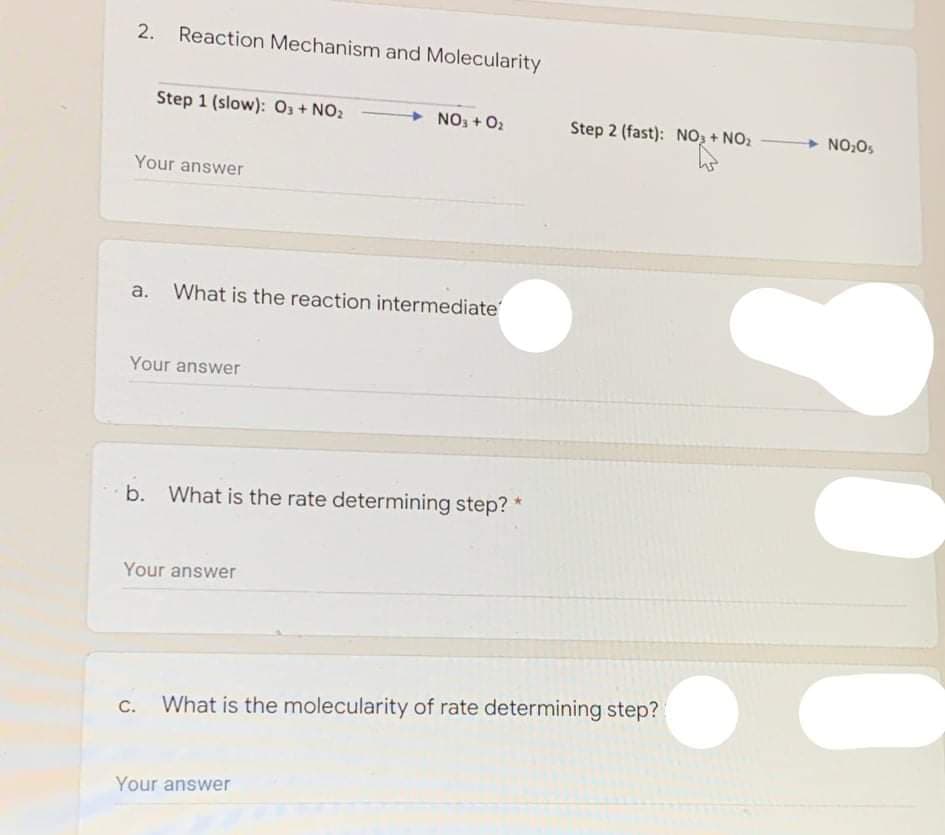
2. Reaction Mechanism and Molecularity Step 1 (slow): O3+ NO2 NO3 + O2 Step 2 (fast): NO, + NO: NO Your answer
Q: D-Galactose.
A: ''Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 38. "The sterol is a precursor to all compounds except" A.ergosterol B.testosterone C.bile salts…
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: What does the last number in the numeric designation of enzymes refer to?
A: Each enzyme is allocated a four digit EC number,an enzyme Commission number does not specify enzymes…
Q: What factors can prolong the coagulation time? What is the reason for the use of siliconized glass…
A: The process by which liquid blood turns into a gel-like substance (blood clot) is referred to as…
Q: Which of the following statements about protein digestion are true? Check all that apply. ► View…
A: Proteins are macronutrients, which act as the building blocks of the body. Proteins can also act as…
Q: True or False: 1. Electron-deficient chlorophyll molecules of PS II receives electron from PS I.…
A: Introduction: The light-absorbing pigment of thylakoids is arranged in functional arrays called…
Q: Given a mixture of three proteins, A, B and C, with isoelectric points pIA = 4.2, pIB = 7.0 and pIC…
A: The isoelectric point (pI) of a protein is defined as the pH at which the net charge of a protein…
Q: How do ADP and ATP regulate the glycolysis pathway?
A: Glycolysis is an important metabolic pathway which participates in the carbohydrate metabolism. This…
Q: interphase FISH is used on fixed material from a solid tumour, rather than metaphase FISH
A: Interphase FISH is a new technique introduced into the molecular cytogenetics. it is used to study…
Q: Predict the relative intensity and wavelength maxima for the given molecules and stated conditions.
A: Intrinsic fluorescence is observed due to three aromatic amino acid residues - phenylalanine,…
Q: Calculate the energy produced (in ATP molecules) achieved by complete oxidation of the hydrolysis…
A: Carbohydrates are the primary energy source for most organisms. Carbohydrates are digested to…
Q: Neutrophils exhibit a respiratory burst of free radicals and strong oxidizers during acute…
A: Free radicals are highly energetic chemical species that go onto react with almost anything.…
Q: Which of the following was added RNA extraction? Buffer RPE Wash buffer guanidine-isothiocyanate…
A: DNA molecule is a sritical methd. It helos in serine, nRNa Nither momt tertibre
Q: Which of the following pairs of amino acids can have intermolecular hydrogen bonding between the…
A: The secondary structure of proteins is stabilized by hydrogen bonding. mainly it…
Q: An inhibitor "I" is added to the enzymatic reaction at a concentration of 1.0 g/L. The data obtained…
A: The reciprocal kinetic data is as follows: 1/[S] L/g 1/V (min-L/g) E = 0.015 g/L (Without…
Q: Propose a reasonable biosynthesis for compound 34 starting from acetyl CoA, alanine CoA, malonyl CoA…
A: Acetyl Co A is involved in my biochemical reactions (carbohydrate and lipid metabolism). It is a…
Q: malate dehydrogenase is interconverted into isozymic forms by oxidizing and reducing agents
A: Malate dehydrogenase is found in a variety of organs in vertebrates. It has been discovered that…
Q: 2. Use your knowledge of amino acids (and the R groups) and tertiary structures of proteins to…
A: Amino acids are monomers of protein they are linked with each other by forming…
Q: How does salt help in the DNA extraction process?
