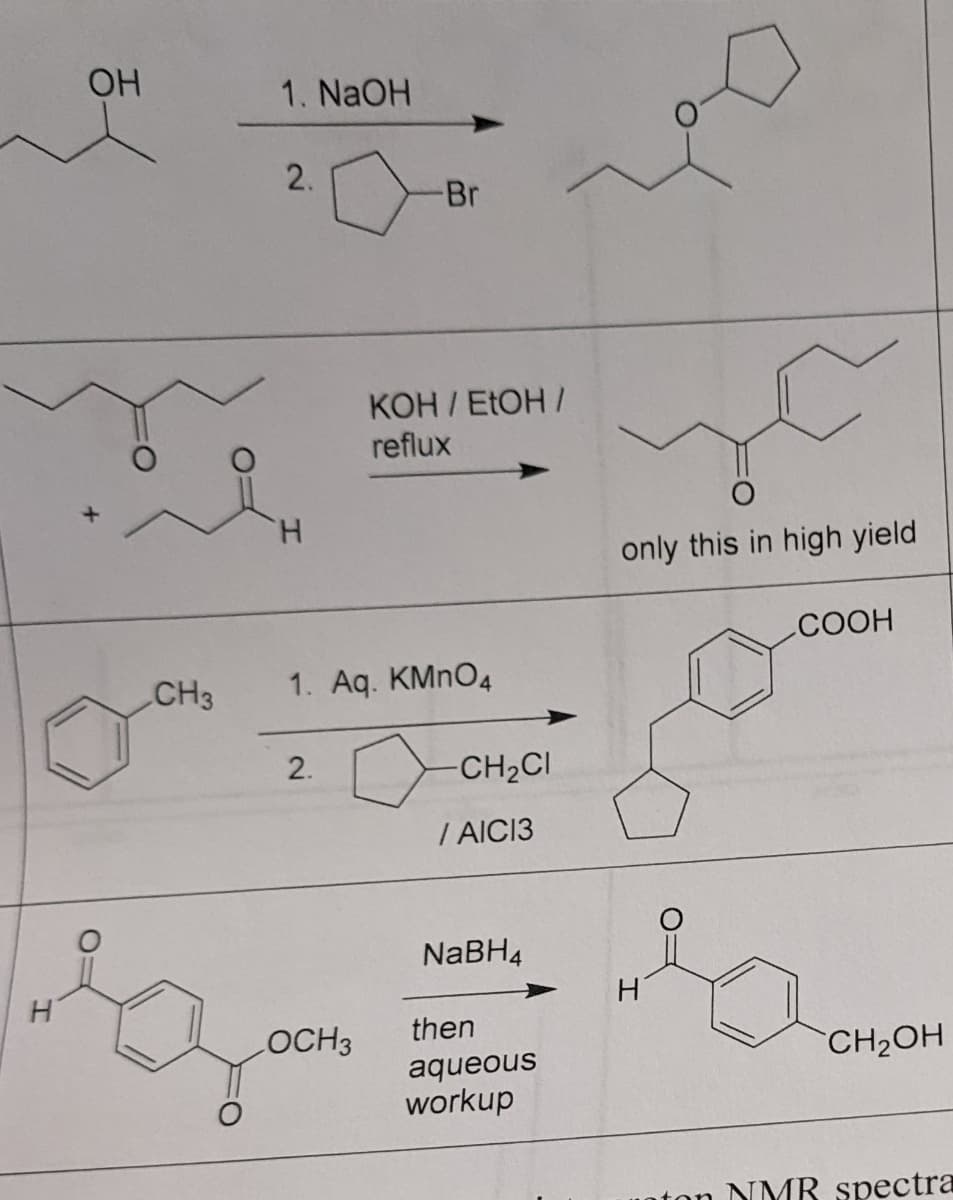
OH CH3 1. NaOH 2. -Br KOH / EtOH / reflux H 1. Aq. KMnO4 2. LOCH 3 -CH,CI /AIC13 NaBH4 then aqueous workup only this in high yield COOH CH₂OH H
Q: 21. For the following equilibrium: H2C=CH2 (g) + H2 (g) €→ C2H6 (g) AH° = -136 kJ Employing Le…
A: Le Chatelier principle states that the equilibrium will be shifted in the direction where the…
Q: 11.) Which of the following entropies are arrange in an increasing order? B. solid < liquid < gas D.…
A: Multiple question given here.
