Q: A gas occupies 15.5 L at 0°C and 879 torr. What temperature would be required to increase the…
A: Given:Volume = 15.5LInitial temperature T1 = 0°CInitial pressure P1 = 879 torrFinal pressure P2 =…
Q: Chemistry
A: Step 1:In an electrochemical cell, oxidation takes place at the anode electrode while reduction…
Q: Question 14
A: The balanced equation for the reaction of zinc with hydrochloric acid is:Zn(s) + 2HCl(aq) →…
Q: Solve all complete solutions
A: Here are the IUPAC names and structures of the esters provided:Propyl propanoate:…
Q: Please give the major product of any of the given letter a, b, c or d. Explain the mechanism
A: 1) a)In the given Image, the overall reaction represents the Williamson ether synthesis, where an…
Q: starting with ethyl acetoacetate and formaldehyde as your only source of carbons synthesize the…
A:
Q: WS-3.3: Draw a detailed free energy diagram for the chlorination of benzene. Include and label the…
A:
Q: Question 16
A: Step 1:Limiting reagent is the reactant that exhaust first in the chemical reaction and decides the…
Q: 5. [8%] Give all IR group absorption frequencies expected for each of the following compounds.…
A: References; Field, L. D., Li, H. L., & Magill, A. M. (2020). Organic structures from spectra.…
Q: Question 19 Predict the expected product for each reaction and provide IUPAC name for the correct…
A: Epoxidation of starting alkene 2-methylbut-1-ene occur to form epoxide intermediate and in step-2…
Q: *Reaction is in image* Questions based on reaction in image: The oxidizing agent is: The reducing…
A:
Q: Write the common (not IUPAC) name and standard abbreviation for each molecule. O-P-O-CH20 H H NH2…
A:
Q: Complete the flow chart below to describe the separation of an acid and base mixture by providing…
A: Step 1: Step 2: Step 3: Step 4:
Q: Most common amines (RNH2) exhibit pKa values between 35 and 45. R represents the rest of the…
A: The presence of a cyano group (-CN) introduces significant electron withdrawal due to its strong…
Q: None
A: Let's analyze each change:An endothermic chemical reaction between two liquids results in gaseous…
Q: Use the following atomic weights and quantities to calculate the overall % yield of…
A:
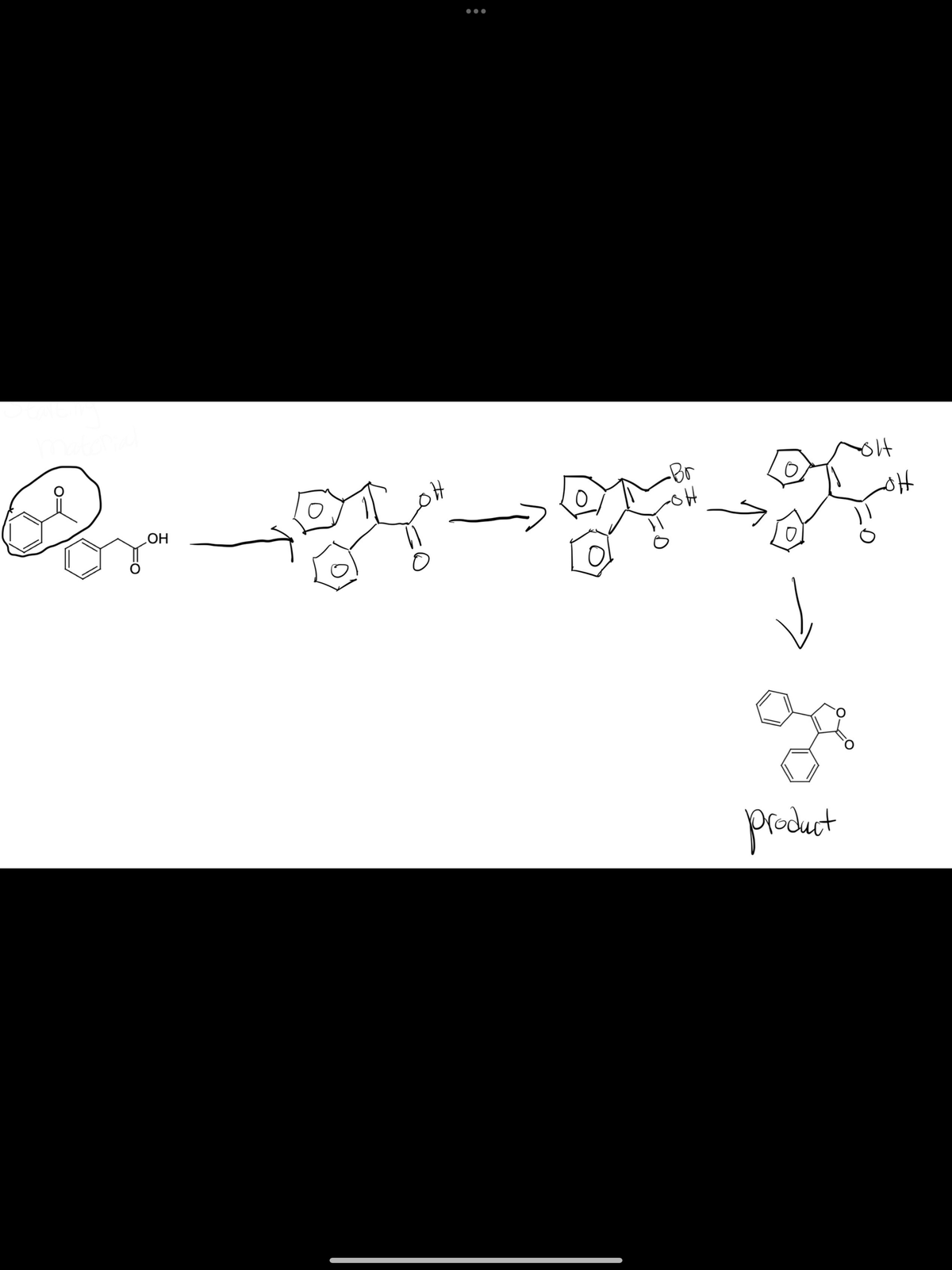
Q: Present a synthesis for the transformation depicted
A: Step 1: Step 2: Step 3: Step 4:
Q: Provide a reaction sequence
A: Step 1: Step 2: Step 3: Step 4:
Q: please can you explain the answer to these questions
A: Step 1: Sp^2 and 2Px :The sp² orbital and the 2px orbital can form both σ and π bonds. When…
Q: 5. Synthesis: Synthesize the following compound from cyclohexane, any carbon pieces that are three…
A: There are a couple of ways to synthesize a molecule containing a cyclohexane ring, smaller carbon…
Q: Draw a structural formula for the major organic product(s) of the reaction shown below. NH2 1. CH3l…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 18 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: Key references: Harder, S., & Langer, J. (2023). Opportunities with calcium Grignard reagents…
Q: Come up with a detailed mechanism for the following cyclization that also includes all resonance…
A: Step 1: The reason why the alkyl bromide portion of a molecule can remain intact in certain…
Q: Please don't provide handwritten solution ...
A: The molecular ion, often represented as M+ (M - plus), is a term used in mass spectrometry and…
Q: 4) i) Fill in the reagents to complete the synthetic sequence below, and draw the final product in…
A:
Q: Draw the structure(s) of the major organic product(s) of the following reaction. CI N₂ KI Draw one…
A: When compound reacts with potassium iodide (KI), a substitution reaction may occur, leading to the…
Q: Please write OUT the MECHANISM and briefly explain in WORDS the mechanism for the following…
A: Step 1:
Q: A 25.0 mL aliquot of 0.0580 M EDTA was added to a 47.0 mL solution containing an unknown…
A: Step 1: Here EDTA will make complex with v+3 and remaining EDTA will react with Ga+3 So the…
Q: Indicate the products X and Y that are obtained in the following reactions. Briefly comment on each…
A: References; Smith, M. B. (2020). March's advanced organic chemistry: reactions, mechanisms, and…
Q: Draw the structure of the major organic product of the reaction. CI (CH3)2₂CuLi ether, -78° C Marvin…
A: Gilman's reagent, typically composed of an organocopper compound such as lithium organocopper…
Q: Draw the structure(s) of the major organic product(s) of the following reaction after aqueous…
A: If you find this answer good please rate it helpful. If any query then raise it.Thank You!
