Q: A metallurgist needs to make 60 kg of architectural bronze, which consist of 12 parts copper, 1 part...
A:
Q: 2-chloro-3,3-dimethylhexane
A:
Q: Nitrous acid is a weak acid. Would you expect solutions of nitrous acid and nitric acid, of equal co...
A:
Q: NaSH Br 1) Hg(OAc)2, CH3CH2OH 2) NABH4 CH;CH,CH,Br
A:
Q: Pls answer letter d
A: The answer is as follows:
Q: The density of methanol, a colorless organic liquid used as solvent, is 0.7918 g/mL. Calculate the m...
A: Given density = 0.7918 g/mL.
Q: Calculate the pH and the concentrations of all species halfway to the first equivalent point in the ...
A: pH is the measure of acidity of a solution. It is defined as negative logarithm (to base 10) of hydr...
Q: At 293K the vapor pressure of A is 30mmHg while that of B is 100mmHg. The two forms an ideal solutio...
A: Vapor pressure of a substance is the pressure exerted by the vapor above its solid or liquid state. ...
Q: Ammonia, NH3, Is toxic to a wide range of aquatic organisms including shails, insects, and fish. The...
A: molarity and pH `
Q: Hydrogen and oxygen react to form water, together they are a _____. - compound - molecule - mix...
A: When two or more types of molecules of different compounds are together then they will be a mixture....
Q: What is the initial rate of the reaction in Experiment 4? Initial rate mol/(L · s)
A: Well, first step for the calculation is to determine the rate equation from the given experiments. F...
Q: The activation energy (E,) for a certain biological reaction is 62.1 kJ/mol. If the rate constant (k...
A:
Q: Draw all of the constitutional isomers for C H.
A: Constitutional isomers are those isomers which have same molecular formula but different structural ...
Q: (2) Using the standard reduction potentials listed in the Table, determine which of the following re...
A: The answer is as follows:
Q: How many seconds are there in a solar year (365.24 days )?
A: 1 day= 24h 1h= 60 min 1 min= 60 sec
Q: Enter your answer in the provided box. Calculate the molality of a solution containing 281 g of HCl ...
A: Molality: The mole of the solute dissolved in one kilogram of the solution is known as the molality ...
Q: 3. Calculate the molality of a 6.55 kg sample of a solution of the solute CH;Cla dissolved in the so...
A: Since you have posted multiple questions, we will solve the 1st one for you. Kindly post the other o...
Q: 1. WHAT IS INTERMOLECULAR FORCE? 2. INTRAMOLECULAR FORCE? 3. WHAT IS KINETIC MOLECULAR THEORY?
A: Given questions are : 1. WHAT IS INTERMOLECULAR FORCE?2. INTRAMOLECULAR FORCE?3. WHAT IS KINETIC MOL...
Q: What is the volume of 0.5 moles of gas at STP? Must attach image of work hand written on paper on Qu...
A: Full form of STP is Standard Temperature and Pressure. Standard temperature is 273K and standard pre...
Q: Sodium metal requires a photon with a minimum energy of 4.41 x10-19 J to emit electrons. a. What is ...
A: Given that - Minimum energy of emitted electron from sodium metal = 4.41×10-19 J. Then, Frequenc...
Q: A solution is prepared by mixing 24.56 g of methanol, CH40 (32.04 g/mol); 62.85 g of water (18.02 g/...
A:
Q: Use the following data to calculate the enthalpy of solution of potassium chloride, KCl: A¡H°(s) = -...
A:
Q: Complete and balance the following equation: Perchloric acid and lithium carbonate Add all of the ...
A: The solution is given below -
Q: 3. For Dan, use sketches of the orbitals to show that d., have B2g symmetry i.e. show how the orbita...
A:
Q: 1. Mg Br 2. Br
A: The reaction given is,
Q: In your opinion how many peaks will appear in the 1H NMR spectrum of the compound below. What signal...
A: Proton NMR spectroscopy is an analytical technique which is used for structural elucidation of a com...
Q: If there are 2.37 moles Fe203 then how many moles of Fe and O2 are there ? if there are 11.11 moles ...
A: Given, 1) Moles of Fe2O3 = 2.37 moles Moles of Fe = ? Moles of O2 = ? 2) Moles of Fe2O3 = 11.11 mole...
Q: 1. Using arrow formalism, show how the McLafferty rearrangement might occur in PHCOCH2CH2CH3. What v...
A: McLafferty rearrange is a characteristic fragmentation of carbonyl compound in mass spectroscopy. fo...
Q: 8. Give IUPAC names for the following compounds: (a) CI „Br (b) CH3 (c) NH2 CH2CH2CHCH3 Br (d) CI CH...
A: IUPAC nomenclature is used for naming organic compounds. Full form of IUPAC is international union o...
Q: When 60.0 mL of a 0.400 M solution of HN03 (ag) is combined with 60.0 mL of a 0.400 M solution of Na...
A: The volume of the final solution is = 60.0 mL + 60.0 mL = 120.0 mL The density of the solution is = ...
Q: What will the [Fe(SCN)2+] be at equilibrium, in M?
A: Equilibrium Reaction Fe3+(aq) + SCN-(aq) ⇔Fe(SCN)2+(aq) ...
Q: kindly help me with one Make a complete schematic diagram for the precipitation, separation, and i...
A: The schematic representation is as follows:
Q: Diamond, lattice constant (a) = 0.3567 nm, shares the same crystal structure as Si, FCC with lattice...
A: The scattering of X-rays occurs from the atoms which provides information about the positions of ato...
Q: Put the following acids in order from strongest to weakest. (1 is the strongest and 5 is the weakest...
A: The acid dissociation constant (Ka) value is used to indicate the strength of acids. Greater the val...
Q: Male 0.48 0.36 0.20 0.55 0.45 0.46 0.47 0.23 Female 0.35 0.37 0.27 0.29 Is there any evidence that c...
A: Soln
Q: Scaling X workshe X 4Conform X EWriting X t Data fo Question 6 of 13 Consider the following reaction...
A: This can be solved as
Q: Choose the best set of conditions for a Friedel-Crafts acylation of aromatic compounds. A) CH3C(O)CI...
A: Friedel-Crafts Reaction
Q: Hydrogen and oxygen react to form water, together they are a .... - molecule - compound - neutro...
A: Hydrogen and oxygen are atoms that combine to form water
Q: For the reaction A2 (aq) + B2 (aq) - 2 AB (ag) The initial rate of appearance of AB was 3.0 M/s. Wha...
A:
Q: How many grams are in 2.36 moles of NaOH? (molar mass of NaOH = 39.997 g/mol)
A: We can solve this problem using the "Mole Concept" Formula for number of moles : Number of Moles = M...
Q: Phenanthrene, C14H10, is an aromatic hydrocarbon. Some phenanthrene is dissolved in 50.0 g of benzen...
A: Given, Mass of phenanthrene (C14H10) = ? Mass of benzene = 50.0 g = 0.05 Kg Boiling point of the sol...
Q: 1. PHCO0OH 2. EtLi, then H30* 3. PCC
A:
Q: Nitrogen monoxide, NO, reacts with hydrogen to give nitrous oxide, N20, and water. 2NO(g) + H2 (g) →...
A:
Q: molar mass
A:
Q: The reaction below reaches equilibrium in a closed reaction vessel. What is the effect of adding an ...
A: Given, 2NaHCO3 ⇌ Na2CO3(s) + CO2(g) + H2O(g)
Q: a) What is the meaning of the following symbols? i. ii. E ii. A b) State TWO (2) conditions necessar...
A: a) The meaning of the given symbol is, i) The given symbol is called psi. It indicates the wave func...
Q: Propose a plan to synthesize 2,2,3-Trimethylpentan-3-ol. Explain.
A:
Q: Can someone help me answer this? thank you Pick an organic molecule that you find interesting and de...
A:
Q: _chloride ion bromide ion Lit F- Na* S2- K* Mg2+ Ca2+ 02- N3- P3- A3+ barium ion boron ion
A: While naming anion, 'ite' 'ate' or 'ide' suffix is used with the name of the element. While naming ...
Q: 20. When a Ca atom loses all of its valence electrons, it is isoelectronic with? (Show the ground st...
A: Atomic number of Ca is 20. Isolelectronic species are those which have same number of electrons.
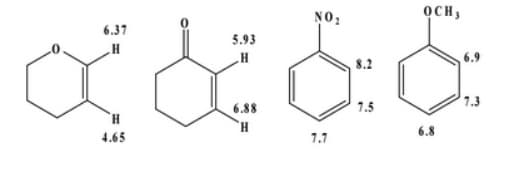
05) How would you explain the chemical shifts for each substance shown below.
Step by step
Solved in 2 steps with 1 images


