Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.16QAP
Related questions
Question
U.4 A. 1-6 part 2
kindly help me with this question please. i also attached the topic for this problem
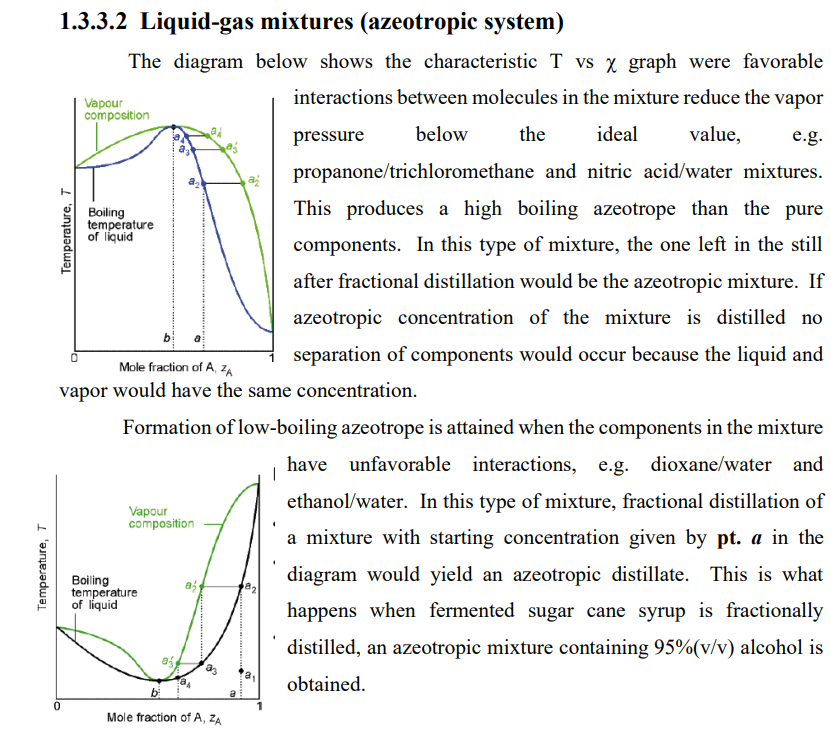
Transcribed Image Text:1.3.3.2 Liquid-gas mixtures (azeotropic system)
The diagram below shows the characteristic T vs x graph were favorable
interactions between molecules in the mixture reduce the vapor
Vapour
composition
pressure
below
the
ideal
value,
e.g.
propanone/trichloromethane and nitric acid/water mixtures.
This produces a high boiling azeotrope than the pure
Boiling
temperature
of liquid
components. In this type of mixture, the one left in the still
after fractional distillation would be the azeotropic mixture. If
azeotropic concentration of the mixture is distilled no
b
separation of components would occur because the liquid and
Mole fraction of A, zA
vapor would have the same concentration.
Formation of low-boiling azeotrope is attained when the components in the mixture
have unfavorable interactions, e.g. dioxane/water and
ethanol/water. In this type of mixture, fractional distillation of
Vapour
composition
a mixture with starting concentration given by pt. a in the
diagram would yield an azeotropic distillate. This is what
Boiling
temperature
of liquid
happens when fermented sugar cane syrup is fractionally
distilled, an azeotropic mixture containing 95%(v/v) alcohol is
obtained.
b
Mole fraction of A, ZA
Temperature, T
la
Temperature, T

Transcribed Image Text:Queetion #z
caly the gosthie & l
Illustrate graphic
behavieurof binary
the positrie &
negatire deñation from icdea
mixtures using T against X plot.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

