Q: use appropriate organic and inorganic compounds, show the synthesis of final product
A: The organic synthesis involves several steps for the conversion of one compound to the other and a f...
Q: What are the common names for (a) 1-propoxybutane and (b) 2-ethoxybutane?
A: The common name of 1-propoxybutane is butyl propyl ether.
Q: Order the following protons from lowest to highest chemical shift value. A) Hc < Hd < Ha <...
A: Answer isHc<Ha<Hd<Hb
Q: (a) How does the average kinetic energy of molecules comparewith the average energy of attraction be...
A: (a) Kinetic energy in the molecules possess due to their motion. The state of matter that has greate...
Q: Draw the structure of following compound. (2E,4E)-3-methylhexa-2,4-diene in the s-cis conformation
A: The representation of arrangement of atoms, group of atoms and bonds (double or triple) in the compo...
Q: P14A.10 Values of the molar polarization of gaseous water at 100kPa as determined from capacitance m...
A:
Q: A Primary alcohol will get converted into what functional group in the following reaction? 1. DMSO, ...
A: Primary alcohol on oxidation give aldehyde.
Q: d. A solution containing 1.00 mol of N2H4 (Kb = 8.9 x10-7) and 0.005 mol of HCl in 1 L of water is a...
A:
Q: Solve this question
A:
Q: Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uter...
A: Amide group is a type of functional group where an amine group is attached to a carbonyl group. The ...
Q: When 148.g of urea CH4N2O are dissolved in 1100.g of a certain mystery liquid X, the freezing point ...
A: When a non-volatile solute is added to a pure solvent, the freezing point of the resulting solution ...
Q: Oil reservoirs below ground frequently are in contact with underground water and, in connection with...
A: Solution
Q: order the following protons from lowest to highestchemical shift value.
A: Chemical Shift value of proton is directly proportional to deshielding of Hydrogen that Means if el...
Q: Define:(i)Elementary reaction in a process (ii) Rate of a reaction.
A: (i) Elementary reaction are those reactions in which two or more reactants forms the product only on...
Q: Can you please help me answer this questions? :<
A: Empirical formula is the formula that shows the elements in a compound in the lowest whole number ra...
Q: Can you please help me answer this question? :<
A:
Q: Question attached
A: (a) The expression used is : ∆G=∆H-T∆SGIVEN:∆H=-125 kJ/mol∆S=-126 J/mol/K ∆G =-125000(J/mol)-(310K)(...
Q: The normal boiling point of a certain liquid X is 131.10°C, but when 32.9g of glycine (C2H5NO2) are ...
A: Normal boiling point of liquid 'X'=131.10°C boiling point of solution (when glycine is dissolved in ...
Q: Find the product by writing down the mechanism of the reactions given below.
A: Alkyl halide gives solvolysis reaction water. The reaction completes in two steps. In the first step...
Q: The following conversion, carried out in the presence of Grubbs catalyst and ethylene gas, involves ...
A: The demonstrated molecules are marked as 4, 8, and 13 in the beginning material. It is an intramolec...
Q: (a) Write the complete β+ decay equation for 11C .(b) Calculate the energy released in the decay. Th...
A: Heavier nuclei are unstable and in order to attain stability,they emit radiation which is known as r...
Q: Find the potential energy for two methyl chloride molecules at 25°C seperated by 1.9°C
A: Given: Separation between two molecules r = 1.9 A° ...
Q: The osmotic pressure of a 0.010 M potassium iodide (KI) solution at 25°C is 0.465 atm. Calculate the...
A:
Q: Name and draw the condensed structure of the compounds formed by condensation reactions between benz...
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In...
Q: Covalent bondsa. link water molecules in ice.b. create only nonpolar molecules.c. bind sodium and ch...
A: Covalent bond: These are chemical bond formed between two atoms in which both atoms share equal pair...
Q: Order the following protons from lowest to highest chemical shift value.
A: The question is based on the concept of proton spectroscopy concept. Each proton has a characteristi...
Q: The pKa of the conjugate acid of guanidine is 13.6, making it one of thestrongest neutral organic ba...
A: Given: The given conjugate acid is guanidine. pKa=13.6
Q: A certain liquid X has a normal boiling point of 129.30°C and a boiling point elevation constant =Kb...
A: We are here given, normal boiling point of liquid X=129.3°C boiling point of solution (solution of X...
Q: What is the condensed electron configuration of iodine? If other, specify. A. [Ar] 5s2 4d10 5p5 ...
A: Electronic configuration is used to represent the distribution of electron density in an atom. In th...
Q: Determine the empirical formulas for the following compounds:(a) caffeine, C8H10N4O2(b) fructose, C1...
A: The empirical formula for the given compounds has to be given.
Q: The acid-catalyzed hydration of the internal alkyne shown here leads to only a single adduct (in low...
A:
Q: pls answer my question
A: The number of signals in proton NMR represents the number of non-equivalent protons in the compound.
Q: Identify solid as molecular, ionic, or atomic. Ar(s)
A: The solids which are composed of molecules that are held together by Van der Waals forces are called...
Q: Analyse the high resolution proton NMR spectrum and suggest a compound and write its name. Aslo writ...
A:
Q: An empty beaker weighs 42.77 g. When completely filled with water, the beaker and its contents have ...
A: Mass of water is equal to the difference in the mass of filled and empty beaker. Mass of the empty b...
Q: I need help with these two questions
A: 1.To obtain pure Cobalt from Cobalt(III) nitrate, the reduction of of Co takes place, and the eleme...
Q: Would you help me with these (4) Thank you.
