Nhich structure is consistent with the following data? Molecular formula: CHa IR absorptions at -3000, 1600 and 1500 cm 3H singet 3H rpiet 2H trplet 2H S pesks IV O A. V O B. II O C. IV O D. II
Nhich structure is consistent with the following data? Molecular formula: CHa IR absorptions at -3000, 1600 and 1500 cm 3H singet 3H rpiet 2H trplet 2H S pesks IV O A. V O B. II O C. IV O D. II
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterL4: Proton (1h) Nmr Spectroscopy
Section: Chapter Questions
Problem 5E: For each structure below, use numbers to indicate chemically equivalent and distinct hydrogens,and...
Related questions
Question
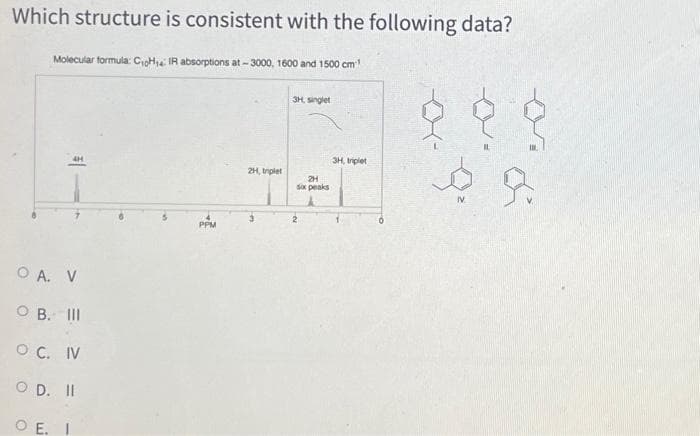
Transcribed Image Text:Which structure is consistent with the following data?
Molecular formula: CioHa IR absorptions at - 3000, 1600 and 1500 cm
3H, singlet
3H, triplet
2H, trplet
2H
Sox peaks
IV
2
PPM
O A. V
O B. II
O C. IV
O D. II
O E. I
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole