Q: What volume of 0.420 mol/L sodium hydroxide is needed to neutralize 100 mL of a 6.50 mol/L hydrobrom...
A: A neutralization reaction can be described as a chemical reaction where an acid and base reacts with...
Q: Based on the solubility rules, which one of these compounds is insoluble in water? ...
A: Solubility rules are a set of rules which tells us the general trend of the solubility of some gener...
Q: What will be the average density for a sample of metal with a mass of 12.6 g and volume calculations...
A:
Q: 5. Propose a synthesis. CN
A:
Q: Identify the oxidation state of H in H2(g). Mg () 2HC(aq) → MgCl2(aq) • H2(8) O+1 O +2 O-1 0-2
A:
Q: 9C (s) + 12H2 (g) + 3/2 O2 (g) → 3C3H7 OH (1) The enthalpy change is -954 kJ and entropy increases. ...
A: Le Chatelier’s principle states that when a system at equilibrium is subjected to a change in concen...
Q: Do a retrosynthetic analysis on the following compound, ending withavailable starting material.
A: The retrosynthetic analysis of the target molecule (heptane-2,6-diol) gives to two moles of availabl...
Q: A gas sample has a volume of 0.185 X 10-6 m3 at 38 °C. What is its volume at 97 °C?
A: Given- Initial volume = 0.185 × 10-6 m3Initial Temperature = 38 °CFinal Temperature = 97 °C To find-...
Q: Hypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that p...
A: Given compound,
Q: Desricbe the Heat conductibility And Brittleness of metallic crystals, ionic crystals, molecular cry...
A: Metallic crystal-Metallic crystals are surrounded by a large no. of delocalized valence electrons . ...
Q: The table to the right gives the number of protons (p) and neutrons (n) for four isotopes. Which iso...
A: The potassium-39 has mass number 39. The isotope which on positron emission gives potassium-39 must ...
Q: The Ksp of CaF2 is 1.6 x 10 What is the concentration of F- in solution?
A: Ksp = 1.6 × 10-6
Q: In fertilizer, 15.0 g of urea (NH2 CONH2) is dissolved in 250 mL of water. A temperature change from...
A: Given that, Amount of urea, = 15.0 g Volume of water, m= 250 mL = 250 g * density of water at roo...
Q: please answer letter b. Please help. Tysm❤️B. Choose one type of solid (metallic crystals, ionic cry...
A: The solids are one of the three states of matter and the presence of constituent particles in close ...
Q: 4 ) When a 5.64 g sample of a non - electrolyte was dissolved in 40.0 g of water , the freezing poin...
A:
Q: Show that a phosphatidic acid is chiral, even though none of its fatty acids are chiral.Where is the...
A:
Q: A 0.3654 g portion of pure formic acid (CH2O2, FW=46.03 g/mol, Ka=1.77 x 10-4) is dissolved in50.00 ...
A: Given that, Mass of formic acid = 0.3654 g Molecular mass of formic acid = 46.03 g/mol Volume of wat...
Q: Show how you would accomplish the following synthetic transformations. Show all intermediates. 2,2@d...
A: The reaction and intermediates for the transformation of 2,2-dibromobutane into but-2-yne are:
Q: What are the principles of radiometric dating methods? detail your descriptions with figures
A: Radiometric dating, radioactive dating or radioisotope dating is a technique which is used to date m...
Q: St unit for the amount of substance is* kelvin mol Kg
A:
Q: I need help with ICE Chart. No matter what I do I don’t get it right, I will show what I have. This ...
A:
Q: Draw Lewis structures for three constitutional isomers for C4H6O2
A: Constitutional isomers are those compounds in which the molecular formula of the compound is same bu...
Q: Consider the compound trans-1,3-di-tert-butylcyclohexane. a. Draw the two chair conformers expected ...
A: Conformers: While any two arrangements of atoms in a molecule that differ by rotation about single b...
Q: Part A Identify each of the following changes of state as melting, freezing, sublimation, or condens...
A: Concept: Melting = It is a physical process in which solid converts into liquid. Freezing = It is a ...
Q: At constant volume, the heat of combustion of a particular compound is - 3800kJ / m * o * l . When 1...
A:
Q: Suppose that the acid HA (with dissociation constant Ka) is partitioned between aqueous phase 1 and ...
A: Distribution coefficient: It is defined as the ratio of the total concentration in phase 2 and the t...
Q: Smaller molecules have a smaller diffusion coefficient. O a. True O b. False
A: False. Diffusion is defined as the net movement of atoms or molecules that are flows from the high c...
Q: 3. Find the density of air at 24°C and 2.026 x 10^5 Pa. One mole of air is assumed to be 79% nitroge...
A: PV = nRT 1Pa = 9.869*10-6 atm P = 2.026 * 105Pa = 2.026*105*9.869*10-6 = 1.999atm n = 1 mole T =...
Q: How many mol of hydrogen are needed to double the volume occupied by 0.32 mol of hydrogen, assuming ...
A: 0.32 moles Given that, at constant temperature T and pressure P ; Moles of Hydrogen gas, n = 0.32 mo...
Q: The volume of a gas is increased from 150.0 mL to 350.0 mL by heating it. If the original temperatu...
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Draw a Lewis structure for the ozone molecule, O3. (The order of atom attachment is O-O-O, and they ...
A: A Lewis Structure is a simplified representation of the valence shell electrons in a molecule. It is...
Q: If it takes 9.47 minutes to fill a 60.0 gal gas tank, calculate the gasoline flow rate in mL/s.
A:
Q: In electro-thermal vaporization technique: Which of the following is incorrect? this technique uses ...
A: Given : Electrothermal vaporization technique. To find: Incorrect statement from the following optio...
Q: How many grams of carbon tetrachlorride are in 1.01 x 1024 molecules?
A: Given: Number of molecules= 1.01×1024 To find: Grams of carbon tetrachloride Formula: Number of mol...
Q: Br CH3 CH,ONa H3C CH2CH3 H3CH2C -write the Newman projection for the sawhorse structure given - Writ...
A: A Newman projection, useful in alkane stereochemistry, visualizes the conformation of a chemical bon...
Q: I just need the product of this reaction.
A: Cyclopentene is a hydrocarbon and combustion of cyclopentene takes place in presence of O2 .
Q: 1- Is it possible to obtain soap from main component of lavender essential oil shown in below? Expla...
A: Soap is the sodium salt o various naturally occurring fatty acids. Soap is produced by the process ...
Q: Is 50% by mass of methanol dissolved in ethanol different from 50% by mass of ethanol dissolved in m...
A: Given, Solution 1: Mass % of methanol = 50% Solution 2: Mass % of ethanol = 50% Mass % means mass of...
Q: José performs an experiment to determine how much sugar can be dissolved in 250 milliliters (ml) of ...
A: Solubility of a compound is the fraction of its concentration that dissolved in a solvent. Solubilit...
Q: 9. A technician is preparing a 40% solution. What volume of stock solution should be use to prepare ...
A:
Q: What is the mass of Lithium Nitrate (30% mass/mass) present in a 50g sample of soil? Conditions when...
A:
Q: What is chemistry
A: Chemistry is a branch of science.
Q: Part A The compound MgS0, Epsom salt, is used to soothe sore feet and muscles. How many grams will y...
A: Solution to Part A: Molar mass of Epsom salt (MgSO4) = 120.366 g / mol Therefore, mass of Epsom salt...
Q: Calculate the solubility of PbI2 obtained by mixing 200 mL of 0.0100 M Pb(NO3)2 and 100 mL of 0.100 ...
A: Given data,Molarity of Pb(NO3)2=0.010MVolume of Pb(NO3)2=200mL=0.200LMolarity of NaI=0.100MVolume of...
Q: Propose a mechanism for the bromination of ethoxybenzene to give o- and p-bromoethoxybenzene.
A: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. ...
Q: Show all the work and setps. Perform the following calculations using your calculator and then expr...
A:
Q: What is the dipole–dipole force? How can you predict the presence of dipole–dipole forces in a compo...
A: There are different types of intermolecular forces which defines , most of the physical and chemical...
Q: Hygrogen cyanide gas (HCN), a power respiratotoxic, inhibitor, is highly toxic. It is a very weak...
A: HCN reacts with NaOH as follows: HCN + NaOH → NaCN + H2O 1 mole of HCN reacts with 1 mole of NaOH...
Q: Propose a chain-reaction mechanism for the decomposition of dimethyl ether to form methane and forma...
A: Given:
Q: Write equations for the formation of (a) MgCO3(s) and (b) C6H12O6(s) from their respective elements ...
A: In the computation of heat of reactions it is a conversion to assume that the heat of formation of e...
Show the product of treating the following g-lactam with reagent
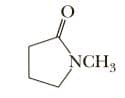
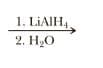
The reagent, Lithium aluminum hydride is a reducing agent. It reduces amide functional group to amine functional group. The product amine depends on the type of substituent already present.
The substrate is a -lactam, it is a cyclic amide. The prefix '' refers to a 5- membered ring.
Step by step
Solved in 2 steps with 1 images


