Q: 14. Which diene and dienophile would react to give the following Diels-Alder product?
A: Given that : We have to determine the Diene and Dinucleophile used to synthesize the below given Die...
Q: Use short hand electronic configuration for the following Se2-
A: We are given Se2-.
Q: www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLqkt1FLlq7wcPWKzBYGfE9IMFjON2fAaf...
A: Ionic compounds are formed by transfer of electrons between two atoms.
Q: 1
A: Streoisomers Stereoisomers are the special arrangements in space . These are the non super-imposbal...
Q: Calculating volume by combining the volume of simple shapes A chemistry student in lab needs to fill...
A: Given Length - 28.0 cm Width - 18.0 cm Height - 13.0 cm Diameter of round botton flask - 10.5cm Radi...
Q: chapter 12: 11) Calculate ΔS° for each of the three following reactions using the data provided bel...
A:
Q: @@ .l D Qe PIAT Question 17: Which of the following is not an advantage/benefit of solid lubricant? ...
A: Dear student I have given answer to your question in the image format.
Q: An aqueous solution of a weak acid HA (MW=100.0 g/mole) has a mole fraction of HA of 0.23, and a den...
A: mole fraction of HA = 0.23 density of HA = 1.24 g/ml Molecular weight of HA = 100.0 g/mole
Q: A carbon-12 atom has a mass of 12.00000 amu. The mass of a proton is 1.00728 amu, and the mass of a ...
A:
Q: Using the data tables provided, 1. Determine the molarity of NaOH for each of the three trials. Reme...
A: NaOH is standardized with respect of KHP. Moles of KHP=Moles of NaOH. Molarity of KHP×Vol of KHP= mo...
Q: Pls help ASAP. WRITE THE IUPAC NAME
A: IUPAC ( International union of pure and applied chemistry ) is an international organisation that ha...
Q: A Lewis structure with placeholder central atom is shown below. If the charge of the molecule is -1,...
A: According to the given lewis structure the identity of the central atom has been identified below.
Q: Draw a reaction coordinate diagram for a fast, stepwise, exothermic reaction. Label reactants, produ...
A: This is the concept of heat of enthalpy is negative
Q: GRAVIMETRIC ANALYSIS OF A TWO COMPONENT MIXTURE A mixture of NaHCO3 and Na2CO3 reacts with hydrochlo...
A: We have to find mass of sodium carbonate and sodium bicarbonate in 0.5 g mixture
Q: Please draw the mechanism for the nitration of benzene by using a mixture of nitric and sulfuric aci...
A: Nitration of benzene is an electrophilic aromatic substitution reaction. HNO3 - Nitric acid H2SO4 - ...
Q: Predict whether the following would be more likely to be miscible or immiscible. a. NaNO2 and CH3CH2...
A: Given data for interpretation; a. NaNO2 and CH3CH2OH b. K3PO4 and C5H12 c. CH3-O-CH3 and CH3COOH
Q: Based on the information below, determine the rate law. N2 + O2 --> ...
A: Given reaction is N2 + O2 -----> 2 NO Given experimental data is [N2] ...
Q: For the decomposition of ammonia on tungsten NH3 on W → ½ N2 + 3/2 H2 the rate constant i...
A:
Q: Which of the following molecules is antiaromatic? A. cycloheptatrieneB. Cyclopentadienyl cationC. cy...
A: The question is based on the concept of aromaticity. we have to identify which of the given molecule...
Q: A compound decomposes by a first order process. If 25.0 % of the compound decomposes in 60.0 minutes...
A: Answer: Half life of the compound is 145 min.
Q: When the Cu2+ concentration is 4.58×10-4 M, the observed cell potential at 298K for an electrochemic...
A: We are going to determine the concentration of Mn2+ as follows
Q: Copper forms two oxides, Cu2O and CuO. Predict over what temperature range this reaction is spontane...
A:
Q: standard thermodynamic data in the tables linked above. Calculate AG for this reaction at 298.15K if...
A: Dear student I have given answer to your question in the image format.
Q: Sample Identity and Rationalization 1 3
A:
Q: Determine the temperature of a reaction if K = 1.2 x 10-6 when AG° = +20.6 kJ/mol.
A: Given: ΔGo=+20.6 kJ/mol=+20.6 x 103 J/mol K=1.2 x 10-6 Temperature, T=?
Q: For the exothermic reaction PCl3(g)+Cl2(g)⇌PCl5(g) Kp= 0.200 at a certain temperature. A flask is ch...
A:
Q: Determine the enthalpy (in kJ/mol) for the reaction 4 NH: (g) + 5 Oz (g) → 4 NO (g) + 6 H2O (g).
A:
Q: (a) Predict the sign of AS with explanation for each process. (i) H2O (g)→H2O (1) (ii) Solid carbon ...
A: Entropy represent degree of randomness or measurement of randomness. It is an extensive properties a...
Q: Which of the following statements is false? As the hydroxide ion concentration increases in a solu...
A: The question is based on the statements. we have to identify which of the given statement is incorre...
