on 13 of 14 > A student was given a solid containing a mixture of nitrate salts. The sample completely dissolved in water, and upon addition of dilute HC, no precipitate formed. The pH was lowered to about 1 and H,S was bubbled through the solution. No precipitate formed. The pH was adjusted to 8 and H,S was again bubbled in. This time, a precipitate formed. Which compounds might have been present in the unknown? Cr(NO,), Ni(NO,)2 Ca(NO3)2 O Cu(NO,), KNO, O Bi(NO,)3 V Zn(NO,), O AGNO, 6:27 1O 80°F Mostly sunny 7/30
on 13 of 14 > A student was given a solid containing a mixture of nitrate salts. The sample completely dissolved in water, and upon addition of dilute HC, no precipitate formed. The pH was lowered to about 1 and H,S was bubbled through the solution. No precipitate formed. The pH was adjusted to 8 and H,S was again bubbled in. This time, a precipitate formed. Which compounds might have been present in the unknown? Cr(NO,), Ni(NO,)2 Ca(NO3)2 O Cu(NO,), KNO, O Bi(NO,)3 V Zn(NO,), O AGNO, 6:27 1O 80°F Mostly sunny 7/30
Chapter8: Polyfunctional Acids And Bases
Section: Chapter Questions
Problem 3P
Related questions
Question
100%
Please see photo and check the correct compounds in the problem. Thank you.
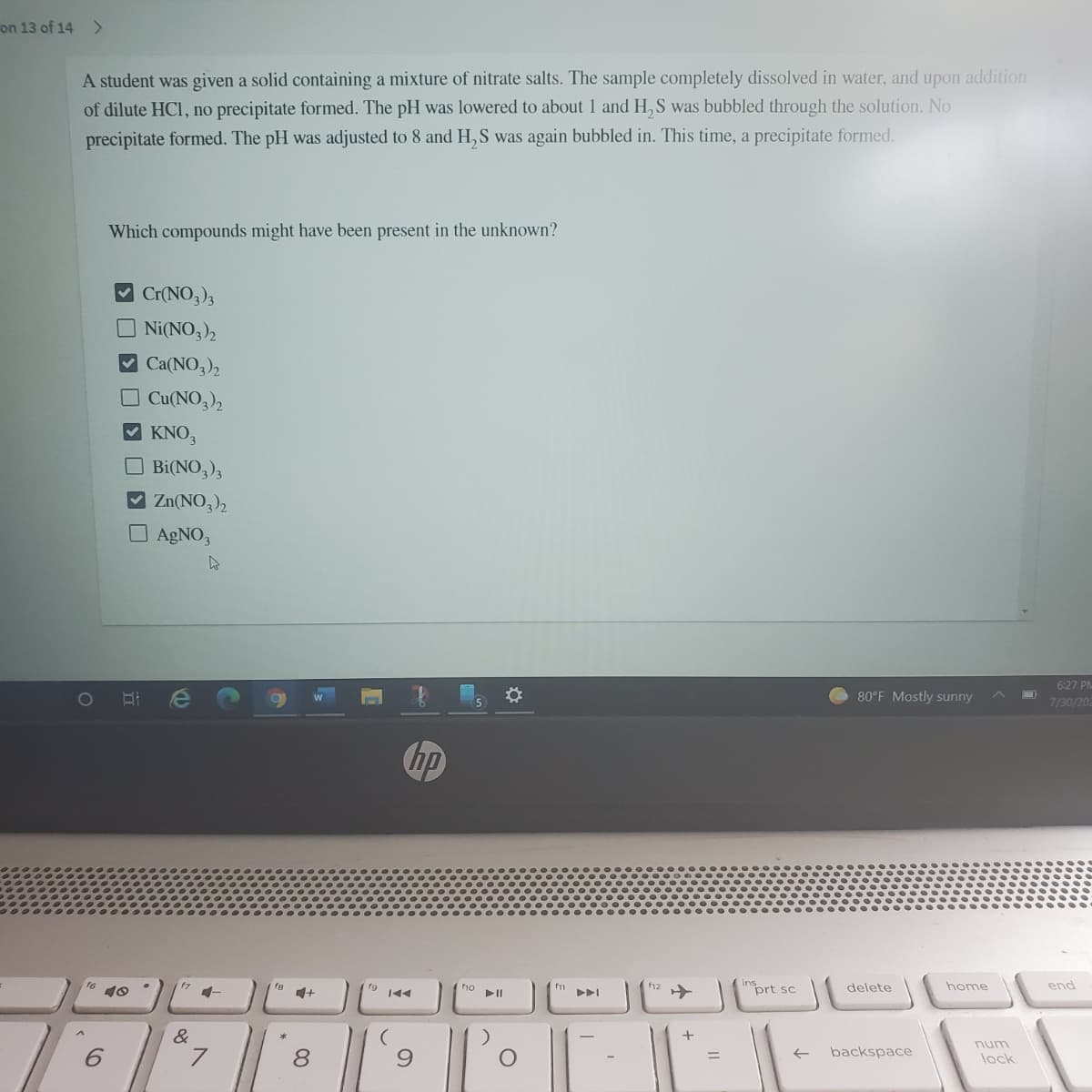
Transcribed Image Text:on 13 of 14 >
A student was given a solid containing a mixture of nitrate salts. The sample completely dissolved in water, and upon addition
of dilute HCl, no precipitate formed. The pH was lowered to about 1 and H,S was bubbled through the solution. No
precipitate formed. The pH was adjusted to 8 and H,S was again bubbled in. This time, a precipitate formed.
Which compounds might have been present in the unknown?
Cr(NO,)3
Ni(NO3)2
Ca(NO3)2
O Cu(NO,),
KNO,
O Bi(NO,),
V Zn(NO,),
O AGNO3
6:27 PM
1O
80°F Mostly sunny
7/30/202
hp
f9
Inprt sc
end
16
「ア
f10
f1
f12
delete
home
&
+
num
7.
8.
9.
backspace
%3D
lock
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

