Q: Give a structure to match the name of each of the following compounds. - butanone -acetaldehyde
A: Ketones are functional groups whose name end with -one suffix and aldehydes are also functional grou...
Q: QUESTION 5 An unhealthy by-product of combustion is carbon monoxide which occurs in incomplete combu...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: The concentration of a drug additive in an animal feed is 12.5 ppm. How many milligrams of the drug ...
A:
Q: To a solution containing 1.0 × 10 ^(- 2) M AgNO3 and 2.0 × 10 ^(- 2) M Pb (NO3) 2 is added, with sti...
A: Given : Concentration of AgNO3 = 1.2 x 10-2 M So, concentration of Ag+ = 1.2 x 10-2 M Concentrati...
Q: Charle's Law A sample of Argon gas is cooled and its volume went from 380 ml to 250 ml. If its fina...
A:
Q: 3. For the reaction, N204(g) 2 nitrogen dioxide (g) At 25°C, the equilibrium concentrations of nitro...
A: Given Equilibrium concentrations [N2O4 ] = 0.0898 M [NO2] = 0.0204 M
Q: The average concentration of sodium ion and sulfate ion in sea water are 1.15ppm and 250ppm respecti...
A:
Q: Calculate the pH of a 0.31 M sodium formate solution (HCOONa). Kb for HCOO− = 5.9 × 10−11
A: Given :- [HCOONa] = 0.31 M Kb for HCOO- = 5.9 × 10−11 To calculate :- pH of solution
Q: What is the difference between coagulation and peptization.
A: Coagulation It is the process of converting a fresh precipitate into colloidal particles by shaking ...
Q: How many grams of phenol should be used to prepare 240 g of a 5% (w/w) solution in water?
A: Formula used :- W/W % = (Mass of phenol ÷ Mass of solution )×100%
Q: moles of ga
A:
Q: for hypochlorous acid, HClO, is 3.0×10-8. Calculate the pH after 40.0 mL of 0.100 M NaOH have been a...
A: Ka(HClO) = 3.0 × 10^-8 Volume of NaOH = 40.0 ml Molarity of NaOH = 0.100 M Volume of HClO = 40.0ml M...
Q: "Strike anywhere" matches contain P4S3, a compound that ignites when heated by friction. It reacts v...
A:
Q: If 24.5 mL of the 0.10 M NaOH solution is required to reach the endpoint in a titration with an unkn...
A:
Q: 3. Draw the splitting diagram for the indicated hydrogen and determine the expected splitting patter...
A: Well, in order to draw splitting pattern we need to look the neighbouring protons which are responsi...
Q: A scientist measures the standard enthalpy change for the following reaction to be -1142.2 kJ: 2H2S...
A:
Q: What is the pH of a 80.0 mL solution which is 0.25 M in IO¯ and 0.43 M in HIO? The Ka for HIO is 2.3...
A: HIO + H2O ⇔ H3O+ + IO- The concentration of conjugate base, [IO-] = 0.25 M the concentration of acid...
Q: 1. Observing pH Changes in Water and Buffer Solutions upon Addition of HCI and NAOH Solutions soluti...
A:
Q: A diprotic acid has a pKa1 = 3.10 and pka2 = 6.50. What is the pH of a 0.10 M solution of this acid ...
A:
Q: Enumerate and describe the types of precipitates. How will you control the size of precipitates
A: Interpretation- We have to enumerate and describe the types of precipitates and how we will control ...
Q: 1. Consider the equilibrium N2O4g) 2NO2G) AH°xn 58.0 kJ In which direction will the equilibrium shif...
A: According to Le-chatelier's principle, when any type of stress is applied at equilibrium then equili...
Q: 7. Identify the element Xwhich forms a polyatomic ion with the following Lewis Structure? [:CI-X-Cl:...
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simpl...
Q: 1. Locate the radial nodes in the 3s orbital of an H atom. Give them in Å.
A: There are two types of nodes, angular and radial nodes. Angular nodes are flat plane (at fixed angle...
Q: Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab, calculat...
A: Solution -
Q: Give the product for the following reaction. CH,CH - 2 CH,OH H* OCH3 CH,CH,- -H OCH3 CH,CH,- -H ÓCH;...
A: Aldehyde react with methanol to form acetal.
Q: Explain how a polyamide is able to bind to minor grooves and disrupt transcription.
A: Polyamide is a polymer in which a repeating unit is an amide group with a general chemical formula C...
Q: What exactly is SEO?
A: Answer What exactly is SEO its means Search engine optimization
Q: A chemist fills a reaction vessel with 0.839 g mercurous chioride (Hg, Cl,) solid, 0.302 M mercury (...
A:
Q: 14. 15. Ha PO 16. Br Br (BuONa (2 equiv) t-BUOH heat 17. HO 312 NO2 O2N
A:
Q: ermine the value of the equilibrium constant fro the reaction A + 2B = 20 L0 mol of A and 1.5 mol of...
A:
Q: mg of OXallo acid (H,C,04), a diprotic acid that can be purchased inexpensively in high purity, and ...
A:
Q: ΔG°
A:
Q: How many mL of resorcinol monoacetate should be used to prepare 1 pint of 15% (v/v) lotion?
A: Given, The volume of resorcinol monoacetate should be used to prepare 1 pint of 15% (v/v) lotion is...
Q: My question is " condensation of an aldehyde and DNP gives a coloured product. What structural featu...
A: Generally, compounds containing chromophore and auxochrome groups have a colour. Chromophore groups ...
Q: An aqueous solution contains NANO3 and KBr. The bromide ion is precipitated as AgBr by the addition ...
A: An aqueous solution contains NaNO3 and KBr. The bromide ion is precipitated as AgBr by addition of A...
Q: at rate
A:
Q: Calculate the HLB value for the surfactant shown below. Use equations specifically designed for non-...
A: Answer - The correct option is (e) = 14.1 Solution - According to the question - Given - In the give...
Q: We started with 20 mL 1 M acetic acid ~ 0.020 L acetic acid in a flask. (Ka acetic acid = 1.8x10^-5...
A: Millimoles of acetic acid taken=Molar concentration of acid×mL of acid=1 M×20 mL=20 mmol
Q: 9. The % w/w of I- in a 0.2144-g sample was determined by Volhard titration. After adding 50.00 mL o...
A: A kind of back titration method which is used for the identification and determination of concentrat...
Q: Compound A is an alcohol that undergoes oxidation to produce compound B. Compound B is a ketone that...
A:
Q: 1-methylcyclobut-1-ene with Cl, in dark conditions 2-methylprop-1-ene with O3, Zn, H;O, H*
A:
Q: Draw the major intermediate and product of the reaction between (2E)-4-methylhex-2-ene and Brz in th...
A:
Q: Substantiate the following: (i). Cyclopentadienyl anion is stable (ii) Cyclopropenyl cation is stabl...
A: Rules For Aromaticity Molecule must be cyclic. Every atom in the ring must be conjugated. Molecule ...
Q: For the reaction CO (g) + H2O (g) → CO2(g) + H2 (g), calculate the following reaction's thermodynami...
A:
Q: excess reactant
A:
Q: Classify each statement about electromagnetic radiation as true or false. True False Answer Bank X-r...
A: Electromagnetic radiations has both magnetic and electric fields and travels in waves .It can vary f...
Give EVERY step of the following reaction FULL names.
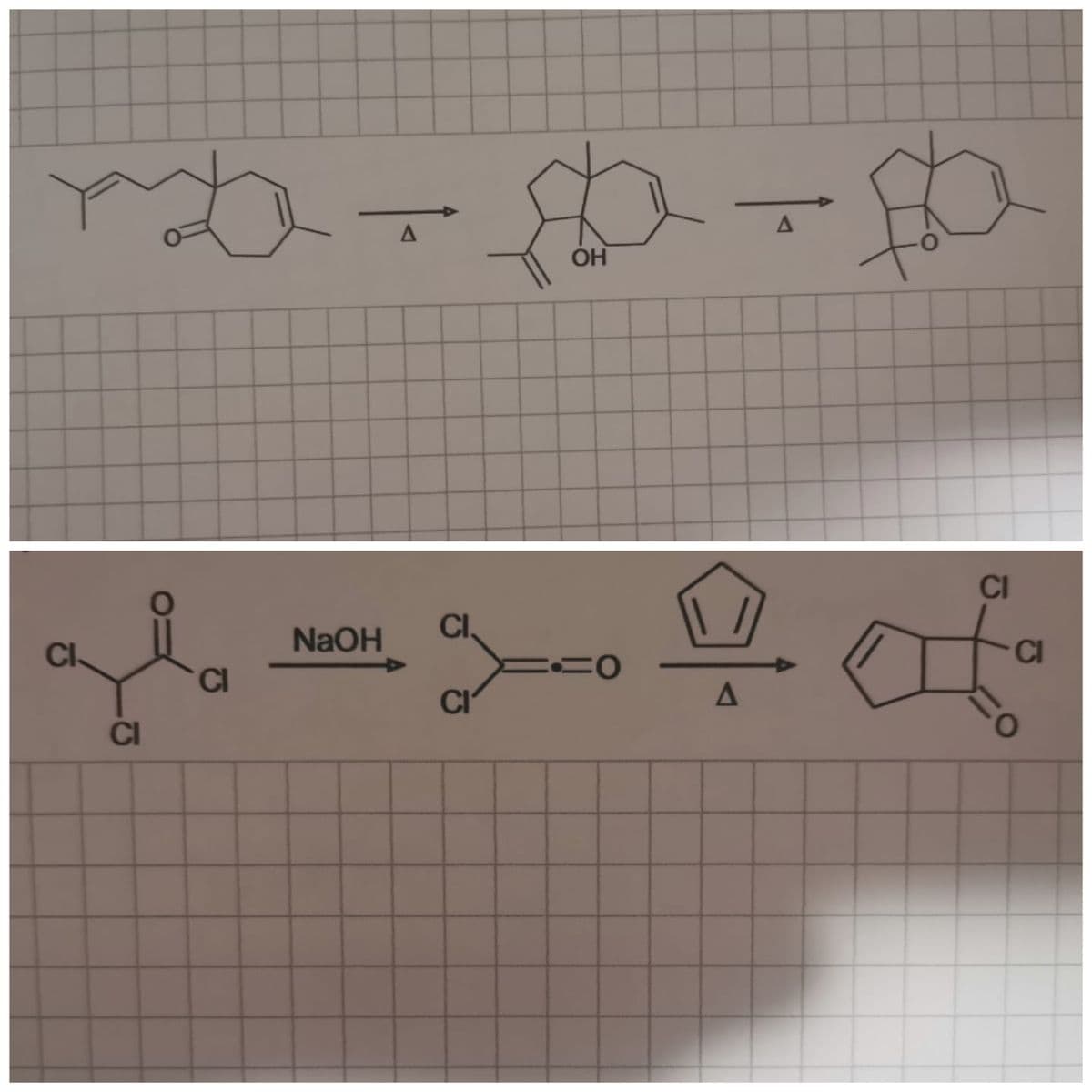
Step by step
Solved in 2 steps with 1 images


