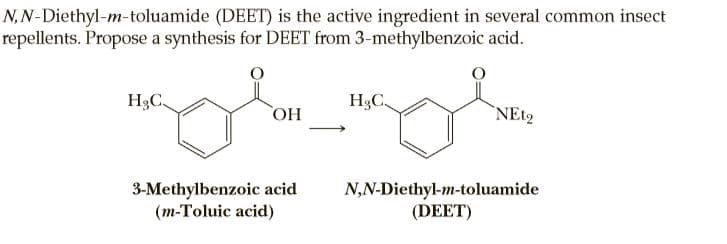
N,N-Diethyl-m-toluamide (DEET) is the active ingredient in several common insect repellents. Propose a synthesis for DEET from 3-methylbenzoic acid. H3C. H3C, HO `NE12 3-Methylbenzoic acid (m-Toluic acid) N,N-Diethyl-m-toluamide (DEET)
Q: 2. Phenobarbital `NH C2H5 Synthesis: OEt EtO NaOEt OEt - co OEt OEt Eto OEt H3C Br NaOEt H2N `NH2…
A: The steps involved in the following synthesis are the abstraction of proton to form nucleophile,…
Q: Starting from nitrobenzene as a starting matenial, the following reaction schemes will lead to the…
A:
Q: Br 1. KOt-Bu KBr HOt-Bu Br Aqueous acetone H20 +. 2. H3C HBr Br H OH optically active racemic a =…
A:
Q: What is the intermediate before the final product of the following reactions? NaOH H* heat Oa. Na O…
A:
Q: 1 Me منا Ci 53 48 O AICI3 dracon CI Acylax 54 CO₂H
A:
Q: What are the reagents to complete this reaction? H;/Pt NABH4, MEOH H2/Lindlar's catalyst Li, EENH2,…
A: Reduction is the process to gain the electrons or to loss the hydrogen Or to gain the hydrogen…
Q: NaOCH; 7. What is the major product? a. b. С. d. e. not a.-d.
A:
Q: Which set of reagents will turn acetic acid into 2-bromoacetic acid? O Br2/NAOH/ETOH O…
A: The given conversion can take place with the help of HVZ Reaction to form a alpha-halo carboxylic…
Q: 1. Treat 2-pentanone with the following reagents. Write the major organic product of the reaction…
A: Since we know that Tollen's test is used to distinguish between aldehyde and ketone. Since we know…
Q: но. OH но. OH HOOC COOH Reagents 2 NANH2 CH2l2, Zn(Cu) a. g. b. 2 HCHO then HCI h. RCO,H H2, Pt H2,…
A:
Q: In the Gabriel synthesis of primary amines, N-potassiophthalimide is used as a source of the…
A: This is a Gabriel phthalamide synthesis reaction. Through this reaction 1° amine is produced by the…
Q: A carboxylic acid is formed when an a-haloketone reacts with hydroxide ion. This reaction is called…
A:
Q: 4. а) Föreslå en sannolik mekanism för reaktionen nedan! O-CHs "CH3 OCHo CH3 Br CH3OH "CH3 ČH3 ČH3…
A:
Q: Which sequence of steps will give the highest yield of a carboxylic ester product? Mix 0.1 mole of…
A: Esterification is based on nucleophilic addition followed by substitution.
Q: b- find A and B form the following :- ALO 1) cyclohexanol- A 250 °C r H₂ Pd Li Cu 3) 2-chloro…
A: The given questions are general organic reactions. The desired products are given below.
Q: 2. Phenobarbital NH C2H5 Synthesis: OEt EtO - co OEt OEt NaOEt OEt OEt Eto H3C `Br NaOEt H2N `NH2…
A:
Q: Propose efficient synthesis for the following reaction. (10 pts) an O2N `NO2
A: The answer is given as follows
Q: The following series of reaction steps will yield 1) But-1-yne plus NaNH2; 2) the product of Step 1…
A:
Q: OH CH3 1. NaOH 2. -Br KOH / EtOH / reflux H 1. Aq. KMnO4 2. LOCH 3 -CH,CI /AIC13 NaBH4 then aqueous…
A:
Q: (a-b) (c-d) (e-f) (g-h) Î Predict the product or provide reagents needed to complete each…
A: Phthalic acid when heated releases H2O to form phthalic anhydride.
Q: An organic chemist reacts (S)-2-butanol with PBr3 in pyridine, removes the major organic product,…
A: The above reaction is proceed by SN2 mechanism in which attack of -OCH3 is done in back side of OH.…
Q: 3-Pentanol KMnOw/A Propionic Acid 2) H ) )H.HS(CH)SH Cyclopentanone 2) Rancy Ni b(cat), H*…
A:
Q: 22. How many of these reactions produce a product named 2-butanol? H₂SO4 H₂O A Br₂ CH₂Cl₂ A. 1 B. 2…
A: In this question we have to tell that how many reaction will produce product as 2-butanol
Q: Me HN From 2-Ethoxybenzoic acid 3. Pr O=S-N NMe
A: This is the synthesis of Viagra. It is a PDE5 inhibitor
Q: Question attached
A: In the 1st step addition of Hbr at the double bond takes place. In the 2nd step, Br is replaced by…
Q: Complete the following synthesis by filling in the missing reactants, reagents, and products.• Boxes…
A: The first step would involve Dieckmann condensation i.e. intramolecular reaction of diesters with…
Q: 1. Which carboxylic acid(s) would decarboxylate when heated to 100-150 °C? OH Ha II a. I b.I с. П d.…
A: Decarboxylation of the carboxylic acid: The carboxylic acid that contains a carbonyl group at the…
Q: Why is it necessary to incorporate the benzyl group on the chloroamine used to convert B to C?
A:
Q: 6. Provide the product resulting from the following reaction. 1. BH3, H20 2. H202 NAOH, H20 OH OH A…
A:
Q: 18) C,H,OK" Write all the elemination and substitution products 19) H;CO, 20) KMnO,
A: "Since, you have posted multiple question, we will solve only first 3 for you, to get other question…
Q: 10. N‚N-Diethyl-m-toluamide (DEET) is the active ingredient in many insect-repellent preparations.…
A: In this question we are asked to synthesise N,N-Diethyl-m-toluamide from Toluene as shown below-
Q: b- find A and B form the following:- ALO 1) cyclohexanol- А 250 °C B₂ Pd Li Cu A+ B- 3) 2-chloro…
A:
Q: Nou are herforming estowification reaction using 5 g of 1- heranoic acid CmW = 116.16 gmol-1) and…
A: Mass of 1-hexanoic acid = 5 g molecular weight of 1-hexanoic acid = 116.16 g/mol Molecular weight…
Q: a) Propose a synthesis route to prepare 3-methylhexan-3-ol from pentan-2-one. Note that more than…
A: For a synthesis of compound 3-methylhexan-3-ol from pentan-2-one in two step reaction we follow a…
Q: e. Br CH,CH,CH3 2. Draw the structures corresponding to the following names: а. 3, 5-Неptadien-1-yne…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: triisopropylsilyl-Cl reagent reacts only once with the starting material. Me Me HO OH Me NH₂ iPr…
A: Given,
Q: Choose the best option for the electrophile precursor to 2-hexanol. OH CH 30 Br nucleophile B O H…
A: During the formation of alcohol from alkyl halide there is water act as a nucleophilic while alkyl…
Q: Select the most appropriate reagents for the transformation. HO, The best reagents are: О 1. NaBH,…
A:
Q: 4) Provide a synthesis of the following compounds using the given starting material and any other…
A:
Q: e) OH OH 1. 2.
A:
Q: 4. using retrosynthetic analysis, determine which compounds could lead to the alkene shows below in…
A: Potassium tert-butoxide can carry out elimination to give least substituted alkene. So the product…
Q: Methamphetamine can exist in two possible forms, shown below: Mr. Mema is trying to convert…
A: A contains nitrogen atom with l.p. on it. Hence, it is basic in nature. B is a salt. We know that…
Q: Which set of reagents will accomplish this reactions? он HO,C CO,H HO,C ČOH он А MCPBA, aqueous…
A: Correct option is ( A ). Mcpba/ aqueous NaOH/H2O
Q: he major product from the reaction of H20 with 2-methyl-2-butene in the presence of acid catalyst is…
A: 2 methyl -2 butanol is the product .
