NH, SH NH, он HN C. H,N HN PEPTIDE RNGCSN NH, PEPTIDE AHIKP
Essentials Health Info Management Principles/Practices
4th Edition
ISBN:9780357191651
Author:Bowie
Publisher:Bowie
Chapter10: Introduction To Coding And Reimbursement
Section10.1: Clinical Classification Systems
Problem 5E
Related questions
Question
Use an arrow to draw the site for cleavage for the following peptides reacted by trypsin. Explain comprehensively.
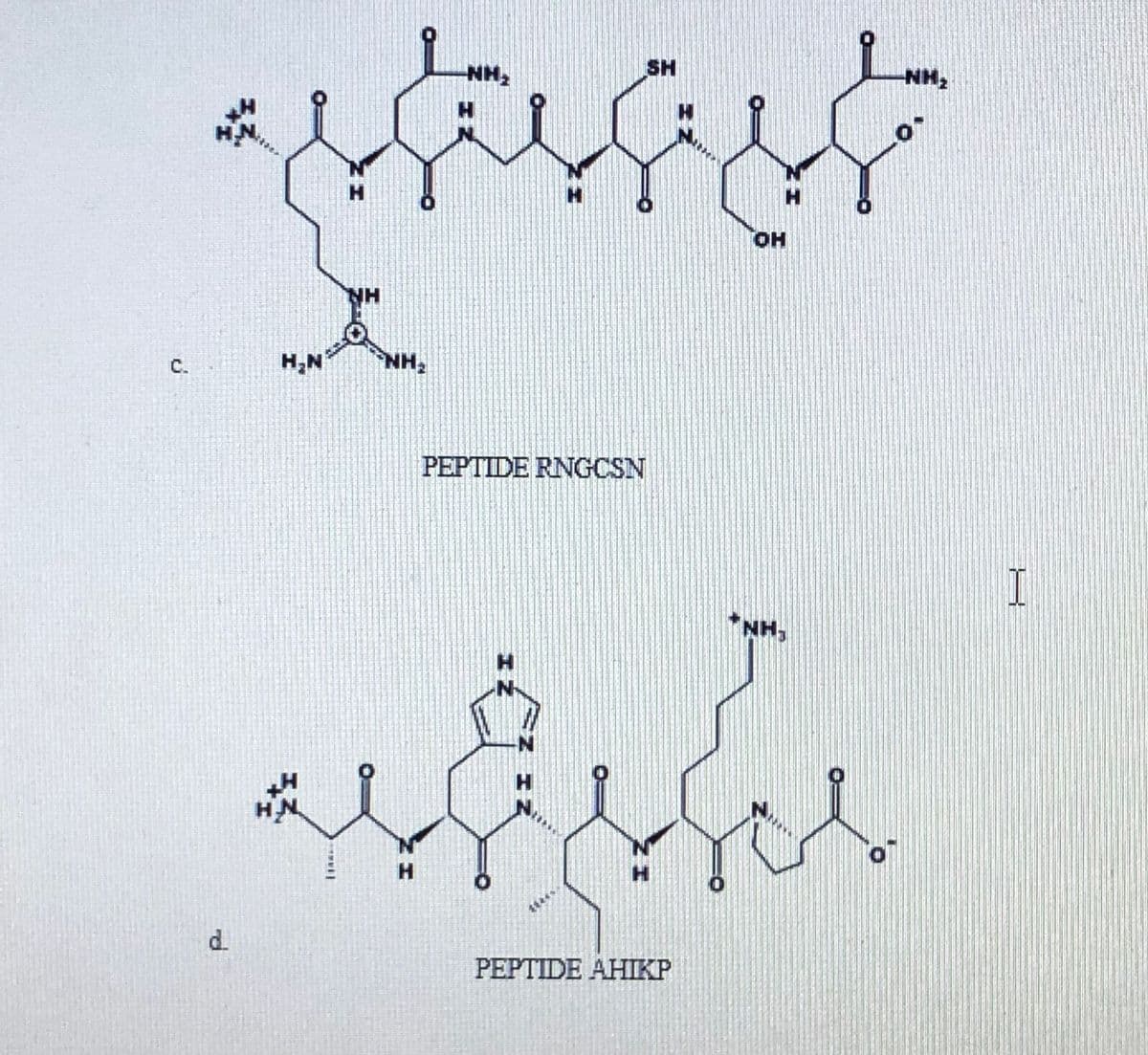
Transcribed Image Text:NH,
SH
NH,
HN
H.
H.
HO.
NH
C.
H,N
NH,
PEPTIDE RNGCSN
I
NH,
HN
d.
PEPTIDE AHIKP
エ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Essentials Health Info Management Principles/Prac…
Health & Nutrition
ISBN:
9780357191651
Author:
Bowie
Publisher:
Cengage



Essentials Health Info Management Principles/Prac…
Health & Nutrition
ISBN:
9780357191651
Author:
Bowie
Publisher:
Cengage




Comprehensive Medical Assisting: Administrative a…
Nursing
ISBN:
9781305964792
Author:
Wilburta Q. Lindh, Carol D. Tamparo, Barbara M. Dahl, Julie Morris, Cindy Correa
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax