Q: Which radioactive decay is most probable?
A: There are different type of decays in nuclear Chemistry to give different type of changes in nuclear...
Q: Which atom in the Br-F molecule has a partial negative charge (8¯)?
A: In a polar molecule the more electronegative atom attracts the shared pare of electrons and partial ...
Q: 6, In the proposed two-step mechanism of decomposition of ozone the oxygen is: step 1: O3=>O2 + O...
A: Since you have asked multiple question, we will solve the first question for you. If youwant any spe...
Q: At 460 K, ∆G = -15.00 kJ/mol for the reaction 2 A (g) → B (g). If the partial pressures of A and B a...
A: Answer: ∆G° for this reaction is 3.42 kJ/mol.
Q: 100 mL of 0.5 M H2SO4 solution is needed. The stock solution is 6 M. How many milliliters of stock...
A: According to law of equivalency V1 × S1 = V2 × S2 Where V1 and V2 are initial and final volum...
Q: The men's world record for swimming 1500.0 m in a long course pool (as of 2007) is 14 min 34.56 s. A...
A: Length of swiming pool = 1500.0 m1 mi = 1609 mLength of swiming pool = 1500.0 m1609 m* 1 mi = 0.9322...
Q: Using bond energies determine the heat generated when 32 grams of methane, CH4, is burned. (in kJ) ...
A:
Q: Balance then answer questions. __ ClO2(g) + __ H2(g) ® __ HCl(g) + __ H2O(g) Calculate the...
A: Given reaction; 2ClO2(g) + 5H2(g) -----> 2HCl(g) + 4H2O(g) Question; To make 0.92 mol o...
Q: Show the reaction mechanisms of the two reactions for each step. Refer to the pictures below.
A:
Q: Question 2 What is the coefficient in front of H2SO, when the following cuation is balanced with the...
A:
Q: what is the full mechanism for hydroboration oxidation of hex-1-yne. state all of the reagents and d...
A:
Q: The equalibrium constant for the following equation HC2H3O2(aq) + H2O(l) ⇌ ...
A: The reaction taking place is given as, => HC2H3O2 (aq) + H2O ⇌ H3O+ (aq) + C2H3O2- (aq)
Q: Identify the final product of this reaction (note: D is deuterium): H2SO4, H2O -D HgSO4 Select one: ...
A:
Q: Ethylenediamine tetraacetate or EDTA is a very common reagent in Analytical Chemistry. As what wasme...
A: Two questions based on EDTA, which are to be accomplished.
Q: Can you give me a step-by-step solution of this problem? I can't quite understand what it means.
A:
Q: Nitrogen and oxygen react to form nitrogen monoxide, like this: N2(9)+O2(g) → 2 NO(9) The reaction i...
A:
Q: what are the products of these reactions, be mindful of which was the arrow is pointing include the...
A: As per the rule, only three questions can be answered.
Q: Can you give me a step by step solution of this problem?
A: Given the volume of distilled water = 50.00 mL Volume of 0.10 M HCl added = 4.00 mL * (1L/1000 mL) ...
Q: 5. Identify the major product A and write a detailed mechanism to explain its formation. What is the...
A: ->In aldol reaction for major product we see electrophilicity of carbonyl center enol form from t...
Q: (a) Write chemical equations to illustrate the following name bearing reactions :(i) Cannizzaro’s re...
A: Given reactions are : (i). Cannizzaro’s reaction (ii). Hell-Volhard-Zelinsky reaction Reactions = ?...
Q: The limiting reactant determines what the actual yield is.
A: About Limiting reactant : When two Reactants reacted with each other but after the Reaction one reac...
Q: In a recent year, 2.84 billion kilograms of polystyrene was produced in the United States. Polystyre...
A: Given: Mass of polystyrene produced = 2.84 billion Kg = 2.84 X 109 Kg = 2.84 X 1012 g ...
Q: Explain Hell-Volhard-Zelinsky reaction ?
A: Given: Hell-Volhard-Zelinsky reaction.
Q: Below is the chemical equation for the combustion of methanol (CH4O). How many grams of methanol ar...
A:
Q: At the same temperature, which of the following gases will have the fastest rate of effusion? A) CO ...
A:
Q: 4
A: Given volume of solution = 4.8 L density of solution = 1.01 g/ml
Q: What is the m/v% of 2000mL of 6M HCl solution?
A: m/v% = (mass of solute/ volume of solution) × 100
Q: A. This is the equilibrium constant for the reactionin which a solid salt dissolves to give its cons...
A:
Q: Based on the reaction below and your answer to Question 3 (-191.2 kJ) from The Additivity of Heats (...
A: We have given that SiO2(s) + 4HF(g) → SiF4(g) + 2H2O(l) Determine the amount of heat (kJ) that c...
Q: What is a limiting reagent and theoretical yield in grams of NO2 when 10.0 g NH3 react with 10.0 g o...
A: Given , mass of NH3 = 10.0g. Mass of O2 = 10.0 g. The balance reaction is 4...
Q: 52) What is (are) the elimination product(s) of the following reaction? Br II A) Only I B) Only II C...
A: Given reaction t-butoxide base with alkyl bromide gives alkene via elimination reaction
Q: solve by filling in blank spots as it asks please
A: In this question, we want to determine the Molecules of C6H8O6 How we can determine, you can see de...
Q: 2. Give IUPAC names to the following compounds. A. CO2H iHH NH2 В. CN ČO,H С. OH
A: Rules for nomenclature: 1) select longest carbon chain as parent chain / base. 2) check the function...
