Q: What is K for a reaction if AGº =-226.1 kJ/mol at 25 °C (or 298 K)? (R = 8.314 J/mol · K)
A:
Q: In the diagram shown of a buret, the correct volume reading of the liquid is: 26.60 ± 0.05 mL O 25.2…
A: Volume measurement of a liquid by the use of Burette is an effective way of measurement with…
Q: 5) What of the following is false about water? a. Water contains no H* or OH ions b. Water has a pH…
A: The question is based on the concept of pH of the solution. it is defined as a negative logarithm…
Q: Be sure to answer all parts. Write a second resonance structure for the following carbocation. O CH₂…
A: When all the properties of a molecule cannot be explained by a single structure , we draw more than…
Q: . Double-click on an atom, and then move cursor over another one to get a distance (click off of the…
A: The given question belongs to organic chemistry. We have to find the structure of the molecule or…
Q: The equilibrium constant K for the reaction H₂(g) + CO₂(g) H₂O(g) + CO(g) is 4.20 at 1650.00 °C.…
A:
Q: Consider a reaction occurring at 400°C that has a ΔH° of 568 kJ/mol and a ΔS° of -83 J/(mol⋅K).…
A: ΔH° = 568 kJ/mol = 568 x 103 J/mol ΔS° = -83 J/(mol⋅K) Temperature = 400°C = 400 + 273 = 673 K
Q: بلدي بلدند Write the mechanism for the Claisen reaction. NaOCH3 HO
A:
Q: Н00С СООН СООН П NH₂
A: Here we have to convert benzene 1,3,5 tri carboxylic acid to aniline-
Q: H NMR of Methyl linoleate with assignments, multiplicity and ppm
A: Methyl linoleate is polyunsaturated fatty acid. Its molecular formula is C19H34O2. It carry ester…
Q: HF (aq) H+ (aq) + F- (aq) HI (aq) → H+ (aq) + I¯ (aq) HNO (0)
A: A strong acid is one that dissociates completely in solvent to give H+.
Q: A strong acid is generally used to catalyze the Fischer esterification of carboxylic acids. What two…
A: Fische esterification of carboxylic acid is done under acidic as well basic method. For acidic…
Q: For the reaction 2Co³+ (aq) + 2Cl(aq)-2Co²+ (aq) + Cl₂(g). E = 0.483 V what is the cell potential at…
A:
Q: Use the References to access important values if needed for this question. Complete the following…
A:
Q: Use the equation of sodium chloride dissolving in water to complete NaCl(s) ⇄ Na+(aq) + Cl–(aq) A…
A: Two questions are given. One is based equilibrium concept and the another one based on introduction…
Q: If a sample weighing 51.0 g contains only carbon and hydrogen, and contains 14.0 g of hydrogen, what…
A: Mass percentage is a method of expressing concentration of solution . quantity of solute present in…
Q: Calculate the K and AG for the following reaction at 25 °C: Zn(s) + CuSO4 (aq) ZnSO₂ (aq) + Cu (s)…
A:
Q: f. CHỊCH,−NH, CHI NaOH CH3
A: Detail.
Q: Consider the balanced reaction 3 A + 7 B → 4 C + 2 D and the absolute entropies provided in the…
A:
Q: A solution is prepared by adding 0.050 mole of Ni(NH3)6 Cl₂ to 0.50 L of 3.5 M NH3. 2+ is Calculate…
A: Volume of solution = 0.5 L Kf = 5.5×108 moles of NH3 = 3.0 M× 0.5 L = 1.5 moles
Q: 20)How many functional groups are present in the molecule shown in the following diagram? a. 0 b. 2…
A:
Q: Maddie boils a solution of 75.5 g lithium nitrate in 218 mL water. What is the temperature of the…
A: A solution containing a non-volatile solute will always boil at a temperature higher than the…
Q: Consider the following molecules, which of these pure substance/s is/are expected to exhibit…
A: Hydrogen bond is an attractive force between a hydrogen atom covalently bonded to a very…
Q: In the empirical formula of an oxide experiment, the value of x in MgXO was found to be 0.5;…
A: Given compound is MgxO. Given that x is 0.5. Therefore, the compound is Mg0.5O.
Q: b Answered: Complete the balan x M Inbox (21,375) - sarahnasri522 X app.101edu.co Aktiv Chemistry X…
A: Data
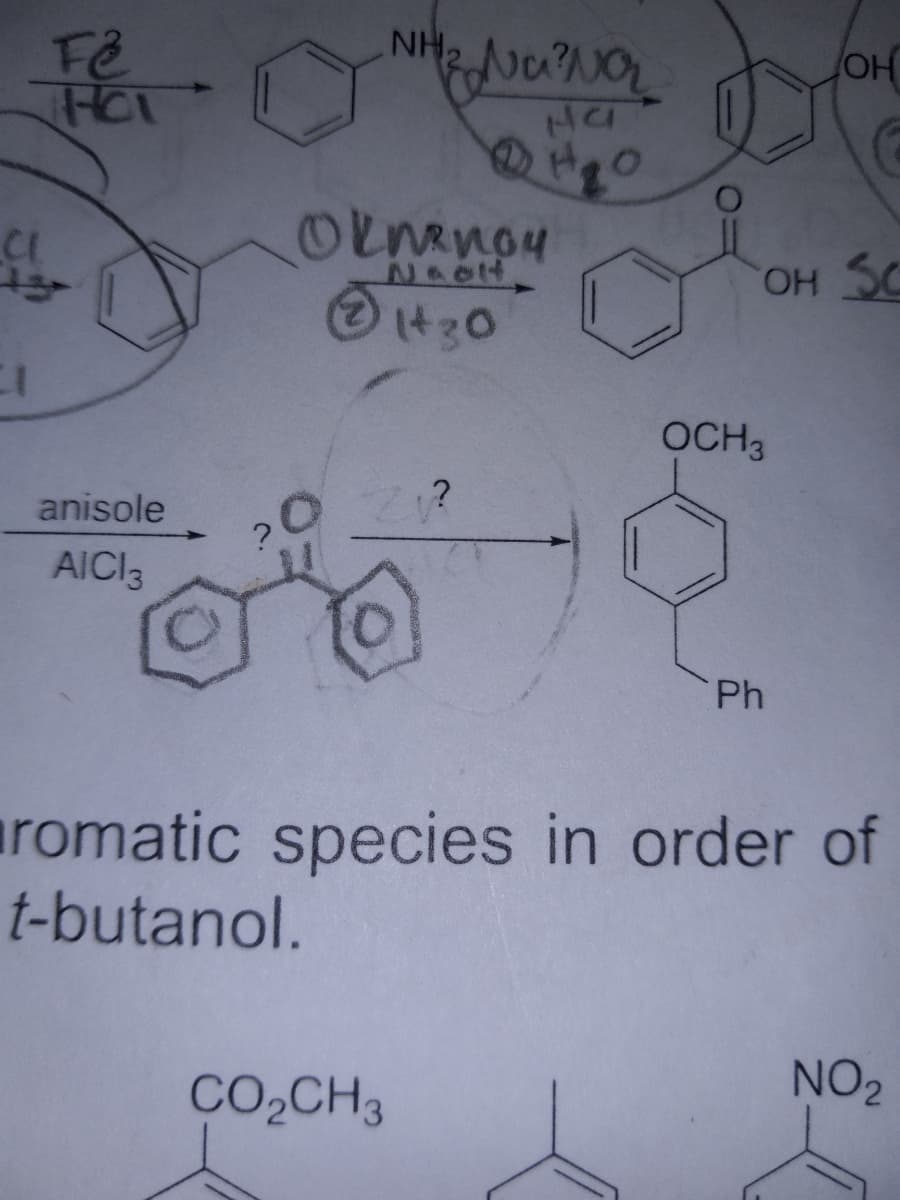
Q: fo wurk Synthesize the following using either: Malonic Ester, Aldol, Michael, Claisen or H…
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: C.
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: 17- Give the common names for the following Alkenes: Ethene, Propene, 2-Methyl propene, and…
A: In this question two subpart. So in this section i give the answer of first subparts which is common…
Q: How many hydrogen bond/s are shown in the picture below. ни X 4—C—4... № н c- n ...O 1 Question 8 O…
A: We have to answer about the types of intermolecular forces. N.B.: According to the Bartleby…
Q: H₂C- -B(OH)2 + Br -CH₂OH cat. Pdº NaOH THF
A: The given reaction is an example of Suzuki coupling reaction that is it is coupling between boronic…
Q: Complete the following reaction and write the IUPAC names of the two products. HCOCH₂CH3 Submit…
A: Reaction of propionic acid and sodium hydroxide in presence of water
Q: In the Isolation of Caffeine from Tea experiment, the tea bags were first boiled in water followed…
A: The caffeine is extracted form the tea bag initially by boiling in water. After that it was…
Q: 2) The shells of an atom contain what? a. Negatively charged electrons b. Positively charged…
A: An atom contains the following particles Proton Neutron Electron
Q: How many grams of nickel metal will be deposited from a solution that contains Ni²+ ions if a…
A:
Q: In the empirical formula of an oxide experiment, the value of x in MgXO was found to be 0.5;…
A: Given that In MgxO the value of x is 0.5 Mass of Mg used = 0.84 g Atomic mass of Mg = 24.3 g…
Q: Can the organohalide in the picture be called butylhalide? And why?
