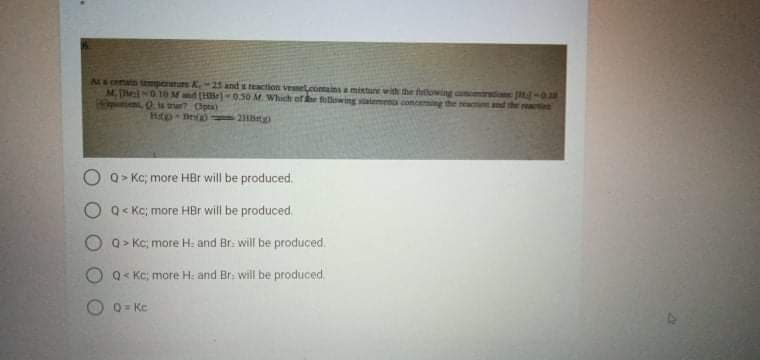
Nsono mpern K-21 and s eaction venet.comainsamisture wit the htowing M. hel010 Md (He)-050 M. Which of folowing e conceng the e, ts r Opta) Hp De 2t and
Q: PUと55UVと 15 100 f a, aud tue yues 15 increas g at avate of 201kP a /hin At what rate is the Uolume…
A: According to Boyle's law, pressure is inversely proportional to volume. Hence PV=constant. As the…
Q: - (i) The mass collision (electronic) stopping power (Sm = S/p) of a 5-MeV proton in silicon is 59.0…
A:
Q: A saturated Na₂CO₃ solution isprepared, and a small excess of solid ispresent (white pile in…
A: SOLUTION: Step 1: (a). the initial solution has the following equilibrium: Na2 12CO3 (s)↔ 2 Na+…
Q: Ex. What mass of AgNO,(mw=169.9) is needed to convert 2.33 g of Na,CO, (mw=106.0)
A: The balanced reaction between AgNO3 and Na2CO3 to form Ag2CO3 and NaNO3 is given as: Na2CO3…
Q: Product isoto prs after nuclear de cay Fill up by di fferent K-deca y 92 B-deay Eleckon Caplave…
A: Nuclear reaction: A nuclear reaction must cause a transformation of at least one nuclide to another.…
Q: arer フ-cト チ子 185 (d) | りう 3-11
A: Solution: We know the proton NMR spectroscopic technique is one of the best and commonly used…
Q: What particle(s) are produced in the following reaction? U238 +016-F250m+. 92 8 -
A: Fermium (Fm) is a man-made element. Its atomic number is 100. Fermium (Fm) is a trans uranium…
Q: iuestion 16 is given 4 0.05 mg dose of wck has a helf-ife of 6 hours. How long will it be until only…
A:
Q: Suppose one food irradiation plant uses a 137Cs source while another uses an equal activity of 60Co…
A: In food irradiation, food is treated with ionising radiations (like gamma rays, X-rays, electrons)…
Q: ralence point. Gi nt (in grams) of t 5738
A:
Q: Calculate the percent yie material S into target T 81% 85% A. B.
A: Interpretation - To calculate the percent yield of the following seven -step synthesis that converts…
Q: nd 107.0 kPa? * D 47.3 g/mol DNone of these is correct.
A: Note : As per guidelines allowed to answer first one Ideal gas equation : It is the hypothetical…
Q: Alec F. Litvin (Student section: 65A) CHEM 111 - Fall 2020 Main Menu Contents Grades Groups Course…
A: The balanced reaction is :3O2(g) →2O3(g)→∆Grxn° = Σ Gf (products)°- Σ Gf (reactants)°…
Q: Suppose there was a release of 0.572 mole of particle A and 0.792 mole of particle B and both…
A: Given the moles of particle-A released = 0.572 mol Given the energy of particle-A = 5.73 MeV We have…
Q: Thor x E THO X Thor X IXL X Inbo x AAsse X A Clas X E THO X A Phot x Laur X Laur x OnC x As X + → C…
A:
Q: Exre 136 Tate 12 147) Calcylate He percont by mae listed firet in the formule Teras Pect amethane,…
A: Applying formula of percentage of elements % of element= atomic weight of element/molecular weight…
Q: Complete the following reaction: 27 Al13 30p15 P +_[al_ + a →
A: In nuclear reaction, Balanced equation means number of proton and mass number in reactant side is…
Q: Xenon-135 is especially troublesome because, (a) It can lead to transient prompt supercritical. (b)…
A: Welcome to bartleby!
Q: What is the process of elution in extracting Ba-137 from the Isotope Generator (2-3 sentences would…
A: Answer: During the process of elution Ba-137 is wiped out of the isotope generator. The elution…
Q: sid ate X OWLV2 | Onlinet X al 112021 X C Ais*war…
A:
Q: An information source with P(Xi)=(0.15, 0.1, 0.03, 0.4, 0.02, .0.1, 0.2) with a time of (1, 4, 2, 3,…
A:
Q: The half-ide of years. Whet fraction of the atoms in a semple of Sr would remain 175 years later?
A: 29 years from this data, we can find the decay constant λ here. lamda=0.693/29=0.24years The T hub…
Q: 5-34. The isotope Cu is unusual in that can decay by y, B*, and ß“, emission. What is the resulting…
A: The gamma decay can of can be given as : The resulting nucleotide is in lower energy state.
Q: sputreometrey ? pieobable fragmentadion patteen 1 [o (co)3 ( NO) what melas fable plale in mau eaues…
A: mass spectrometry
Q: How long will it take for 98.6% of a sample of 18879 Au to decompose (t1/2 = 8.8 mins)?
A:
Q: ♡21 Cinn tre to llowing correctly Balaned ractin Hlow many grams of watr are produed from 89grams of…
A: Applying concept of mole and limiting reagent.
Q: ll InCl/Cu(OT1 Dod. CH Cl, n OMe Meo OMe Meo 77% InCly/Cu(OTf D00. CH Cl, it Meo NO2 NO2 Meo- 72% dr…
A: Solution has been provided in 1st and 2nd step.InCl3/Cu(OTf)2 work as a Lewis acid and help making…
Q: In an alpha particle (42He) scattering experiment, using a thin gold (19779Au) foil, the initial…
A: Given, kinetic energy = 2.0MeV
Q: The radioisotope 41Ar decays by β-(negative beta) emission to an excited level of 41K that is 1.293…
A: Beta decay results in isobars and in beta minus decay neutron is converted into a proton, electron…
Q: The sample of 4x.is a B emitter has an activity of 0.5 uCi. In10 days the activity of the sample has…
A:
Q: Homewock (1) When a sample ol 4-heplanone WAS iradjilad iiE 150 s wah 1 radiation wath4 power outpul…
A:
Q: 31; logP = 4.56) on some- eldrin has a half-life of ab ermeability of about 3 x 1 ate during the…
A: The partial pressure as, A= 0.400 atm, B= 0.400 atm.
Q: IMG_9523.HEIC Open with Preview y.dorlocator=assig ot The following are unbalanced equations. They…
A: The fundamental types of chemical reactions are: 1. Combination reactions: In these reactions, two…
Q: 9. Using the reaction HPO42– + 2H+ + 2e– ⇌ HPO32–+ H2O(l) E°= –0.234 V And…
A:
Q: Tyes 28.94 A a0 Sr activity today is S.76 dpm was calibrated to 1.00Mc, its. im=605 O Calculate how…
A:
Q: A radioisotope of argon, 37Ar18, lies below the “band of stability.” One would predict that it…
A: Isotopes are elements having the same atomic number but different atomic mass compared to that…
Q: Suppose there was a release of 0.572 mole of particle A and 0.792 mole of particle B and both…
A: Given the accelerated energy of both the particles = 5.73 MeV We have to convert 5.73 MeV to kJ/mol
Q: What's More Activity 4 - Complete the nucleosynthesis reactions below Given: Composition of each…
A:
Q: 6- Radiation source in AAS device is hallow cathode lamp. 7- The PH of a solution Mg(OH)2 can affect…
A:
Q: old
A:
Q: Question 15
A: Given: ΔHo of the reaction = -890.0 KJ Mass of CH4 = 82.1 g Molar mass of CH4 = 16.04 g/mol Chemical…
Q: Suppose there was a release of 0.572 mole of particle A and 0.792 mole of particle B and both…
A: Given the moles of particle-A released = 0.572 mol Given the energy of particle-A = 5.73 MeV
Q: Suppose there was a release of 0.572 mole of particle A and 0.792 mole of particle B and both…
A: GIVEN: Moles of particle A = 0.572 mol Moles of particle B = 0.792 mol Energy = 5.73 MeV. To…
Q: In the equationP - S +X, the particle 15 16 represented by x is A. He В. n C. H D. e
A: Since you are posted with multiple questions. As per the rule, I am answering the first question…
Q: 103 + AsO33- l2 12 + AsOs2-
A:
Q: 225AC? 17. By what series of emissions does Pa disintegrate to
A:
Q: A T8Kx Undexgats a i)- write decy euatim ? ii)-For a ameral double E-Cab tade decap ,e tas ting from…
A: Electron capture decay reaction is a first order radio active decay process in which by capturing…
Q: The nitrogen and protein content of a 2.8391 g sample of milk was determined using Kjeldahl…
A: Given: Volume of HCl = 25.00 mL Concentration of HCl = 0.250 M Volume of NaOH = 31.65 mL…
Q: the W the att Vactive between two 12 13 adjacent ions of an atom is 14 15 Jand the rePuisive energy…
A: Attraction is the stabilization energy and repulsion is disstabilization energy. Binding energy is…
Q: Choose the correct answer to fill in the product blank ("???") below. 1on → 14156BA 9236Kr 3 on ???…
A: Interpretation: The blank space of the given reaction is to be completed. Given reaction: n01 +…
Step by step
Solved in 2 steps with 1 images

- page 5) can you give me the example for initial HA: 1.00M in table?Solve correctly please, (With explanation need, Gpt/Ai wrong ans not allowed)Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)
- In the synthesis of hydrocarbons, the carbon source is carbon dioxide. Although the CO2 concentra?on in the atmosphere raises at a drama?c speed, point sources are probably the easier sources for a PtX process. Iden?fy 3 possible point sources, explain why CO2 is formed and what challenges each of the three CO2 streams presentsEe.62. With explanation please ....PLS SEND ME ANSWER WITH DETAILED EXPLANATIONS AND I WILL GIVE YOU LIKE SURE.