Q: Calculate the standard enthalpy change for the reaction where the heats of formation are given in th...
A:
Q: 1,The increase in the rate of the reaction with the increase in the concentrations of the reactants ...
A: The rate of reaction represent the disappearance of reactant molecule while appearance of product mo...
Q: Chemistry Question
A: Given An artificial sweetener = Aspartame Aspartame is = Methyl ester of a dipeptide Amino acid in...
Q: What is the effect of adding C2H5OH to a container in which the following reaction has reached equil...
A: The equilibrium reaction given is, => CH3COOH + C2H5OH --------> CH3COOC2H5 + H2OO Given: C2H5...
Q: Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 signifi...
A:
Q: Using the unrounded answer from the previous problem determine q (kJ)(-3512.25kj) for the following...
A: The question is based on the concept of thermodynamics . We have to calculate heat released for form...
Q: 2. The decomposition of a pollutant in water at 15°C occurs with a rate constant of 2.39 y', followi...
A:
Q: Hydrogenation of chlorobenzene in the presence of an auxiliary action is: (Chlorocyclohexane) (Chlor...
A: In this question, we will see the product name after Hydrogenation of chlorobenzene in the presence ...
Q: What is the molarity of a KCl solution made by diluting 35.0 mL of a 0.500 M solution to a final vol...
A: Given M1 = 0.500 M V1 = 35 ml M2 = ? V2 = 150 ml
Q: a) When a voltage is applied and current passes through sodium vapor, electrons are raised from the ...
A:
Q: If the vacuum pump has sufficient capacity and is left on for an extended period of time, the liquid...
A: The boiling point of liquid nitrogen at atmospheric pressure is 77K.
Q: Mass of copper(II) sulfate pentahydrate: 1.5201g Mass of weigh boat: 2.1872g Mass of weigh boat with...
A: The reaction taking place can be written as, => [Cu(H2O)4]SO4•H2O (aq) + 4 NH3 (aq) -------> [...
Q: Consider the following isomers of hexene: H3C CH;CH2 CH3 CH;CH2 H CH,CH2 CH,CH, CH;CH2 CH,CH,CH, H3C...
A: Has the highest MP(melting point ) : ISO Explanation : This has the highest melting point because i...
Q: Which of the following is an example of a covalent bond? A) K - CI B) AI - CI C) H - CI D) Al - Al
A: We have to find the option that contains the covalent bond.
Q: Calculate the restoring force when the ends of a one-dimensional polyethene chain of molar mass 85 k...
A:
Q: determine the standard emf of a cell whose standard reduction potential at the anode is -3.05 V and ...
A: Given standard reduction potential at the anode = -3.05 V standard reduction potential at the cathod...
Q: 2
A: Equilibrium - It is a state in which the concentration of the reactants and the products tend to rem...
Using diagrams draw a full retrosynthetic scheme for the synthesis of the following Target molecule. it should lead back to ethyl acetoacetate (CH3COCH2CO2Et) and 4-nitrobenzyl bromide (4-O2NPhCH2Br). include disconnections, retrosynthetic arrows, synthons, FGIs and synthetic equivalents and suggested reagents.
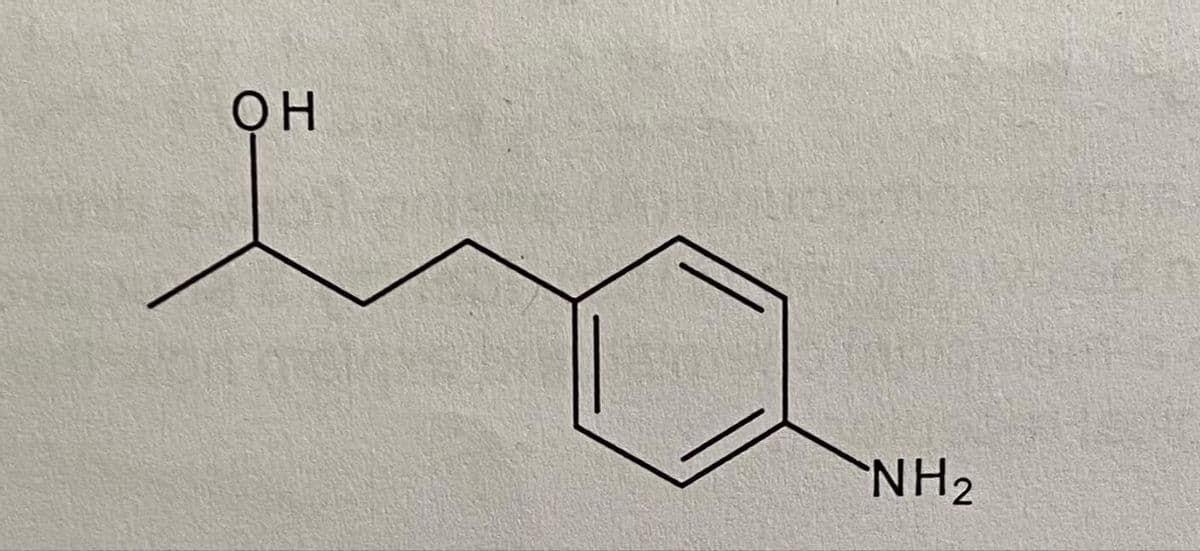
Step by step
Solved in 2 steps with 2 images


