Q: Which of these is not a conjugate acid-base pair? O NaH2PO4 ; NazHPO4 O HCIO2; HCIO3 O CH3CH2NH;CI;…
A: Acid base conjugate acid base pairs have a difference of one H+ ion.
Q: Please sort the following values from most to least basic Most Basic | [H*] = 3.52 x 10-9 E H] =…
A: Given: The concentration of H+ ion, OH- ion, pH, and pOH values are given. They are to be sorted…
Q: Draw the conjugate base for the acid, HC≡CH. Draw all hydrogens. Remember to include charges and…
A: Conjugate base of any acid is formed when the acid donates 1 proton in the solution.
Q: True or false NH4+ is the conjugate base of NH3.
A: Conjugate base : it is a species having one less Proton that is H+ . Hence conjugate base of NH3 is…
Q: In the following reaction: HCIO4 + H2O → H30* + CIO4 O H20 is the conjugate acid of H30* O CI04 is…
A: Given: HClO4+H2O→H3O++ClO4- To find: conjugate base or conjugate acid.
Q: Find the acid and base and the conjugate acid and base? 1.NaH2PO4+F- ——-> NaHPO4+HF
A:
Q: The conjugate to a strong acid is a O Weak acid Strong acid O Negligible acid Weak base Strong base…
A: Conjugate to a strong acid is weak base. For example let's assume a acid HA HA + H2O⇄A-+ H3O+ where…
Q: Identify the acid, conjugate acid, base, and conjugate base in each reaction below. CH₂OH + NaH…
A: A conjugate acid-base Conjugate acid is formed after a base accept a proton from an acid, On the…
Q: Indicate acids and bases 1) HCl(aq)= H+(Aq) + Cl- (aq) -I know HCl would be an acid, so would that…
A: This is acid base reaction.
Q: Use the below reaction to answer the following question. HCl + H2O H3O+ + Cl- Write down…
A: The reaction given is HCl + H2O <——-> H3O+ + Cl- The substance which donates a proton…
Q: What is the conjugate acid and base for this reaction?
A:
Q: Is H2S an example of a strong or weak acid? What is its conjugate base? Select one: Strong acid, H3s…
A:
Q: Identify the base and conjugate acid in the equation. Explain in detail how you came to your…
A: According to the Brønsted-Lowry concept of an acid-base :- An acid is a proton donor A base is a…
Q: OH- base conjugate acid is: a) H + b) H2O c) OH + d) H3O +
A: When the atom donates it's proton then it is called the conjugate bases of the acid .
Q: Label the acid, base, conjugate acid, and conjugate base in the following reactions: CH3NH2 + Н2О…
A: According to Bronsted-Lowry theroy: An acid is the substance which donate proton and base is the…
Q: In the following unbalanced reaction, which is a conjugate acid-base pair? H3PO3 (aq) + H2O (1) -->…
A:
Q: Given Some acid, choose the weakest acid a (1) butyric acid (ka = 10 5x10-5) (ii) ascorbic acid (ka…
A:
Q: Select the stronger base from each pair of compounds.
A: Basicity of a substance is defined by its power to accept a proton or to release a hydroxide ion.…
Q: Draw the conjugate base of each acid: HBr, HSO,, CH3OH.
A: To find: The conjugate base of given acids
Q: KNO2 (aq)+ H2SO4 (aq) -> Complete the balanced chemical equation for the following reaction between…
A: Given reaction: KNO2 + H2SO4 → ?
Q: In the following acid-base reaction NaHSO4 + NazHPO4 Na2SO4 + NaH2PO4 The acid is and its conjugate…
A: ->Acid is species which can give H^+ whereas base which can accept H^+.
Q: Indicate each as either strong acid, strong base, weak acid, or weak base. a. KOH(aq) --> K+(aq) +…
A: For strong acid or base complete dissociation take place
Q: 1. Label the acid (A), base (B), conjugate acid (CA), and conjugate base (CB) in each of the…
A: Dear student , since you have posted multiple questions we are allowed to solve only first question…
Q: 12. For the reaction shown below, draw the conjugate acid and conjugate base in the appropriate…
A: Acid after loosing hydrogen ion it becomes conjugate base . Base after gaining hydrogen ion it…
Q: Draw the products formed when 2-propanol [(CH3)2CHOH], the main ingredient of rubbing alcohol, is…
A: Bronsted-Lowry acid-base theory: According to the Bronsted-Lowry acid-base theory, acid is a…
Q: Complete the reaction when ammonia is dissolved into water NH3 + H20 a. NH4*1 b. Он1 C. H30*1 d. H20…
A: Given, NH3 + H2O ------>
Q: 1. Consider the reaction below CH3COOH H20 H3O* ---> CH3COO CH3COOH [Choose is the conjugate acid is…
A: Acid It is a substance containing hydrogen and have the ability to donate it to another su8bstance.…
Q: In the following reaction, what compound is the conjugate base? HBr + NH3 →NH4+ + Br−
A: In this question, we have to find out the correct answer of given problem by the help of conjugate…
Q: A. A conjugate acid is the protonated form of a Bronsted base. B. A conjugate base is the ionized…
A:
Q: In the following reaction: NH, (aq) + H,0(aq) = NH,(aq) + H,0 (aq) Select one: O a. H,0 is a base…
A: A number of scientists put forward theories to explain the acidic and basic nature of the…
Q: Which pKb value corresponds to a conjugate acid-base pair with the strongest acid?
A: Difference between conjugate acid &base is one proton According to bronsted proton donar acid…
Q: TRUE OR FALSE Conjugate base of NH4+ is NH3
A: Conjugate base of a compound can be written by removing H+ from the compound.