Q: Which of the following gas samples would have the largest volume, if all samples are at the same…
A: The objective of the question is to determine which of the given gas samples would have the largest…
Q: Question 26 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: To predict the final product of a multi-step organic synthesis reaction, let's go through the…
Q: Question 11 Propose best Williamson ether syntheses for the following compounds. Select all…
A:
Q: Please do question 2 and classify as either aromatic or nonaromatic. Explain the mechanism/ process
A: Step 1: Huckel's ruleAromatic: (4n+2)π electrons in the system and the system should be fully…
Q: None
A:
Q: What are the uses of NMR in new drug discoveries?
A: Uses of NMR in New Drug DiscoveriesStructure Determination: NMR spectroscopy is used to determine…
Q: Identify the type of polymerization in each of the following:
A: Step 1:Answer a) This is an example of Condensation Polymerisation . Because in this two polymers of…
Q: Give the major organic product or missing starting material for the following reactions. Do…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. σ=[(Σd2)/(n-1)]½ n= number of trials d= measured value - mean…
A: There are two formulas in finding Standard Deviation, population standard deviation denoted by σ…
Q: Rewrote handwriting same clean it
A:
Q: Question 26 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: What are the uses of NMR in new drug discoveries? Please answer at your own easy words. Please don't…
A: Nuclear Magnetic Resonance (NMR) is a powerful technique used in drug discovery for several key…
Q: 5 Write the nuclear reaction for the beta decay of Th-234. Who is the daughter nuclide? 234 91 Pa…
A: The beta decay of Th-234 involves the emission of a beta particle (β⁻). Here's the complete nuclear…
Q: Incorrect Your answer is incorrect. Tag the carbons in this molecule that are bonded to the H atoms…
A: Thank you.
Q: 1. Assuming the validity of Raoult's law, do the following calculations for the benzene/toluene…
A:
Q: What is the concentration of A after 10.5 minutes for the reaction A → Products when the initial…
A: The objective of this question is to find the concentration of reactant A after a certain time…
Q: 1. A pure solvent freezes at 6.2oC. Determine the molar mass of a 0.8712 g nonelectrolyte dissolved…
A: Step 1: Step 2: Step 3: Step 4:
Q: The young expert Hand written solution is not allowed please
A: The given reaction is:- 2NO2(g) → 2NO + O2 This is the balanced reaction. Given S(entropy) of O2 =…
Q: The following shows 1.00L bulbs connected by valves. Each bulb contains neon gas with amounts…
A: To solve this problem, we'll use the ideal gas law, which states:PV=nRTWhere:- P is the pressure…
What is the mechanism and reagents for this reaction? with the circled molecule being attacked first since it more electronegative.
Step by step
Solved in 2 steps with 2 images

- Chemistry A stoppered flask in your laboratory drawer is stamped by the manufacturer with the notation “TC 25 250 mL”. Explain what this notation means.What volume of 95.0% alcohol by weight (density: 0.809g/cm^3) must be used to prepare150 cm^3 of 30.0% alcohol by weight (density: 0.957g/cm^3)On the occasion of World Health Day, Dr. Satpal organized a ‘health camp’ for the poor farmers living in a nearby village. After check-up, he was shocked to see that most of the farmers suffered from cancer due to regular exposure to pesticides and many were diabetic. They distributed free medicines to them. Dr. Satpal immediately reported the matter to the National Human Rights Commission (NHRC). On the suggestions of NHRC, the government decided to provide medical care, financial assistance, setting up of superspeciality hospitals for treatment and prevention of the deadly disease in the affected villages all over India. (i) Write the values shown by (a) Dr. Satpal (b) NHRC. (ii) What type of analgesics are chiefly used for the relief of pain of terminal cancer? (iii) Give an example of artificial sweetener that could have been recommended to diabetic patients.
- The average body temperature of a red-tailed hawk is 41.1C what is the temperature in K?The mass is 15.1 g. Please show workChemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.

