OH NH2 Неpt + CH3SO,CI + CH3(CH2)NHET H3C (a) (b) (c) (d) (c) SOCI2 (a) + (b) H3PO4 OH Et Неpt (d) (i) (h) H3C Work out the details of the above retrosynthesis on a separate sheet of paper, and then draw the structure of (h). • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Include counterions for diazonium salts. opy aste Previous Next
OH NH2 Неpt + CH3SO,CI + CH3(CH2)NHET H3C (a) (b) (c) (d) (c) SOCI2 (a) + (b) H3PO4 OH Et Неpt (d) (i) (h) H3C Work out the details of the above retrosynthesis on a separate sheet of paper, and then draw the structure of (h). • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Include counterions for diazonium salts. opy aste Previous Next
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section: Chapter Questions
Problem 22.59P
Related questions
Question
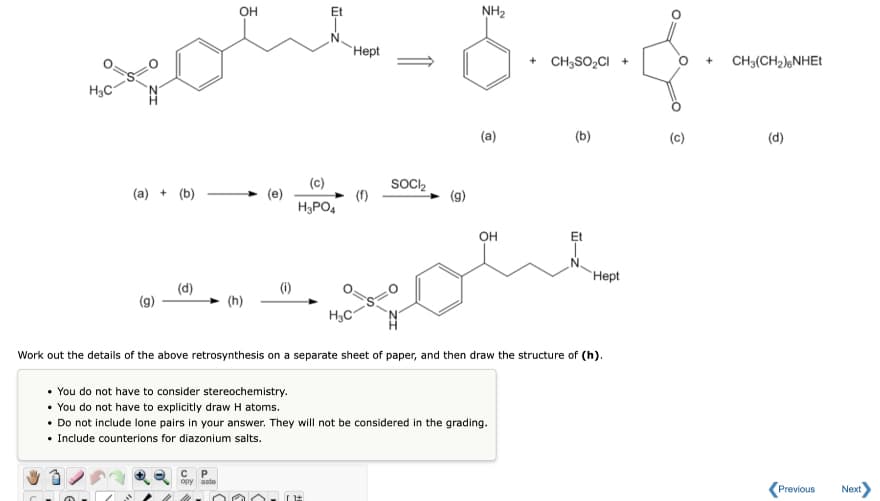
Transcribed Image Text:OH
NH2
Hept
+ CH3SO,CI +
CH3(CH2);NHET
(a)
(b)
(c)
(d)
(c)
SOCI2
(a) + (b)
H3PO4
OH
Et
Нept
(d)
(i)
(g)
(h)
H3C
Work out the details of the above retrosynthesis on a separate sheet of paper, and then draw the structure of (h).
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
• Do not include lone pairs in your answer. They will not be considered in the grading.
• Include counterions for diazonium salts.
opy aste
Previous
Next
-
Expert Solution
Step 1
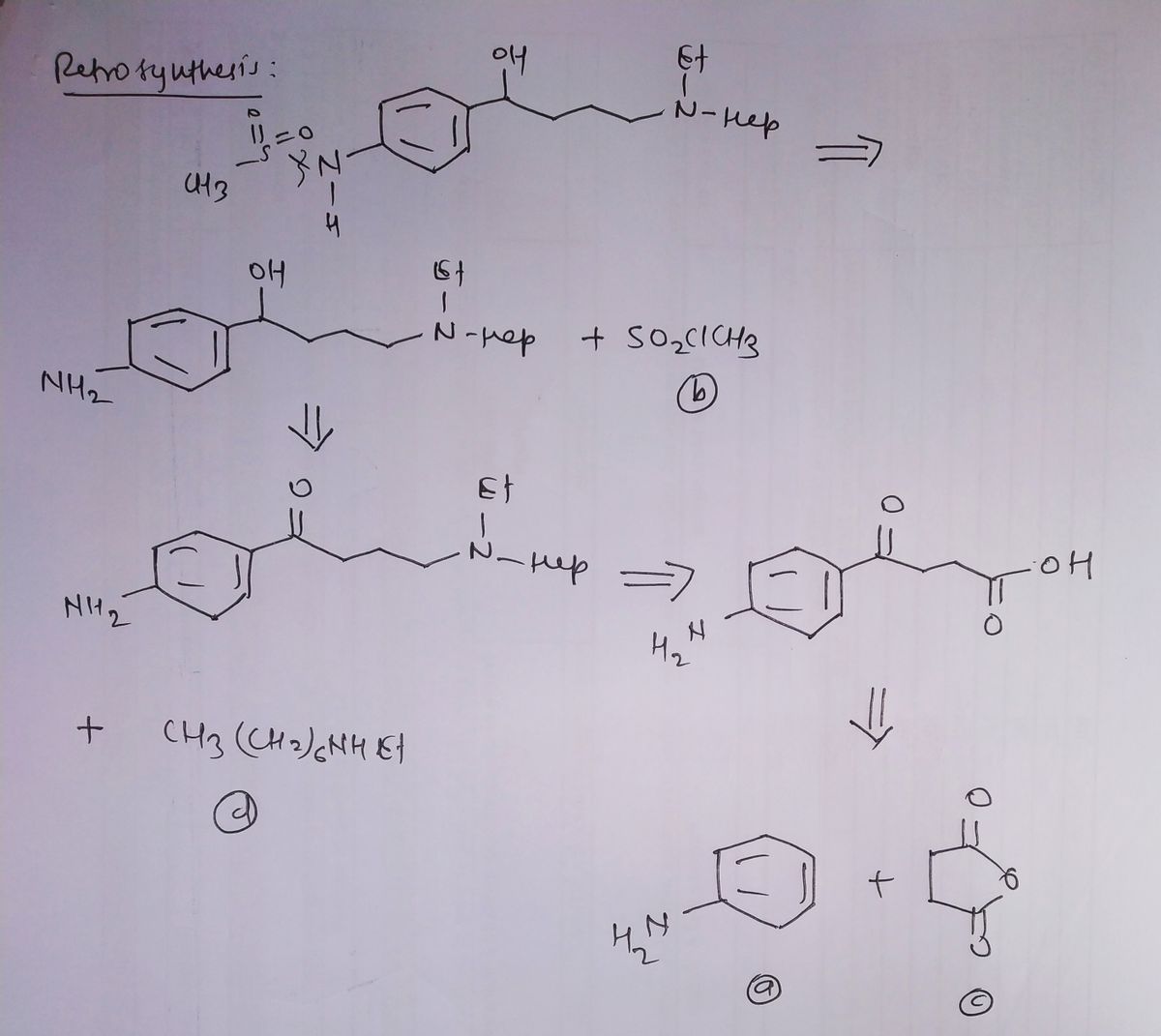
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning