Q: 3. Provide stepwise mechanisms, using curved arrows to show the movement of electrons, for the…
A: The given reactions areWe have to provide the mechanisms of those reactions.Concept: Radical…
Q: Magnesium metal reacts with gaseous oxygen in a combination reaction. Write a balanced equation to…
A: A combination reaction, also known as a synthesis reaction, is a type of chemical reaction in which…
Q: Nitrogenous Mustard has been used as an alkylating anti-cancer agent. nitrogen mustard Demonstrate…
A: Nitrogen mustards are alkylating agents that can covalently bind to DNA, leading to DNA damage and…
Q: At a certain temperature, 0.900 mol SO, is placed in a 5.00 L container. 2SO₂(g)2SO₂(g) + O₂(g) At…
A: The equilibrium reaction is as follows:The initial moles of = 0.900 molThe equilibrium moles of =…
Q: 50. What is the specific heat of a substance if 7600 cal are required to raise the temperature of a…
A:
Q: What is Kc for the following reaction at 25C? Br₂(l) + Cal₂(s) CaBr₂(s) + 1₂(s) K=4 x 1023 AH=-149…
A:
Q: In a study of the rearrangement of cyclopropane to propene at 500 °C (CH2)3 CH3CH=CH₂ the…
A: Rearrangement reaction of cyclopropane Graph :- In[(CH2)3] versus time in seconds gives a straight…
Q: A chemist dissolves 527. mg of pure perchloric acid in enough water to make up 120. mL of solution.…
A: To calculate the pH of a solution of perchloric acid, we need to determine the concentration of the…
Q: A chemist titrates 210.0 mL of a 0.3206M ammonia (NH3) solution with 0.3768M HCl solution at 25 °C.…
A:
Q: Question 4 ohe object The initial volume of water in a graduated cylinder is 20.0 mL. You place an…
A:
Q: 19. The half-life of carbon-14 is 5730 years. A plant fell into a tar pit 18,400 years ago before…
A:
Q: What is the basic reaction for; Cr2O72—(aq) + HNO2 (aq) → Cr3+ (aq) + NO2— (aq
A: Cr2O72- (aq) + HNO2 (aq) → Cr3+ (aq) + NO3- (aq)At reduction half: Cr2O72- (aq) → Cr3+ (aq)Balance…
Q: 47. The heat content of a system is equal to the enthalpy only for a system that is at constant A.…
A: The mathematical expression of the First law of thermodynamics is dU = dq + dW = dq - pdV.The…
Q: The molecule shown to the right is: a. a secondary amide D. a secondary amine . a primary amide d. a…
A: Amine functional group consists of a nitrogen atom bonded to one or more alkyl or aryl groups, while…
Q: Consider the 50 mL 0.1 M phosphate buffer at a pH of 7.4. If 0.5 mL of 1 M HCI was added to the…
A:
Q: 4. In the reaction mechanism below: a) Which step is the rate determining one? b) What is the…
A: Reaction mechanism of a reaction is given.
Q: 3.Complete the reactions below by drawing the major product or products. Indicate product…
A: In a SN2 reaction, the nucleophile attacks from the opposite side of that of the leaving group. The…
Q: 21. The initial oxidation number of the element oxidized is initial oxidation number of the element…
A:
Q: One of the emission lines in the Balmer series has a wavelength of 656.47 nm. This corresponds to…
A: The given wavelength is = 656.47 nm = 656.47.We know that for the Balmer series, the value of the…
Q: The addition of a reactant to a system can accelerate the rate of the forward direction. True O…
A: According to Le Chatelier's principle, if we disturb a reaction at equilibrium by changing the…
Q: You are titrating a 10.00 mL aliquot of a 0.3675 M solution of a diprotic weak acid (pKal = 2.125,…
A: When a diprotic acid is titrated with a strong base, two equivalence points are observed due to the…
Q: Br ph + OH CI r Reaction Br H₂O - + H₂O OH OH₂ OH₂ + Br OH + ci + Br franqer + OH + Relative Rate 1…
A: Polar aprotic solvent is used in SN2 type reaction. Rate of SN2 mechanism depends on both the…
Q: n the box below give the Newman projection of the most stable conformer of the molecule hown below…
A: In the molecule, 2-Methyl-3-Ethyl-hexane. The Bond across C2 and C3 is free to rotate and the exist…
Q: A student performed the first determination in Part 2. They reacted 59.11 g of 1 M HCl with 0.7347 g…
A: Mass of Hcl = 59.11 g Molarity of HCl =1M Mass of magnesium oxide = 0.7347 g Initial temperature Ti=…
Q: pls explain what other compounds or reagents would be needed to form it given you have HCl and…
A: The given compound and reagents are 4-methyl-2-hexene and HCl.We have to synthesized…
Q: The J = 2 to J = 3 rotational transition in a certain diatomic molecule occurs at 120 GHz. Find the…
A:
Q: A conductivity cell filled with 0.1 M KCI at 25°C has a measured resistance of 24.36 2. The…
A:
Q: After propyne reacts with excess Br2 in chloroform, the product eventually becomes
A:
Q: Arrange the single covalent bonds within each set in order of INCREASING polarity. If: • most polar…
A: Higher the electronegativity difference between the atoms, higher will be the polarity of the bond.
Q: What are the compounds A and B? Br KOH/EtOH heat A (Major) H₂O. H* B
A: The more stable form of 2-methyl bromocyclohexane is trans. In this reaction, KOH in EtOH acts as a…
Q: 20-The functional unit of mature glycogen is tropocollagen .which structure is found between these…
A: Mature glycogen refers to fully formed glycogen granules found in cells, particularly in liver and…
Q: 1 of 253 What is the difference between 6.32 - 2.334 ? A B с D LU E 4.0 3.9 4 3.99 3.986
A: Here, in this question we determine the difference between 6.32 - 2.334 using significant figures.