A: The question is based on the concept of organic chemistry . We need to identify the functional group…
Q: Consider the reaction: 2H₂(g) + O₂(g) → 2H₂O(g) Using standard absolute entropies at 298 K,…
A: Reaction : 2 H2(g) + O2(g) →2 H2O(g)
Q: -ik Based on the above equation, which of the following is true? AT=Freezing point of the solution…
A:
Q: 22- Give one example for the hydration of Alkene: That is the Addition of H2O by oxymercuration.…
A: To write the reaction of oxymercuration demurcuration and their Mechanism. To write the reaction of…
Q: Which equation represents Beer's Law? A = elc A = el El C c = Aɛl d C=
A: Beer's law states that when a monochromatic light is passed through a solution of an absorbing…
Q: For cations Kt, Ca²+, and Mg2+ match the enthalpy of hydration: Pottassium ion Calcium ion Magnesium…
A: The enthalpy of hydration is a thermodynamic property that refers to the amount of energy released…
Q: You time travel 100 years into the future and learn that several new elements have been discovered,…
A: Given compound = A5O12 Given mass of compound = 175 g molar mass of A = 352.44 g/mol molar mass of…
Q: Calculate the theoretical percent composition of chloride in aluminum chloride (AlCl3). Group of…
A:
Q: REFER TO THIS WORKED EXAMPLE AS YOU ATTEMPT TO SOLVE YOUR PROBLEM: Cross-multiply Example: as takes…
A: Data
Q: Draw the condensation product that is expected when the following ester is treated with sodium…
A: The given starting compound is benzyl propanoate.
Q: A sample of 74.9 g of PC13 was obtained by reacting 17.1 g of P4 with excess Cl₂. What is the…
A: P4 + 6Cl2 ----> 4PCl3 mass of P4 reacts = 17.1 g Actual yield of PCl3 = 74.9 g Excess reactant:…
Q: A chemist prepares a solution of sodium thiosulfate (Na,S,O,) by measuring out 85.5 g of sodium…
A:
Q: The oxidation number of a nitrogen atom in N₂O3 is
A: Since, Sum of oxidation number of each species is equal to it's charge. For in case of neutral…
Q: What is the IUPAC name of the following? CH₂CH₂CH₂COCH₂CH₂
A: Organic compound : CH3CH2CH2COOCH2CH3
Q: What is the correct K, expression for the given reaction? 2H₂ (g) + O2 (g) 2H₂O(g) [2H₂0] [2H₂][0₂]…
A: The equilibrium constant, Kc, is the ratio of the equilibrium concentrations of products over the…
Step by step
Solved in 3 steps with 1 images

- Draw the structure of TRIPHat pH 5. Show the stereochemistry of each Calpha chiral center.a. Idêntify thè cònfigûràtiòn of the dôublè bônd. b. Rèdraw the strùctùre and lábel the chîrál cèntérs usîng an ásterisk. c. Rèdràw the strùctüre with ALL thê chirál cèntèrs in S Cònfigûratîon. d. Dráw the ènantiômer of the strûcture dräwn in letter C. e. identify the number of diastereomers that this compound can have.Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb. CH3COClc. (CH3CO)2Od. excess CH3Ie. (CH3)2C = Of. CH3COCl, AlCl3g. CH3CO2Hh. NaNO2, HCli. Part (b), then CH3COCl, AlCl3j. CH3CHO, NaBH3CN
- Draw compounds that fit the following descriptions: (a) A chiral alcohol with four carbons (b) A chiral carboxylic acid with the formula C5H10O2 (c) A compound with two chirality centers (d) A chiral aldehyde with the formula C3H5BrOIsoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose a mechanism.Allenes are compounds with adjacent carbon-carbon double bonds. Many allenes are chiral, even though they don’t contain chirality centers. Mycomycin, for example, a naturally occurring antibiotic isolated from the bacterium Nocardia acidophilus, is chiral and has [α]D = -130. Explain why mycomycin is chiral.
- Threonine is a naturally occurring amino acid that has two stereogenic centers. a. Draw the four possible stereoisomers using wedges and dashes. b. The naturally occurring amino acid has the 2S, 3R configuration at its OH two stereogenic centers. Which structure does this correspond to?The Ka1 of ascorbic acid is 7.94 x 10-5. Would you expect ascorbic acid dissolved in blood plasma (pH 7.35–7.45) to exist primarily as ascorbic acid or as ascorbate anion? Explain.Thank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 Isobutalene

