Q: O Resources Hint Identify the products formed in this Brønsted-Lowry reaction. HSO, + F acid + base ...
A: A bronsted lowry base is a substance which accept a proton are H^+ ions from the other compound and ...
Q: fill in problem as requested
A:
Q: What are the errors in the following answer?
A: A question based on chain extension of epoxide, which is to be accomplished.
Q: refer to image, please explain in detail, showing all steps. will rate!
A: In this question, we will Identify which Compound ( A or B ) give carbocation intermediate ( C ) wit...
Q: what is the balanced chemical reaction for the galvanic cell that contains the two half reactions. ...
A:
Q: A fellow lab student is attempting to identify his unknown organic compound by using the Tollens' te...
A: For tollens' positive test aldehyde must have alpha hydrogen.
Q: In one process used for electroplating silver, a current of 10.23 A was passed through an electrolyt...
A: Dear student, this question is related to electroplating process.
Q: What is the conjugate acid of HPO4 2-?
A: Given :- base = H2PO42- To identify :- conjugate acid of the respective base
Q: A 1.00ml aliquot of fish odl was andyrd for N using the Kieldahl methad: After chqashim, the dishill...
A: Mili equvalent of acid=100ml×0.0503 M =5.03mili equvalent Miliequvalent of base=28.3ml×0.124M =3.50...
Q: An important step in the production of sulfuric acid is the oxidation of SO2 to SO3. SO2 (g) + 1/2 O...
A:
Q: If Kc = 0.0084 for the reaction below at 298.0 K, then what is the value of Kp? (R = 0.0821 L・atm/mo...
A: We have Kc = 0.0084 , temperature as 298 K, We have to calculate the value of Kp.
Q: 4
A: In naming a compound we select the longest carbon chain to name the base alkyl group. Then we look f...
Q: Chemistry Question
A: Dear student this question is related to organic chemistry.
Q: Need help
A: This question is related to pH.
Q: Which of the following reactions occurs more rapidly?
A: Applying concept of epoxide formation.
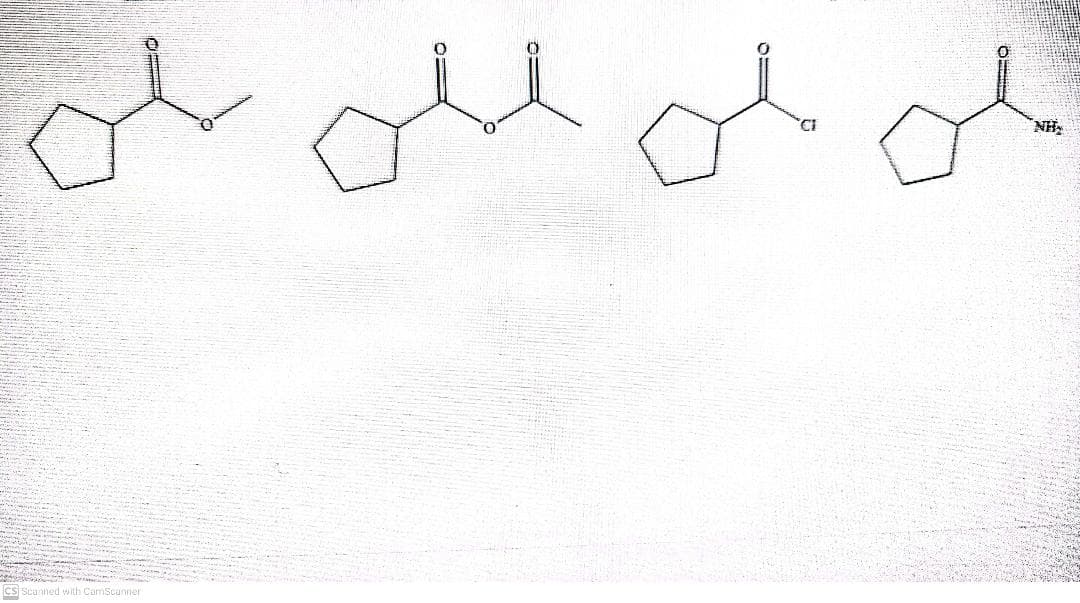
Q: Show the hydrolysis in basic conditions with the following carboxylic acid derivatives.Identify the ...
A: Carboxylic acid functional group is RCOOH R-CONH2 is amide , R-COOR is called as ester , RCO3R i...
Q: A sample of argon has a volume of 1.20 L at STP. If the temperature is increased to 22.0°C and the p...
A: The ideal gas equation is PV=nRT , by using ideal gas equation and modifying it according to given c...
Q: Given ΔGorxn = -1,114.8 kJ/mol for the following reaction and the listed ΔGof values below each subs...
A: Given ΔG0rxn = -1,114.8 kJ/mol T = 25 oC. 2 A (ℓ) + 3 B (g) →→ 2 C (g) + 4 D (ℓ) ΔGof (kJ/m...
Q: d If nickel(II) formate is heated to 300 °C in the absence of air for 30 minutes, the salt decompose...
A: Nickel formate decomposed to form nickel powder. Each mole nickel formate produces 1 mol of nickel. ...
Q: Write the equilibrium constant expression, K
A: the state at which rate of forword reaction equal to rate of backword reaction is called " equlib...
Q: Methanol 1CH3OH2 can be made by the controlled oxidation of methane: CH41g2 + 1 2 O21g2 ¡ CH3OH1g2 C...
A: 1) For CH4 - ∆G = -50.72 KJ/mol 2) For O2 - ∆G = 0 3) For CH3OH - ∆G = -166.27 KJ/mol
Q: Show how you might use the Williamson ether synthesis to prepare ether
A: Applying concept of Williamson ether synthesis.
Q: A certain shade of blue has a frequency of 7.28×1014 Hz.7.28×1014 Hz. What is the energy of exactly ...
A: For a wave with frequency (v) has energy (E) can be mathematically expressed using Planck hypothesis...
Q: 10
A: ]Given reactionN2(g) + O2(g) + Br2 (g) ⇔ 2 NOBr (g)Equilibrium constant of this reaction is give...
Q: The followings methods are useful to induce crystallization EXCEPT adding a small crystal of the des...
A: Crystallization is defined as a process where solid crystal forms from the solution. There are vario...
Q: Describe one example in real life of a factor affecting the rate of a specific reaction.
A: There are many examples in real life of a factor affecting the rate of a specific reaction.
Q: fill in problem as requested!
A:
Q: d If you synthesize 3.0 micromoles of NaTcO4, what mass of compound do you have? If the compound is ...
A: We will convert moles to grams. Then calculate molarity of solution.
Q: What amount of heat, in kJ, is required to vaporize 121.45 g of ethanol (C₂H₅OH)? (∆Hvap = 43.3 kJ/m...
A: Answer:- This question is answered by using the simple concept of stoichiometry for the thermochemic...
Q: The equilibrium constant, K, for the following reaction is 4.89 at 397 K. 2CH,Cl2(g)CH,(g) + CCl4(g)...
A: We have Kc for given reaction as 4.89. We have to calculate the Kc for the new given reaction.
Q: Match the substance to its classification in aqueous solution. NH3 NH4Br 1. Weak acid HC,H5 2. Stron...
A: Different type of compounds have different nature on dissolving them in water. Some of the Substance...
Q: You are asked to determine the amounts of calcium ion in a 200 mL water sample using EDTA to titrate...
A: The equation representing titration of Ca2+ ion with EDTA: Ca2+ (aq) + EDTA4- (aq) → Ca(EDTA)2- (aq)
Q: Charles' Law explores the effects of temperature on the volume of an ideal gas. Charles’ Law is desc...
A:
Q: Hello! how can i derive this formula if I am to find the mL of silver nitrate? Thank you!
A: A numerical problem based on normality, which is to be accomplished.
Q: I do not understand how to work out this problem.
A:
Q: Magnesium carbonate decomposes into magnesium oxide and carbon dioxide. What is the sum of the coeff...
A: Interpretation - To tell about the sum of the coefficients for the reactants and the products when M...
Q: Iaterial: Block Mass: Сорper 5.0 g 10.0 g Heating Duration: D 2s T initial T final 71.9 °C Heat Rese...
A: Specific heat capacity is the amount of heat required to raise 1°C temperature of 1 g of a substance...
Q: 2
A: Following transition is not allowed in vibrational it spectra .
Q: At 25.0° C, a 10.00 L vessel is filled with 7.00 atm of Gas A and 5.89 atm of Gas B. What is the mol...
A: In this question, we want to determine the mole fraction of gas B. You can see details Solution bel...
Q: The reaction below shows the Haber process used in the commercial production of ammonia from nitroge...
A:
Q: A sample of indium chloride weighing 0.5000 g is found to contain 0.2404 g of chlorine. What is the ...
A: Solving only first question in accordance with guidelines. We will calculate weight of indium and ch...
Q: At the same temperature, which of the following gases will effuse the slowest? A) Hydrogen B) Helium...
A: We need to determine the gas which effuse slowest among the options.
Q: 1. How many ATOMS of nitrogen are present in 6.98 grams of nitrogen monoxide, NO? of nitrogen atoms
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Identify the conjugate acid for each base. conjugate acid of HS−: conjugate acid of SO2−...
A: According to Bronsted-Lowry concept Acid is a species which can donate a proton to another species a...
Q: 10
A: We have to named the following given compounds 23-30.
Q: Provide answer to the attached
A: Applying concept of SN2 mechanism.
Q: Is it possible for a single molecule to test true positive in all the qualitative assays? Why or why...
A: A question based on qualitative analysis, which is to be accomplished.
Q: Calculate the wavelength of the green line in the visible spectrum of excited H atoms. The green lin...
A: The question is based on the concept of atomic structure. we have to calculate wavelength for the gi...
Q: Chemistry Question
A: Dear student this question is related concept of enzyme models.
Q: What is the product of a Grignard reaction? O A 2° or 3° amine with the nitrogen bound to the R grou...
A: ->farmaldehyde react with grignard reagent to give primary alcohol. ->aldehyde or ketone react...
Draw the step-by-step mechanism using curved arrows of the following reaction.
Step by step
Solved in 4 steps with 4 images

- 1. Define sparging. What is the significance of this step in an HPLC analysis? 2. What are the two types of pre-column? What is the purpose of each type? 3. What are the two types of packing used in HPLC column? Describe each type. Please answer numbers 1-3. Need asapDefine the following terms used in HPLC: (d) sampling loops (e) bonded-phase packing (f) gel filtrationWhat are the retention times and areas of all the attached peaks?