Q: Science Books presented by Macmillan Leaning Consider the table. Metal Tm (k) All (kJ/mol) T All…
A:
Q: 8. Provide reasonable mechanisms for the conversions below. CHs H,SO,
A: We have to provide the mechanism of the given reaction.
Q: molecular formula of the compound?
A:
Q: Therefore, the rate of reaction is 0.005 mol/s 1. Iron reacts with oxygen as shown in the balanced…
A:
Q: Exercise 34. Show the reaction of the carbonyl group of each aldehyde or ketone with one molecule of…
A:
Q: The graph shows the titrations of two unknown acids with the same base. Identify the stronger acid…
A: At half equivalence point, pH = pKa = -log(Ka) Below is the curve showing half equivalence point-
Q: Why is it necessary to have different methods for appl
A:
Q: b. Thermodynamics Circle the correct choice or fill in the blanks in each of the following: f. Which…
A: All the given problems are related to the chemical thermodynamics. Entropy is a measure of…
Q: Which wavelength of light has the highest frequency? a. 16 mm b. 4 nm c. 16 nm d. 4 mm
A:
Q: 39. DNA isolated from human cheek cells was found to contain 20% guanine. What percent of the…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Circle one of the following substances which would be most soluble in H2O?
A:
Q: Add or subtract the following measurements. Be sure each answer you enter contains the correct…
A: Answer giving correct number of significant digit of add or subtract of this given questions.
Q: Chemistry What is the name of the following structure? C0-CH2 CH2 CH3 CH3
A: First of all we have to select main chain .Then we will give numbering and during writting name of…
Q: 10. Write a complete and balanced nuclear equation for the following. (Note: Z= # of protons of that…
A:
Q: 30. How would you best classify the following molecule? CH OH HO A) hydrolyzable lipid B)…
A: a) Hydrolysable lipids contain phospholipids, neutral waxes, glycolipids and waxes. It can be…
Q: Show total valence electrons and draw the Lewis structure for the following compounds. 1. KrF4 2.…
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [N] [H.]…
A: For the reaction, aA + bB ----> Products Rate law for the reaction is, Rate = k [A]x [B]y…
Q: Provide the structural formula for the following: (skeletal formula is fine)…
A:
Q: coordination number
A:
Q: Neither the change in enthalpy nor the change in entropy is, by itself, sufficient to determine…
A:
Q: 15.) Determine the change in entropy of the reaction: SO2 (g) + H202 (1) H2SO4 (1). The standard…
A:
Q: The wavelength associated with the 3rd line in the Lyman series is [ Select ] nm. This wavelength is…
A: Each line of the Lyman series ends to ground state i.e n=1. Each line of Balmer series ends to…
Q: rate law ar
A:
Q: ne the heat associated with a decrease in the temperature of 375g of 5°C to 24.6°C. The specific…
A: Given, Mass of water = 375 g Initial Temperature = 31.5 °c Final Temperature = 24.6 °c Heat…
Q: Le Chatelier’s principle allows scientists to predict shifts in equilibrium. If you increase the…
A: Le Chatelier's principle says that if any change is happen to a reaction (system) at equilibrium,…
Q: What is the pressure of 1.51 g of nitrogen gas confined to a volume of 0.105 LL at 30 ∘C?
A:
Q: Indicate the two most probable reagents for the following substrate/product pair. S-
A: Thioacetal formation
Q: Identify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this…
A:
Q: 1.) The enthalpy of formation of BaCl2(s) is -858.6 kJ/mol, which of the following st
A: Since you have posted multiple questions, the answer for first question is given below. Kindly…
Q: oue Matchthe tollowing cleavage with ithe corre. ct goro lucts. 1 Eth CH eth
A:
Q: If a student prepares 10 liters of a 0.01M NaOH solution by starting with 25 mL of a stock solution,…
A:
Q: For the reaction CH;CHO (g) → CHạ (8) + CO (g) @ 60OK, the rate expression is Rate = k[CH;CHO]".…
A: Given reaction is CH3CHO (g) -----> CH4 (g) + CO (g) The rate expression is rate = k[CH3CHO]m…
Q: What is the ratio of [CH3CO0"]/[CH3COOH] in a acetate buffer (pkK, = 4.76) at pH 4.20? %3D [CH3COO…
A:
Q: 1-bromo-1,3,5-triethylcyclohexane + ethanol >?