Q: Give typed full explanation not a single word hand written otherwise leave it
A: The given reaction is an example of Friedel-Craft alkylation.-OCH3 is an electron donating group…
Q: The image at right shows which of the following aspects of enzymatic catalysis? Active site +…
A: To describe the substrate specificity , various enzyme catalyzed reactions occur . So we will…
Q: Which of the following reaction doesn't give benzaldehyde: as a product?
A: Oxidation of primary alcohol using pyridinium chloro chromate (PCC) gives aldehyde.
Q: In chemical reactions, the rate of reaction is often influenced by various factors. Consider a…
A: Introduction:Catalysts play a vital role in accelerating chemical reactions by providing an…
Q: A mixture containing 0.0392 M A(g) and 0.0452 M Big) is allowed to come to equilibrium at 300 K The…
A: Ans-Given that-Initial concentration [A] = 0.0392M [B] = 0.0452MAt…
Q: Calculate the pH of a 0.10 M solution of NaCIO.
A: NaClO is basically a salt, prepared form weak acid HClO and strong base NaOH. When NaClO or ClO- is…
Q: The positron has charge +e and mass equal to the electron mass. Calculate in electronvolts the…
A: Here we have Bohr's Energy Formula of nth electron, Energy of the electron in nth orbit, En
Q: Use the data provided below for the data in your lab (Final report, calculate the percent recovery…
A: Initial mass of the mixture (mixture mass) = 0.8 g Final mass of iron = 0.23 g Final mass of zinc…
Q: What are the solutions to the followin x^(2)+8x+15=0
A: The solutions of the given equation are:--5 and -3
Q: Each of the following organic compound names is incorrect. Explain why it's incorrect, then give the…
A: IUPAC nomenclature rules for alkanes:• Identify the longest continuous carbon chain.• Identify the…
Q: For the following reaction, 49.3 grams of potassium hydrogen sulfate are allowed to react with 18.7…
A: Given:Mass of potassium hydrogen sulfate = 49.3 gMass of potassium hydroxide = 18.7 gKnown:Molar…
Q: Consider a hypothetical chemical reaction: A + B ⇌ C + D. The reaction is carried out in a closed…
A: In this explanation, we will explore the factors contributing to the change in reaction rates and…
Q: The water pressure on Mustafa as he dives is increasing at a rate of 0.992 atmospheres (atm) per…
A:
Q: An analyst has timed an operation for 50 cycles. The average time per cycle was 11.5 minutes, and…
A: To find the standard time for the metal-cutting operation, we need to calculate the normal time and…
Q: 1. Ammonium Phosphate reacts with Aluminum Acetate 2. Ammonium Phosphate reacts with Potassium…
A: Chemical equation:Ammonium phosphate reacts with aluminium acetate to form ammonium acetate and…
Q: H3SCH3(g) + OH(g) → Products For a constant O2 concentration of 150 torr, it has an activation…
A: The relation between rate constant and temperature is given by the Arrhenius equation----The unit of…
Q: The pressure above a pure sample of solid Substance X at -10. °C is lowered. At what pressure will…
A: The phase diagram represents the different phases of the substance and the pressure and temperature…
Q: Use full-headed or half-headed curved arrows to show the movement of electrons in each reaction. a.…
A: The full-headed curved arrow indicates the transfer of 2 (two) electrons.The half-headed curved…
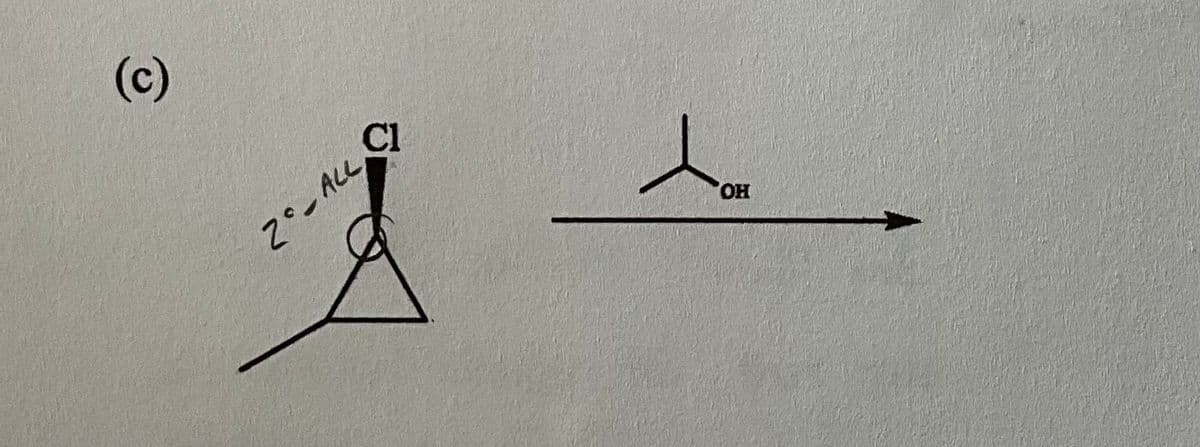
Can someone help help walk me through how to do this problem?
For the reaction below: (1) draw the product(s) formed under the stated conditions, (2) determine the dominate mechanism followed (E1, E2, Sn1, or Sn2) and (3) explain your choice.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images