Q: Ketones, Aldehydes and Epoxides: Assume "then H,0" is included if a protonation step is needed R K V…
A: The answer is given as follows
Q: Steroids are a class of organic compounds containing a specific tetracyclic skeleton (four rings),…
A: Functional groups in a drug have different properties. They have been altered to change Water /lipid…
Q: In the Gabriel synthesis of primary amines, N-potassiophthalimide is used as a source of the…
A: Here we have to write products formed in step-1 and step-2 in the following given Gabriel synthesis.…
Q: What is the major organic product obtained from the following sequence of reactions? Mg H20 Br Et,0…
A:
Q: drw product followng reactions w starting material 1-3
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Starting with benzene, what is a possible product of this series of reactions?: 1) HNO3 & H2SO4 2) Fe, HCl 3) (CH3)2CH-Br, FeBr3 4) NaNO2, HCl, 0-10 oC 5) HBF4 2-fluoro-3-isopropylaniline m-fluoro-benzenesulfonic acid o-fluoro-isopropylbenzene 2-amino-benzenesulfonic acidWhen certain aromatic compounds are treated with formaldehyde (CH2O), and HCl, the CH2Cl group is introduced onto the ring. This reaction is known as chloromethylation. (a) Propose a mechanism for this example of chloromethylation. (b) The product of this chloromethylation can be converted to piperonal, which is used in perfumery and in artificial cherry and vanilla flavors. How might the CH2Cl group of the chloromethylation product be converted to a CHO group?Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):
- A carboxylic acid is formed when an a-haloketone reacts with hydroxide ion. This reaction is called a Favorskii reaction. Propose a mechanism for thefollowing Favorskii reaction. (Hint: In the first step, HO- removes a proton from the a-carbon that is not bonded to Br; a three-membered ring is formedin the second step; and HO- is a nucleophile in the third step.)Phenol plus 1) 1/2 I2, HNO3; 2) Br2 + AlCl3; 3) HNO3/H2SO4; can yield ___________. 2-bromo-6-iodo-4-nitrophenol 1-bromo-3-iodo-4-amino-phenol 3-bromo-5-iodo-4-nitrobenzene 2-bromo-3-iodo-4-hydroxy-aniline 2-bromo-3-iodo-4-nitrophenolScheme 1: Reagents: (a) acetic anhydride, sulfamic acid, 60°C,30min,91%; (b) Et3SiH,I2,0-5°C,30min,82%. 1) Draw the FULL, ARROW-PUSHING MECHANISM 2) Provide a verbalization of the mechanism for your reaction mechanism 3) What is happening at each step? What atom is nucleophilic? Which atom is electrophilic? Which bonds are made? Which bonds are broken? 4) Imagine you are trying to describe the reaction mechanism and I can't see your display - how would you describe the mechanism in a way that I know, that you know, what you are talking about.
- i) 3-methyloctane-2,3-diol 11) 3-chloro-4-ethoxy-2-methylcyclopentanamine iii) N, N, 3,3-tetramethylbutan-2-amine iv) 1-ethoxy-2,5-difluorohexan-3-ol 6) 5-isopropoxy-3-isopropyl-6-propoxy-4-propylcyclohexane -1, 2-diol draw of the following names in skeletal formsgas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.A carboxylic acid is formed when an a-haloketone reacts with hydroxide ion. This reaction is called a Favorskii reaction. Propose a mechanism for the following Favorskii reaction. (Hint: In the first step, HO- removes a proton from the a-carbon that is not bonded to Br; a three-membered ring is formed in the second step; and HO- is a nucleophile in the third step.)
- The Baylis–Hillman reaction is a DABCO (1,4-diazabicyclo[2.2.2]octane) catalyzed reaction of an a,b-unsaturated carbonyl compound with an aldehyde to form an allylic alcohol. Propose a mechanism for the reaction. (Hint: DABCO serves as both a nucleophile and as a base in the reaction.)Which of the following reactions will not produce a carboxylic acid? Benzaldehyde + hot, conc'd KMnO4 1) Ph-N2+ + KI 2) BrMgCH=CH2 in ether, followed by H3O+ sec-butylBenzene + hot, conc'd KMnO4 1) Benzene, HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2; 4) CuCN; 5) dilute acid and heat 1) Benzene and acetyl chloride & AlCl3; 2) HCl & Ni Thank you!From 10 to 14. can i get an explanation on how to choose the appropriate products for these reactions? thank you