A: DNA is extracted to compare the DNAs from different sources and to study the sequence differences.…
Q: Frank weighs 112 kg and John weighs 56 kg. Both are exposed to 5 mGy of gamma radiation. Do they…
A: The dosage of radiation absorbed per kilogramme. The level of radiation does not mean anything in…
Q: Describe in detail the four levels of protein structure
A: Folding of protein is necessary for its biological activity. Protein folding occurs spontaneously,…
Q: How does the DNA hold information?
A: Deoxyribonucleic acid (DNA) is a polymer made up of two polynucleotide chains that coil around each…
Q: Draw the complementary DNA strand for the given: 5'-A-T-C-C-G-A-A-T-T-G-3' Draw the complementary…
A: Let us first understand the one letter codes for the given nucleotides: A is Adenine T is Thymine…
Q: Long explanations are not needed. True or False a. One NADH is produced for every pyruvate…
A: The pentose phosphate pathway is an alternative pathway for glucose oxidation. It provides NADPH for…
Q: What is the correct sequence of steps in the light reaction? ADP + Pi --> ATP oxidation of water…
A: In plants, photosynthesis occurs in two stages one is light-dependent and the other is…
Q: 16. Within the chloroplast, grana are ____. Select one: A. stacks of enfolded thylakoid membrane…
A: A chloroplast is a plastid, which is a source of plant cell organelle. Plastids aid in the storage…
Q: Which of the following steps does NOT require ATP? O A. Conversion of glucose to glucose 6-phosphate…
A: Metabolic pathways are a series of process which includes chemical reactions occurring in a cell.…
Q: During exercise when the body lacks an adequate supply of oxygen to support energy production, the…
A: When a person does workout, then the body has a tendency to lack a very high amount of oxygen…
Q: Interpret the two chromatograms below. Which of the two chromatograms will provide good sequencing…
A: In Automated DNA sequencing, using a laser the different fluorescent labelled nucleotides at the end…
Q: Failure of anti-oxidant function results in the hydroxylation of an aromatic acid of Enzyme Z and…
A: It isn't mentioned in the question on which strand of the c-DNA was taken for Sanger sequencing. So…
Q: Which post-transcriptional modification is NOT shared by all eukaryotic RNAs? a. Modifying bases…
A: Posttranscriptional modification is the process by which the RNA primary transcript is chemically…
Q: What is the maximum number of possible intra-peptide disulfide bonds for any given peptide form?
A: The side chains of Cysteine residues in proteins undergo oxidation to form disulfide linkages.
Q: Chemistry A poison has been found to severely compromise the energy production in its victims.…
A: Energy is produced in the mitochondrial matrix through the process of oxidative phosphorylation.…
Q: During plasmid extraction, after the addition of acetate-conta in ing neutralization buffer the…
A: Plasmids are extrachromosomal DNA in the bacterial cells. They are double stranded circular DNA,…
Q: Given the following pKa data for the individual amino acids, estimate the pI for the tripeptide…
A: In the given data pKa of amino acids Thr-Asp-Arg is: Thr-? Asp-3.86 Arg-12.48 N-terminal group-…
Q: Use the data below to determine the maximum velocity [in mM/s] of a certain enzyme-catalyzed…
A: We must know the Michaelis Menten equation: V=Vmax [S]Km+ [S]
Q: The lipase substrate emulsion contains 0.500 mg of olive oil per 3 mL Also, the molar mass of the…
A: The number of moles of a substance is calculated by using the equation, n=mM, where, "n" is the…
Q: Complete the table
A: Polysaccharides is one of the important component in living organisms, which is found many diverse…
Q: 14. In an a-helix, a hydrogen bond is formed between the carbonyl oxygen of the residue and the…
A: The alpha helix is a type of secondary structure of the protein. They are stabilized by hydrogen…
Q: depletion of NAD+
A: Nicotinamide adenine dinucleotide ( NAD) is a coenzyme central to metabolism. It is found in all…
Q: Which of the following describes a Z-DNA helix? a. It is inhibited by methylation of bases b. It is…
A: DNA is a polymer made up of two polynucleotide chains that coil around each other to form a double…
Q: Which critical product/s of the light reaction are required for the Calvin cycle? О ATP O NADH O O O…
A: Photosynthesis is divided into light and dark reactions. In photosynthesis carbon dioxide is used to…
Q: Polyunsaturated fatty acids a). are solid at room temperature b). have carbon-carbon double bonds…
A: Fatty acids are carboxylic acids with long hydrocarbon chain. There are two types of fatty acids -…
Q: Given the knowledge of biochemistry, make a sample outline of a new 24-hour healthy meal. Provide…
A: The human body can be viewed as an engine that releases the energy contained in the foods it…
Q: lease help me answer questions 71 & 77 71. Which of the following are examples of kinetic energy? A.…
A: Introduction: Kinetic energy: The term energy is the ability to do work or cause change. There are…
Q: What are the products of the Krebs Cycle?
