Octocrylene is an ingredient found in topical sunscreens. It is a water-resistant molecule that helps protect skin against harmful UVA and UVB radiation. (a) What is the hybridization of each nonhydrogen atom? (b) Circle all atoms bonded to the acyclic C=C double bond that are required to be in the same plane. (c) Are there two unique configurations possible about the acyclic C=C double bond? Explain. (d) Which of the two C-C single bonds indicated by arrows would you expect to be shorter? Explain. Octocrylene
Octocrylene is an ingredient found in topical sunscreens. It is a water-resistant molecule that helps protect skin against harmful UVA and UVB radiation. (a) What is the hybridization of each nonhydrogen atom? (b) Circle all atoms bonded to the acyclic C=C double bond that are required to be in the same plane. (c) Are there two unique configurations possible about the acyclic C=C double bond? Explain. (d) Which of the two C-C single bonds indicated by arrows would you expect to be shorter? Explain. Octocrylene
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.SE: Something Extra
Problem 24VC: A trisubstituted cyclohexane with three substituents-red, green, and blue-undergoes a ring-flip to...
Related questions
Question
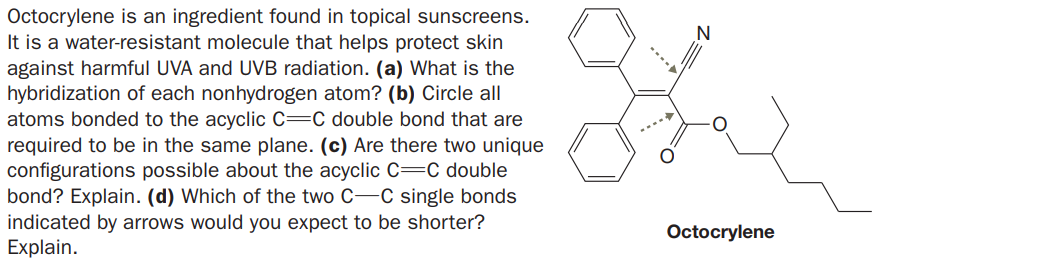
Transcribed Image Text:Octocrylene is an ingredient found in topical sunscreens.
It is a water-resistant molecule that helps protect skin
against harmful UVA and UVB radiation. (a) What is the
hybridization of each nonhydrogen atom? (b) Circle all
atoms bonded to the acyclic C=C double bond that are
required to be in the same plane. (c) Are there two unique
configurations possible about the acyclic C=C double
bond? Explain. (d) Which of the two C-C single bonds
indicated by arrows would you expect to be shorter?
Explain.
Octocrylene
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 4 images

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning