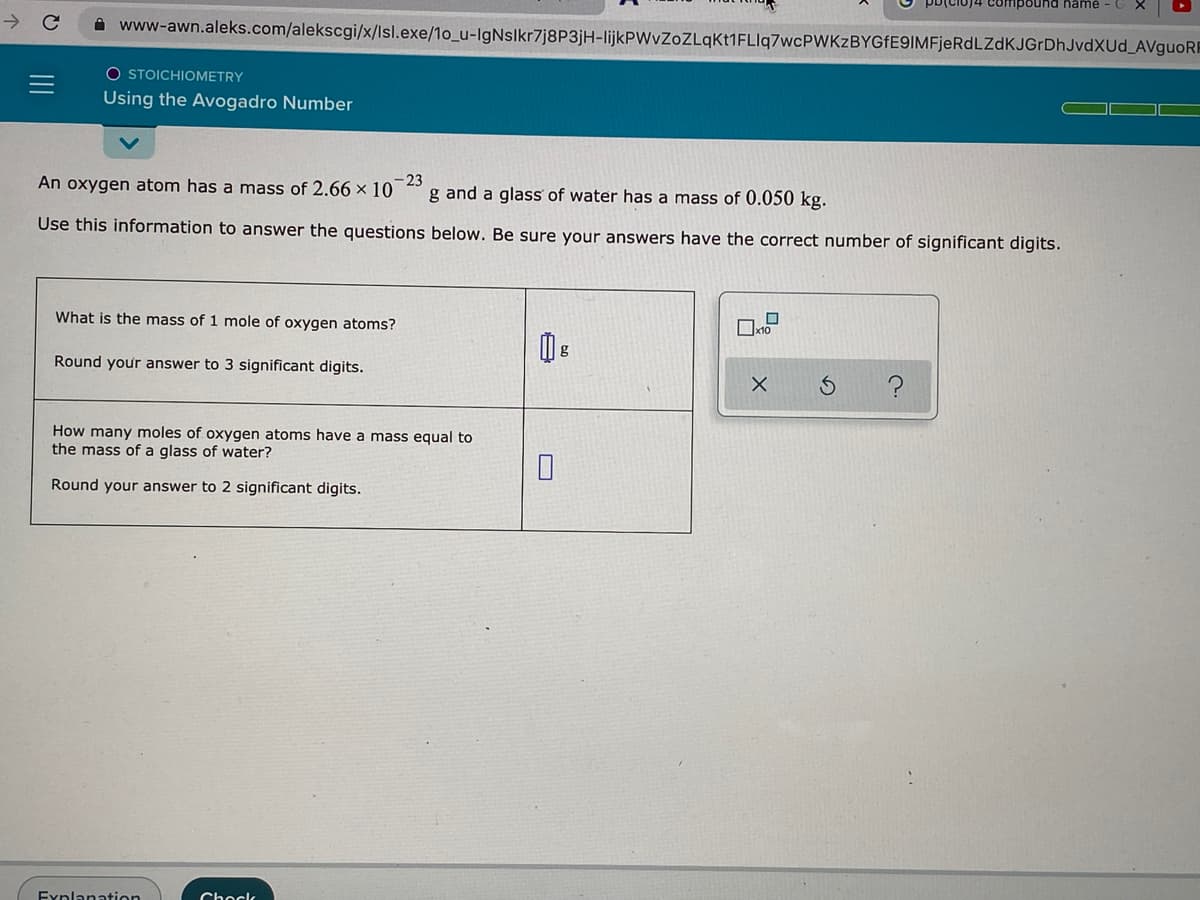
O STOICHIOMETRY Using the Avogadro Number An oxygen atom has a mass of 2.66 x 10 - 23 g and a glass of water has a mass of 0.050 kg. Use this information to answer the questions below. Be sure your answers have the correct number o What is the mass of 1 mole of oxygen atoms? g Round your answer to 3 significant digits. How many moles of oxygen atoms have a mass equal to the mass of a glass of water? Round your answer to 2 significant digits.
O STOICHIOMETRY Using the Avogadro Number An oxygen atom has a mass of 2.66 x 10 - 23 g and a glass of water has a mass of 0.050 kg. Use this information to answer the questions below. Be sure your answers have the correct number o What is the mass of 1 mole of oxygen atoms? g Round your answer to 3 significant digits. How many moles of oxygen atoms have a mass equal to the mass of a glass of water? Round your answer to 2 significant digits.
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter8: Gases
Section: Chapter Questions
Problem 155CP: Methane (CH4) gas flows into a combustion chamber at a rate of 200. L/min at 1.50 atm and ambient...
Related questions
Question
Transcribed Image Text:pound hame
->
A www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLlq7wcPWKzBYGfE9IMFjeRdLZdKJGrDhJvdXUd_AVguoRE
O STOICHIOMETRY
Using the Avogadro Number
An oxygen atom has a mass of 2.66 × 10
23
g and a glass of water has a mass of 0.050 kg.
Use this information to answer the questions below. Be sure your answers have the correct number of significant digits.
What is the mass of 1 mole of oxygen atoms?
Round your answer to 3 significant digits.
How many moles of oxygen atoms have a mass equal to
the mass of a glass of water?
Round your answer to 2 significant digits.
Fynlanation
Chock
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