A: Krebs cycle is one of the most important metabolic pathway. It is also known as citric acid cycle or…
Q: Give the functions of both water-soluble and fat-soluble vitamins
A: The B vitamins (folate, thiamine, riboflavin, niacin, pantothenic acid, biotin, vitamin B6, and…
Q: Aldolase enzymes have the following rate constants. k₁ = 10³M ¹s¹, k-₁ = 4.4x10 s ¹, k₂ = 10's-¹ -1…
A: Consider the following single substrate (S) enzyme (E) catalyzed reaction mechanism given below E +…
Q: How many enyzymatic reactions are there in glycolysis pathway?
A: There are 10 enzymatic reactions in glycolysis pathway.
Q: pls help ASAP, thanks!
A: In the anaplerotic reaction the oxaloacetate metabolite is converted to PEP by the PEP carboxykinase…
Step by step
Solved in 3 steps

- Which of the following is the best way to judge the relative activation energies between two given chemical reactions? Compare the ?G values between the two reactions Compare their reaction rates Compare their ideal environmental conditions Compare the spontaneity between the two reactions.Which of the following methods is not used by enzymes to increase the rate of reactions? a. covalent bonding with the substrate at their active site b. bringing reacting molecules into close prosimity c. orienting reactants into positions to favor transition states d. changing charges on reactants to hasten their reactivity e. increasing fit of enzyme and substrate that reduces the energy of activationIn the following graph: A represents the product. B represents the energy of activation when enzymes are present. C is the free energy difference between A and D. C is the energy of activation without enzymes. E is the difference in free energy between the reactant and the products.
- 1) what is the equilibrium constant for the reaction A + CT AT+ C? Show calculations. 2) the imageWhen two glucose molecules react to form maltose: a. the reaction represents a negative G. b. free energy had to be available to allow the reaction to proceed. c. the reaction is exothermic. d. it supports the second law of thermodynamics, which states there is tendency of the universe toward disorder. e. the resulting product has less potential energy than the reactants.Which of the following comparisons or contrasts between endergonic and exergonic reactions is false? Endergonic reactions have a positive ?G and exergonic reactions have a negative ?G Endergonic reactions consume energy and exergonic reactions release energy Both endergonic and exergonic reactions require a small amount of energy to overcome an activation barrier Endergonic reactions take place slowly and exergonic reactions take place quickly.
- Which of the following statements about the allosteric site is true? a. The allosteric site is a second active site on a substrate in a metabolic pathway. b. The allosteric site on an enzyme can allow the product of a metabolic pathway to inhibit that enzyme and stop the pathway. c. When the allosteric site of an enzyme is occupied, the reaction is irreversible and the enzyme cannot react again. d. An allosteric activator prevents binding at the active site. e. An enzyme that possesses allosteric sites does not possess an active site.In an enzymatic reaction: a. the enzyme leaves the reaction chemically unchanged. b. if the enzyme molecules approach maximal rate, and the substrate is continually increased, the rate of the reaction does not reach saturation. c. in the stomach, enzymes would have an optimal activity at a neutral pH. d. increasing temperature above the optimal value slows the reaction rate. e. the least important level of organization for an enzyme is its tertiary structure.1. Consider the following parameters related to an enzyme that follows Michaelis-Menten kinetics for the reaction: k(1) k(2) S ----> ES ----> P k(-1)
- 1. a) Fill in the blanks: a ____________ reaction is one that requires energy input, whereas a spontaneous reaction is one that _____________ energy. A spontaneous reaction is also called an ___________ reaction, whereas an endergonic reaction requires energy input. b) Briefly, why do living cells and organisms require an input of energy? c) How do animals like humans get their energy if they cannot obtain it directly from the sun like photosynthetic organisms? How do bacteria obtain energy? d) Chemical energy is the form of energy we will discuss. Where is the chemical energy stored in a molecule of glucose?1a-Which of the following is/are true for all exergonic reactions? Select all that apply Select one or more: a. The products have more total free energy than the reactants. b. The reaction proceeds with a net release of free energy c. The reaction requires energy from a coupled endergonic reaction d. A net input of energy from the surroundings is required for the reactions to proceed 1b-Which of the following can change the activity of a single enzyme? Select all that apply Select one or more: a. An inhibiting molecule noncovalently binding to the active site. b. A regulatory molecule noncovalently binding to a location other then the active site (an allosteric site). c. Another enzyme phosphorylating the enzyme of interest. d-Removal of a small section of the enzyme.1. Which of the following terms best describes the progress of the reaction with respect to free energy change?



