OH 1. Hg(OAc)2, H2O 2. NABH4 .CH3 CH3 H2C H3C Acid-catalyzed addition of water to an alkene yields an alcohol with Markovnikov regiochemistry. The electrophilic H* adds to the sp car hydrogens to yield the most stable carbocation intermediate, which then adds water to give the product alcohol. Because a carbocation inte rearrangements can occur prior to the addition of water. To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in a subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which cannot rearra the cyclic intermediate at the most substituted carbon to give an organomercury alcohol. The reduction step with sodium borohydride is co: radicals. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions + Aco-Hg-OAc AcO-Hg OAc
OH 1. Hg(OAc)2, H2O 2. NABH4 .CH3 CH3 H2C H3C Acid-catalyzed addition of water to an alkene yields an alcohol with Markovnikov regiochemistry. The electrophilic H* adds to the sp car hydrogens to yield the most stable carbocation intermediate, which then adds water to give the product alcohol. Because a carbocation inte rearrangements can occur prior to the addition of water. To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in a subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which cannot rearra the cyclic intermediate at the most substituted carbon to give an organomercury alcohol. The reduction step with sodium borohydride is co: radicals. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions + Aco-Hg-OAc AcO-Hg OAc
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter10: Alcohols
Section: Chapter Questions
Problem 10.52P: Alcohols are important for organic synthesis, especially in situations involving alkenes. The...
Related questions
Question
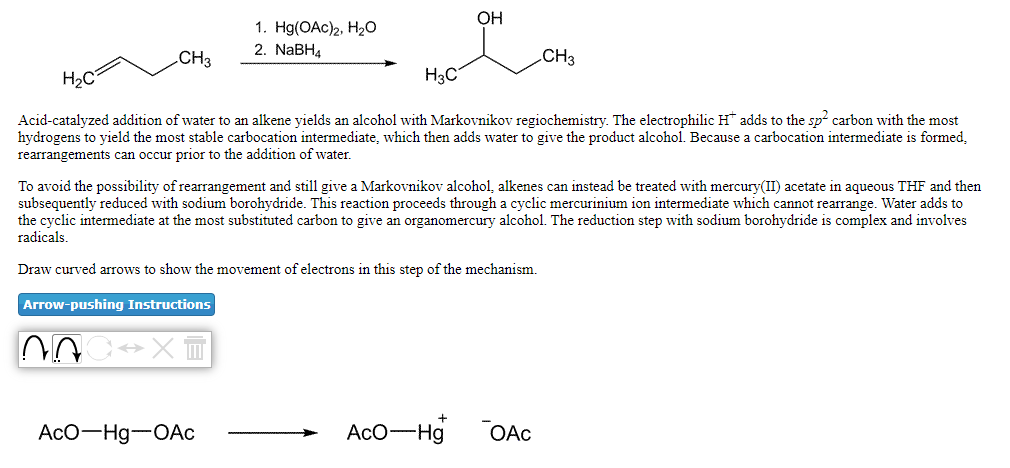
Transcribed Image Text:ОН
1. H(OAc)2, На0
2. NABH4
.CH3
CH3
H2C
H3C
Acid-catalyzed addition of water to an alkene yields an alcohol with Markovnikov regiochemistry. The electrophilic H adds to the sp carbon with the most
hydrogens to yield the most stable carbocation intermediate, which then adds water to give the product alcohol. Because a carbocation intermediate is formed,
rearrangements can occur prior to the addition of water.
To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in aqueous THF and then
subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which cannot rearrange. Water adds to
the cyclic intermediate at the most substituted carbon to give an organomercury alcohol. The reduction step with sodium borohydride is complex and involves
radicals.
Draw curved arrows to show the movement of electrons in this step of the mechanism.
Arrow-pushing Instructions
AcO-Hg-OAc
AcO-Hg
OAc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