A:
Q: If pH of a solution increases what happens to the following things; [H3O*] РОН and [OH¯] O a. falls;…
A:
Q: Show the chain reaction (initiation ,propagation and termination) step in a polymer known as PMMA
A: Mechanism of chain polymerization consists of three phases, called initiation, propagation, and…
Q: 0-2M sodium A salution contains 101x 10 Subfide and 5.12 x10-3M ammonjum cabonde. Saled barium…
A: Answer: When the ionic product of a sparingly soluble salt becomes equal to its solubility product,…
Q: 2. Helium diffuses at a rate of 80.0 mL/s. How fast would krypton diffuse under the identical…
A:
Q: Select ALL the terms that apply to the relationship of the indicated protons. CH3 но- Hb H- На- H-…
A:
Q: A solution is prepared at 25 °C that is initially 0.47M in chloroacetic acid (HCH,CICO,), a weak…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: 3. The compound C4H6 has several isomers. Draw each of the isomers and determine the molecular…
A:
Q: A gas sample at STP contains 1.47 g oxygen and 1.66 g nitrogen. What is the volume of the gas…
A: We are given the mass of two gases , we have to find the volume at STP
Q: Nhat is the maximum number of electrons contained in an orbital of type (x)? Of type How many…
A: According to the concept of quantum chemistry, orbitals are the places in which probability of…
Q: Item Answer Total Number of ATP Produced from one Glucose Molecule Total Number of NADH Obtained…
A:
Q: Proteins vary widely in structure, whereas nucleic acids have rather uniform structures. How do you…
A: Answer - Protein - Protein is an important part of a healthy diet. Proteins are made up of chemical…
Q: 9. Consider the reaction CH4 (g) + H20 (g) = CO (g) + 3H2 (g). Which of the following will happen if…
A: As per Boyles law, at constant temperature and fixed mass of gas, the volume of the gas is…
Q: What is the correct IUPAC name of the following compound? Problem viewing the image, Click Preview…
A: The ester is formed from alcohol and carboxylic acid. The name of an ester is written as an alkyl…
Q: Exercise 43. Explain why the radical polymerization of styrene forms branched chains with quaternary…
A: The mechanism of free radical polymerization of styrene to form linear polystyrene is shown below:
Step by step
Solved in 3 steps with 3 images

- L.5 Hi Experts! Can you please help me complete the tables? please kindly also include some sample calculations. THANK YOUUU SOO MUCHH!! Results (GIVEN) Solvent Trial Volume of NaOH (mL) Concentration of NaOH (M) Volume of Solution (mL) Concentration of Solution (M) Water 1 15.6 0.4664 10 2 15.4 0.4664 10 Average Ether 1 31.9 0.0823 10 2 31.8 0.0823 10 Average Table 1. Data for 1 M Acetic acid. Solvent Trial Volume of NaOH (mL) Concentration of NaOH (M) Volume of Solution (mL) Concentration of Solution (M) Water 1 7.6 0.4664 10 2 7.7 0.4664 10 Average Ether 1 20.2 0.0823 10 2 18.3 0.0851 10 Average Table 2. Data for 0.5 M Acetic acid. Solvent Trial Volume of NaOH (mL) Concentration of NaOH (M) Volume of Solution (mL)…Hi! can anyone here help me to create a schematic diagram about the experiments: " 21. Preparation of Ethyne" by Pdst Chemistry, and "Activity 10: Functional Group Tests for Hydrocarbons" by FRETZ RMT since i dont know what a schematic diagram is, and what processes/contents should be in it. thank youwith a theoretical yield of 80% compute for the percent error of the following data Vial weight= 18.3501 vial + fluorescein weight=18.442 resorcinol = 0.1502g Phthalic anhydride= 0.1022g
- outline a separation scheme for isolating pure aspirin. lab video: https://www.youtube.com/watch?v=Y4NMpO1xI8U(9.29 × 105/8)-20.81 = with correct sig figsPls solve this question correctly in 5 min i will give u like for sure What evidence supports or refutes that during an experiment, purified extract is pure caffeine?
- Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)! ( give full derivation asap within 15 min, otherwise dislike)Any special precautions or purifications of the reagents required? Limiting and excess reagent? Why is excess necessary? 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude…