A:
Q: Complete the reactions given below
A:
Q: Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-...
A: In balanced chemical equation, number of each and every atoms present in the equation is equal on ri...
Q: Draw the organic products formed in attached reactions?
A: Synthesis of phenyl(pyrrolidin-1-yl)methanone: When benzoyl chloride is treated with pyrrolidine in ...
Q: Find the product by writing down the mechanism of the reactions given below
A:
Q: Question attached
A: Given, The structure is; H2/Pd is a reducing agent. Palladium is an excellent catalyst.
Q: Explain Electron Spin and the Pauli Exclusion Principle?
A: A quantum property of electrons is known as electron spin. A form of angular moment is electron spin...
Q: Silicon has a band gap of 1.1 eV at room temperature.(a) What wavelength of light would a photon of ...
A: As your question consists of multiple subparts, we are solving only the first two, to then get the s...
Q: Q-2 Describe briefly various mechanism of breakdown in solids.
A: Dielectrics is a material which does not allow the flow of current. The spacing between the conducti...
Q: Label the protons in attached highlighted CH2 group as enantiotopic, diastereotopic, or homotopic.
A: Enantiomers are a pair of stereoisomers that are not superimposable on their mirror images. Enantiom...
Q: Balance each redox reaction occurring in acidic aqueous solution. b. Al(s) + Fe*(aq) Al*(aq) + Fe(s)
A: Redox reactions are the chemical reactions in which gain of electron(reduction) and loss of electron...
Q: 1. The concentration of sulfur dioxide is 8,800µg/scm. Convert this value to a) ppmv and bug/acm at ...
A:
Q: The following are the general characteristics of polar compunds, exceptA. Low melting point B. Low B...
A: Polar compounds are compounds in which there is an electronegative difference exists between two ato...
Q: 2. Hydrogen reacts with nitrogen to form ammonia according to the equation 3 H2(g) + Nag)2 NH(g) a) ...
A: a) The mass of NH3 produced from 3.55 moles N2 can be determined as, From the equation, it is clear ...
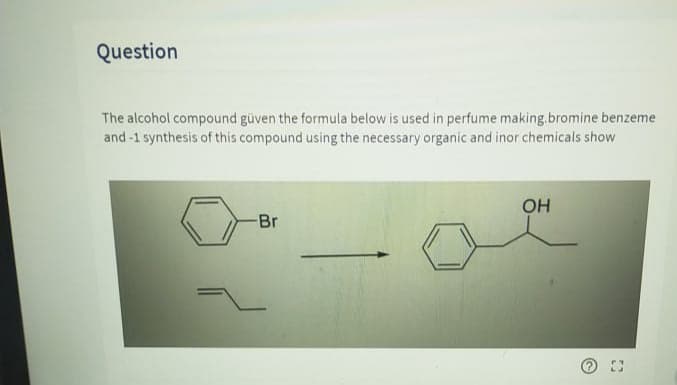
The alcohol compound güven the formula below iş used in perfume making. Bromine benzeme and -1 synthesis of this compound using the necessary organı and indir chemicals show
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 5 images

- Question 1 Carboxylic acids and its derivatives are reduced by sodium borohydride. True False Question 2 Less reactive derivatives cannot synthesize more reactive derivatives of RCOOH. True FalseOrganic Chemistry question; Please provide a well explained and correct answer for the following. the thiol has lower pKa compared with the alcohol in equal carbon number, which means the thiol has stronger acidity. Try to explain why -SH has such behavior in comparison to -OH.Which structure is best described as a carboxyl group? Question 5 options: a) –COOH b) –NH2 c) –OH d) None of the above
- 1. Give IUPAC names for the following compoundsWhat functional group(s) are needed to synthesis the following products (CHOOSE ALL THAT APPLY)Please include the letters with the answers. a. tertiary alcohol b. amide c. primary alcohol d. aldehyde e. secondary alcohol f. carboxylic acid g. aromatic h. ketoneQUESTION 3Starting with 2-pentyne, write the chemical equation for the synthesis of: a) Pentaneb) 2,2,3,3-tetrabromopentanec) 2,2-dibromopentaned) trans-2-pentenee) cis-2-pentene
- Give IUPAC names for the following compounds: b. c. e. f.Question 4: The reaction of propylbenzene with NBS gives: 1-bromo-3-phenylpropane 2-bromo-1-phenylpropane 1-bromo-1-phenylpropane A mixture of all three of the above compound The reaction of 1-phenyl-1-propene with HBr gives: Question 2 options: 1-bromo-3-phenylpropane 2-bromo-1-phenylpropane 1-bromo-1-phenylpropane A mixture of all three of the above compounds Question 17: The pKa of cyclopentadiene is much smaller than typical for an allylic hydrocarbon. The best explanation for this observation is: Question 17 options: Cyclopentadiene is much more stable than expected. Cyclopentadiene is much less stable than expected. The cyclopentadienyl anion is aromatic. The cyclopentadienyl anion is antiaromatic.OChem help with IUPAC names involving Ph and Bn The phenyl group (Ph-R, C6H5-R) can be formed by removing a hydrogen from benzene and attaching a substituent to where the hydrogen was removed. The benzyl group (abbv. Bn), similar to the phenyl group, is formed by manipulating the benzene ring. In the case of the benzyl group, it is formed by taking the phenyl group and adding a CH2 group to where the hydrogen was removed. Its molecular fragment can be written as C6H5CH2-R, PhCH2-R, or Bn-R. Please provide the IUPAC name for the following: (Ph)2CHC(CH3)2CC(CH2)3CH(Bn)CHO