Q: Which substance is the acid? Which substance is the base? Which substance is the conjugate acid?…
A:
Q: Draw the conjugate base of the following "acids": он OH
A: Conjugate base is defined as substance that forms when an acid loses a hydrogen ion.
Q: 1. Classify each reactant as an acid or base. CF + NH4* a. HCI + NH3 b. CH3COOH+ H2O CH3COO" + H;O*…
A:
Q: Al(OH)3 has an OH-1 in the formula so we don't use water in the reaction because it just cancels out…
A: Al(OH)3 has OH- in the formula, so it dissociates into Al3+ ions and OH- ions.
Q: In the given reaction, determine the correct conjugate acid-base pairs. HCIO4 + NaHCO3 = H2CO3 +…
A:
Q: In the following reaction, what compound is the base? HBr + NH3 →NH4+ + Br−
A:
Q: 8. How do I identify the conjugate acid for each of the following bases? Base Conjugate acid Base…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Label each reactant and product in this reaction as a Brønsted acid or base. HCN + NH, CN + NH,…
A:
Q: Solve this
A: Substances can be classified as acids and bases by different concepts. Accordind to Bronsted lowry…
Q: In the reaction below, identify the acid, base, conjugate acid and conjugate base : H2O + NH3 -->…
A: According to the bronsted- Lowry acid-base theory. A species is called acid if it can donate a…
Q: Of the following reactions, classify who is the acid and who is the base, as well as the bases and…
A: According to Bronsted Lowry theory a acid is a substance which can donate H+ and base is a…
Q: Which pair is not a conjugate acid–base pair?(a) (CH3)3 N; (CH3)3 NH+ (b) H2SO4; H2SO3 (c) HNO2;…
A: Acids and bases are electrolytic species that dissolve in water to dissociate into their constituent…
Q: What is the conjugate acid of attached each base?
A: According to Bronsted lowry concept of acid and bases: An acid is a substance that donated a proton…
Q: Base on the reaction what is the conjugate acid? HNO₃ + H₂O ⟶ H₃O⁺ + NO₃⁻
A: The given reactions is, HNO3(aq) + H2O(l )<------>H3O+(aq) + NO3-(aq)
Q: Draw the conjugate base of the following: Acids: Conjugate Base: Bases: Conjugate Acid: H;SO4 HSO4…
A: Conjugate base is formed when an acid loses a proton H+Acid conjugate…
Q: Which conjugate base is the most stable? O: OH NH2 (i) (ii) (ii) (iv)
A: There are two types of bases/acids. One is strong base/acid which is completely dissociable in into…
Q: What acid has the strongest conjugate base? CH3COOH C6H5OH H2SO4 H3PO4
A: The conjugate base is formed by the removal of H+ ions. The conjugate base of CH3COOH is CH3COO- The…
Q: 1- Select the one which is neither an acid nor base а) CHCOОН b) НCI c) KCI d) CH3OH
A: Acid is species which donates proton Base is species which accepts proton
Can you help me complete this acid base reaction? And label the acid,base,conjugate acid,and conjugate base.
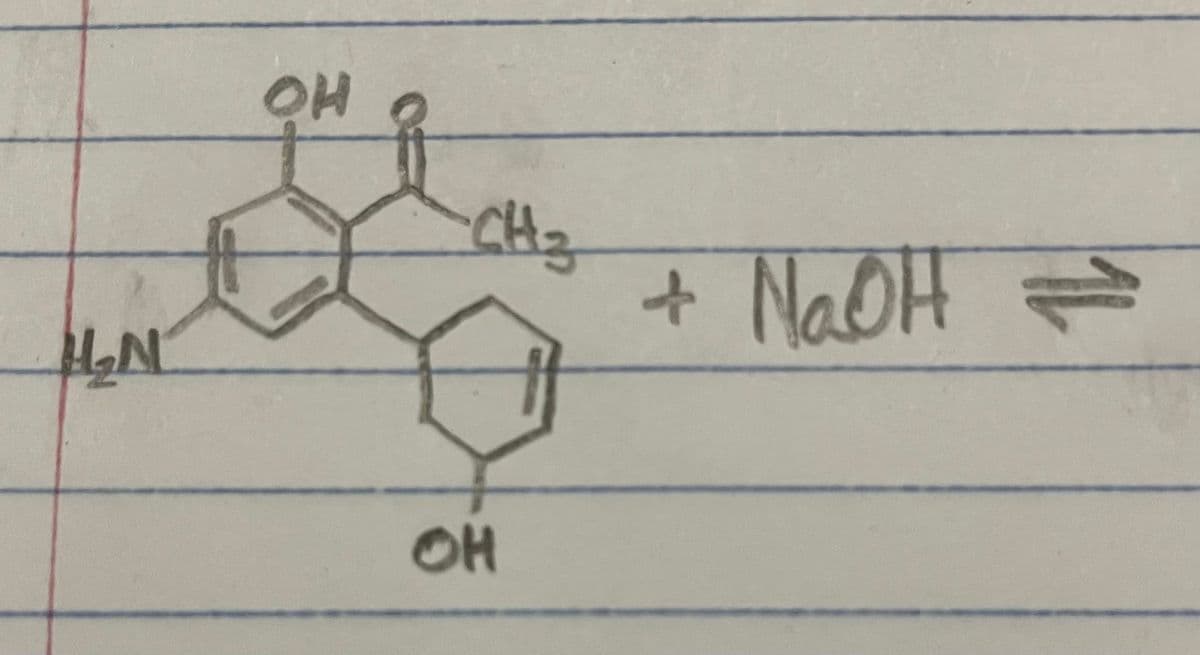
Step by step
Solved in 2 steps with 1 images


