL2: Mass Spectrometry
Section: Chapter Questions
Problem 20CTQ
Related questions
Question
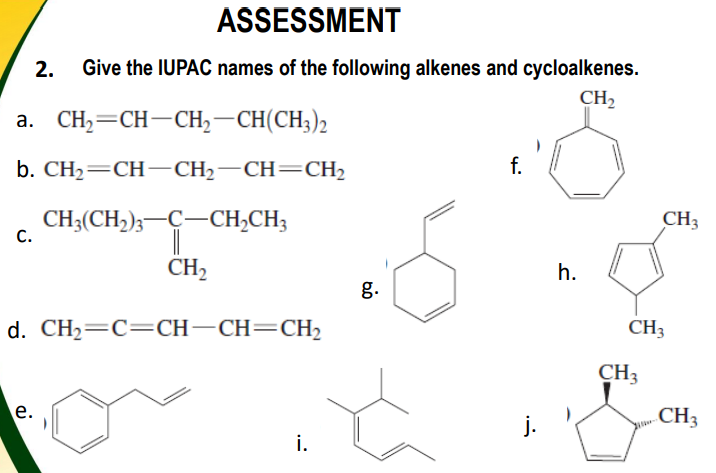
Transcribed Image Text:ASSESSMENT
2.
Give the IUPAC names of the following alkenes and cycloalkenes.
CH,
a. CH2=CH-CH,–CH(CH;)2
b. CH— CH— CH— СH—CH2
f.
CH3(CH,);-C-CH,CH3
CH3
С.
CH2
h.
g.
d. CH2=C=CH-CH=CH2
CH3
CH3
е.
CH3
j.
i.
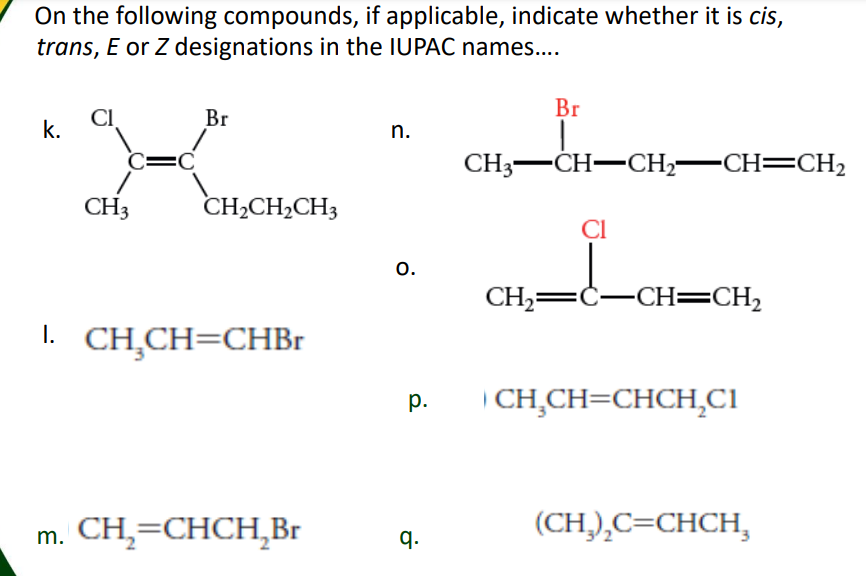
Transcribed Image Text:On the following compounds, if applicable, indicate whether it is cis,
trans, E or Z designations in the IUPAC names..
Br
Br
k.
n.
=D
CH3-CH-CH,-CH=CH2
CH3
CH2CH2CH3
CI
o.
CH,=Ċ-CH==CH,
I. CH,CH=CHB.
| CH,CH=CHCH,CI
р.
m. CH,=CHCH,Br
q.
(CH,),C=CHCH,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning