NaOH (aq) + HCI (aq) - NaCl (aq) + H20 (1) Volume of 1.00 M HCI (mL) 45.0 Volume of 1.00 M NAOH (mL) 50.0 Initial Temperature ("C) 22.1 Final Temperature ('C) 31.6 (a) Calculate the moles of HCI used in the reaction. Moles of HCI = 0.0450 mol (b) Calculate the moles of NaOH used in the reaction. Moles NaOH= 0.0500 mol (c) Which reactant is the Limiting Reactant, HCI or NaOH? Limiting Reactant = HCI (d) Is this acid-base neutralization reaction exothermic or endothermic? exothermic NOTE: DO Include the correct number of significant figures or you will be marked wrong and DO NOT include units in your answer.
NaOH (aq) + HCI (aq) - NaCl (aq) + H20 (1) Volume of 1.00 M HCI (mL) 45.0 Volume of 1.00 M NAOH (mL) 50.0 Initial Temperature ("C) 22.1 Final Temperature ('C) 31.6 (a) Calculate the moles of HCI used in the reaction. Moles of HCI = 0.0450 mol (b) Calculate the moles of NaOH used in the reaction. Moles NaOH= 0.0500 mol (c) Which reactant is the Limiting Reactant, HCI or NaOH? Limiting Reactant = HCI (d) Is this acid-base neutralization reaction exothermic or endothermic? exothermic NOTE: DO Include the correct number of significant figures or you will be marked wrong and DO NOT include units in your answer.
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 3ALQ: ou place hot metal into a beaker of cold water. ol type='a'> Eventually what is true about the...
Related questions
Question
did i do this right?
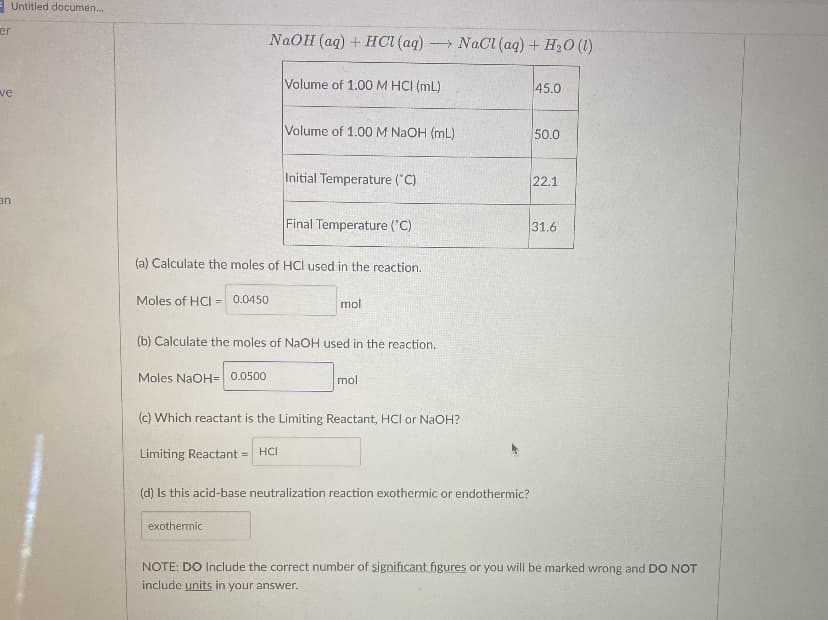
Transcribed Image Text:EUntitled documen.
er
NAOH (aq) + HCI (aq) NaCI (aq) + H20 (1)
Volume of 1.00 M HCI (mL)
45.0
ve
Volume of 1.00 M NaOH (mL)
50.0
Initial Temperature ("C)
22.1
an
Final Temperature ('C)
31.6
(a) Calculate the moles of HCI used in the reaction.
Moles of HCI =
0.0450
mol
(b) Calculate the moles of NaOH used in the reaction.
Moles NaOH= 0.0500
mol
(c) Which reactant is the Limiting Reactant, HCl or NaOH?
Limiting Reactant =
HCI
(d) Is this acid-base neutralization reaction exothermic or endothermic?
exothermic
NOTE: DO Include the correct number of significant figures or you will be marked wrong and DO NOT
include units in your answer.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning