Q: 100 Transmittance %T 0 A 4000 cyclobutanol 3000 2-butanone. OH ethyl vinyl ether…
A: Ir spectrum gives information about functional group which are present in given molecule. This…
Q: will
A: According to the question, we need to determine the redox reaction.
Q: 7. Consider these three labelled specific volume vs. temperature plots that may be displayed by…
A: The curve a shows amorphous solid. The curve b shows semicrystalline solid The curve c shows…
Q: Identification Complete each statement. Write your answer at the end of each question. 101.Chemical…
A: The correct answer is given below
Q: Rank the following groups in order of decreasing priority according to the Cahn-Ingold-Prelog…
A:
Q: The synthesis is a simple two-stage process with catalytic hydrogenation used for reduction of the…
A: First step is Micheal addition group. Second step is reduction of nitrile to amine.
Q: For the following reaction, ide Xo 요 i 1 || IV Acid Base Acid I and II B. I and III I and IV D. II…
A: 2) Bronsted-Lowry base is a base which accepts a proton(H+). Compound I accepts a proton from…
Q: 2. The following rates of reaction were obtained in three experiments with the reaction 2 NO(g) +…
A:
Q: In the measurement 0.5030 L, which digit is the last significant figure? 3 the 0 at the end the 0…
A: The correct answer is given below
Q: Identification Complete each statement. Write your answer at the end of each question. 92. To…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Calculate K, and pK, for water. H₂O acid (HA) + H₂O solvent H₂O+ + OH conjugate base (A)
A:
Q: Given that the pKa of carbonic acid (H2CO3) is 6.4, is sodium bicarbonate strong enough of a base to…
A: Sodium bicarbonate, NaHCO3(aq) is a conjugate base of carbonic acid, H2CO3(aq). For carbonic acid,…
Q: Describe what is mass spectrometer in your own words.
A: Spectroscopy is branch of chemistry in which we deal with electromagnetic spectra that results from…
Q: At a certain temperature the rate of this reaction is first order in HI with a rate constant of…
A:
Q: 98. It is the determination of the normality or molarity of a solution accomplished by the use of…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Multiple Choice Each of the numbered items or incomplete statements is followed by answers or by…
A: The correct answer is given below
Q: 25.00 mL of 0.041 32 M Hg₂(NO3)2 were titrated with 0.057 89 M KIO3. Hg2+ + 2103 → Hg₂(103)2(S)…
A:
Q: Hydrogen and fluorine react to form hydrogen fluoride, like this: H₂(g)+F₂(g) → → 2 HF(g) The…
A: Le Chatelier's principle: According to the Le Chatelier's principle when a change in concentration…
Q: 13.In sampling, a producer's risk (type 1 error) is usually occur when a bad batch was accepted 14.A…
A: Producer's Risk is the basically the probability of rejecting a compliant lot ,having a level of…
Q: Multiple Choice Each of the numbered items or incomplete statements is followed by answers or by…
A: #72: In titration, a particular volume of unknown concentration of an analyte is titrated with a…
Q: 56-57. What mass of Al(OH)3 would be produced if 350.0 mL of 0.4500 M KOH were added to a solution…
A: Here we are required to find the mass of aluminum hydroxide formed .
Q: Using the blank galvanic cell below, label the cell based on the following reaction: 3Cu(NO3)2(aq) +…
A: In a Galvanic cell, anode is the negative terminal where oxidation takes place and cathode is the…
Q: A dosage requires a patient to receive 59.2 mg of medicine for every 8 kg of body weight for every 4…
A: Given, A dosage requires a patient to receive 59.2 mg of medicine for every 8 kg of body weight for…
Q: What mass of sodium chloride is found in 0.2 L of a 1.45 M aqueous sodium chloride solution? The…
A: Given that - Volume of sodium chloride solution = 0.2 L We know that 1 L = 1000 mL , then - =>…
Q: he one lettered completion each case 69. The presence of a cotton fiber in a liquid preparation is…
A: In reference to the question 69. Defects are categorised on the basis of nature as- Ocular defect…
Q: حلة الاول كن نمي كول وبات الي الاستقرار مائل اوشش What if There Is No Carbonyl Group? The amino…
A:
Q: How many moles of HCl (molar mass = 36.46 g/mol) are present in 0.70 L of 0.5 M HCl?
A: Number of moles is calculated by dividing the given mass with molecular mass
Q: The control of the pesticides residue in fruits and vegetables has been emphasized by the standard…
A: Ans: Dispersive Liquid–Liquid Microextraction
Q: Write the concentration equilibrium constant expression for this reaction. 3+ 3 1₂(s)+2 Cr³+…
A: The expression of concentration equilibrium constant is given below
Q: Calculate the silver lon concentration in terms of pAg during the titration of 50.00 ml. of 0.05000…
A: Given in following question 50ml of 0.05000M Nacl with 0.1000M AgNO3 after the addition calculate…
Q: 101.Chemical processes in which a proton donor reacts with a proton acceptor 102. Distilled water…
A: Requirement from question: Three objective questions and answers.
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of…
A:
Q: What is the final product
A: LiAlH4 reduce the acid to form alcohol which was then attack with the carbonyl group form the…
Q: Multiple Choice Each of the numbered items or incomplete statements is followed by answers or by…
A: We have to tell which indicator is used in the assay of diluted phosphoric acid, material to be…
Q: 12. What is the IUPAC name of the following compound? ytheplane 3-isopropyl-6 2-methyl heptane G44
A: A species (molecule or ion) that can take a pair of electrons is referred to as a Lewis acid,…
Q: Describes how the Covid-19 virus can spread rapidly in an aircraft cabin using appropriate gas laws.
A: The principle states that at a given condition (i.e, fixed temperature and pressure), the rate of…
Q: A solution has a hydroxide-ion concentration of 0.043 M. What is its pH?
A:
Q: Q2) Determine if each of the reactions shown are oxidations, reductions or neither. OH OH NH ·ly
A: In organic chemistry oxidation is the process in which number of C and electronegative atom…
Q: Draw a diagram showing the changes in potential energy that occur during a full 360 degrees…
A:
Q: What is the molarity of a solution that contains 126 g NaCl (molar mass = 58.44 g/mol) in 0.2 L…
A: Molarity is a concentration term for a solution. The molarity of the given solution is defined as…
Q: Based on the molecular orbital model, choose all of the following molecules or ions that are stable…
A: Choose stable molecules or ions--
Q: A chemical bond for which one of the bonded atoms provides both electrons is referred to as a:…
A: Given that , The chemical bond is formed by the shareing of electrons but both the electrons are…
Q: Calculate the standard cell potential produced by a galvanic cell consisting of a nickel electrode…
A:
Q: Predict the elimination products for the reaction. Identify the major and minor stereoisomers and…
A:
Q: Solve for G, H and S by using/manipulating and solving for any of these variables in the equation…
A: Gibb's free energy is a state function and it is an extensive property. It is given mathematically…
Q: 16. A physician orders that a child should be given potassium chloride 1.6 milliequivalents per…
A: Given that - Prescribed amount of potassium chloride by physician = 1.6 milliequivalent per…
Q: Determine the mass of chlorine produced when a 200 A current flows for 24.0 h through a cell…
A:
Q: 101.Chemical processes in which a proton donor reacts with a proton acceptor
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What is the van't hoff factor of barium chloride?
A: The Van’t Hoff factor is used in the calculation of osmotic pressure, elevation of boiling point,…
Q: 4. The following data were obtained for the gas-phase decomposition of NO₂ at 300°C: NO2(g) NO(g) +…
A:
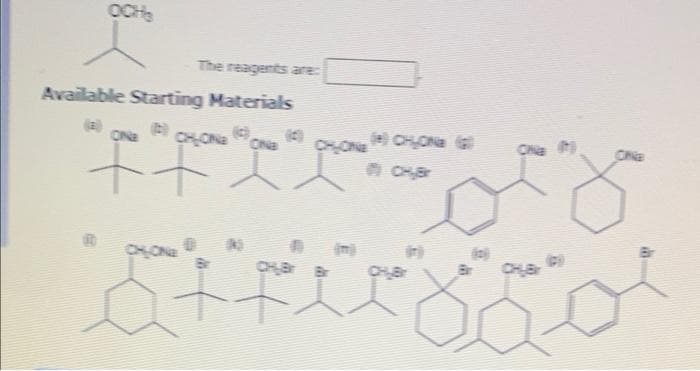
Step by step
Solved in 3 steps with 1 images

- Melamine, used as a fire retardant and a component of the writing surface of white boards, can be prepared from s-trichlorotriazine through a series of SNAr reactions with ammonia. The first substitution takes place rapidly at room temperature. The second substitution takes place near 100 °C, and the third substitution requires even higher temperature and pressure. Provide an explanation fatr this reactivity.Fill in the missing reagents a-h in the following scheme:2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3
- Which substituent is the strongest deactivator to EAS reactions? -NO2 -Cl -CH3 -OHM 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :1a) What’s the product in the following reaction? a. 4 b. 1 c. 2 d. 5 e. 3 1b) What reagents would you use to prepare product A with good yield? a. CH3CH2CH2CH2I; 2) Li, NH3 (liq), -33 °C b. KNH2; 2) CH3I; 3) H2, Pd/C c. HBr; 2) (CH3CH2CH2CH2)2CuLi d. NaNH2; 2) CH3CH2CH2CH2Br; 3) H2, Lindlar catalyst e. (CH3CH2CH2CH2)2CuLi; 2) H2, Lindlar catalyst
- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat(2)Draw the product(s) for each reaction. Provide stereochemistry where appropriate. Indicate if the reactionproceeds via S N 1 or S N 2Draw the products formed when CH3CH2C=C Na+ reacts with each compound. a. CH3CH2CH2Br b.(CH3)2CHCH2CH2Cl c.(CH3CH2)3CCl d.BrCH2CH2CH2CH2OH e. ethylene oxide followed by H2O f.propene oxide followed by H2O