Q: Pls pls aolve question-2 pls solve accurate it's important...if you can do both pls do both i will t...
A: Equation representing reaction of Mg (excess) with HCl : Mg (escess) + 2 HCl → MgCl2 + H2
Q: The Kb of a weak base, Z, is 3.75 x 10−12. What is the Ka for its conjugate acid, HZ+?
A:
Q: Consider the reaction: 3FE203(s) + H2(g)– →2F@304(s) + H20(g) Using standard thermodynamic data at 2...
A:
Q: Fill in the name and empirical formula of each ionic compound that could be formed from the ions in ...
A: The solution is given below -
Q: Calculate the half-life at 100 molecules, 50 molecules and 25 molecules for a 1st order reaction and...
A:
Q: Help
A:
Q: Consider the following reaction at 25 °C: 3 NiO(s) + 2 NH3(g) → 3 Ni(s) + N2(g) + 3 H20(g) If AG° = ...
A:
Q: What is the value of ΔG°rxn for the reaction: C--->2A+B given: 2A+B--->ΔG°rxn=150.5kJ/mol
A: Given: Reaction-1: 2 A + B --> C ΔG°rxn=150.5kJ/mol Reaction-2: C--> 2 A + B ΔG°rxn=?
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction:Fe3+(aq) + ...
A: We have to find the value of equilibrium constant for given reaction and predict about G°
Q: Which of the following can behave as an electrophile? A. HBr B. Cl- C. CH3O- D. CH3CH2CH2CH3
A: During the reaction , mainly two type of Substances or species play an important role to starting an...
Q: Determine K for a reaction at 200 K if AG° =22.8 kJ/mol. (R = 8.314 J/mol · K)
A: Given is ΔG° = 22.8 kJ/mol = 22800 J/mol T = 200 K R = 8.314 J/mol-K We are to find the equilibrium ...
Q: Write the empirical formula for at least four ionic compounds that could be formed from the followin...
A: Ionic compounds are those which are formed by complete transfer of electrons.
Q: Benzocaine (ethyl p-amino benzoate) is a local anesthetic compound. Design its synthesis using the n...
A: In organic chemistry, inter conversion of organic molecule takes place from one form to another form...
Q: Can u please explain how to solve it ! Thanks.
A: Multiplication of significant figures : The answer of multiplication must be reported in the same ...
Q: Compound X has a molar mass of 134.12 g•mol -1 and the following composition: element mass % carbon ...
A: Element Mass % Carbon 44.77 % Hydrogen 7.52 % Oxygen 47.72 %
Q: What is the pH of a 0.193 M w. acid HA solution? Ka = 6.61 x 10−6?
A: The acid is weak so it weakly dissociates in the solution. That is it gives an H+ ions slowly in to ...
Q: What will be the pH of a solution containing 0.200 M NaH2PO4 and 0.200 M Na2HPO4? Ka(NaH2PO4) = 6.2 ...
A: Given, Molar concentration of weak acid, NaH2PO4 or H2PO4- = [H2PO4-] = 0.200 M Molar concentration...
Q: A robot spacecraft returned samples from the planetesimal 98765 ALEKS, located in the outer Solar Sy...
A: Given data, Mass of 101Ru = 100.9 amu Mass of 102Ru = 101.9 amu Percentage abundance of 101Ru = 90.9...
Q: If the number of electrons in an atom is 51 and its atomic mass number is 125, calculate How could y...
A: The element of the electrons 51 and mass number 125 represents the Antimony (Sb). Antimony belongs t...
Q: 16. For the following reactions, determine if they will follow SN1 or SN2 mechanism (explain your an...
A: The question is based on the concept of organic reaction mechanism. we have to predict the mechanism...
Q: a-Draw the chair structure of the compound. b-Draw the ring-flipped structure of the compound. c-lde...
A:
Write the following synthesis with the mechanism using the appropriate reagents.
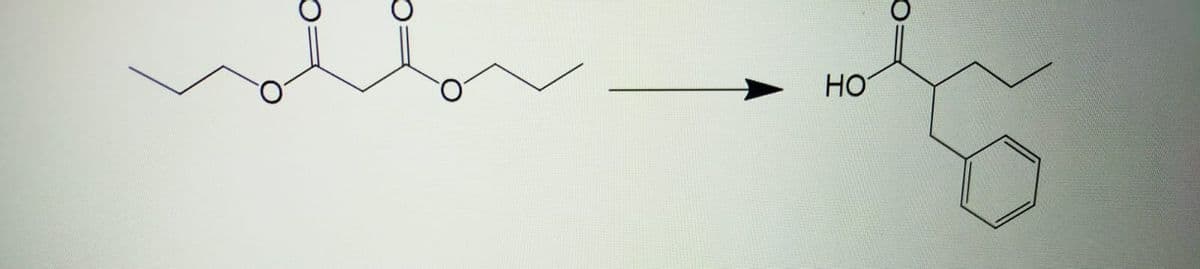
Step by step
Solved in 2 steps with 2 images


