Q: Correct each molecule in the drawing area below so that it has the structure it would have if it…
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: 7) Propose a synthesis. H D
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: If a solution of 0.10 M Mn²+ and 0.10 M Cd²+ is gradually made basic, what will the concentration of…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: © Macmillan Learning Classify each aqueous solution as acidic, basic, or neutral at 25 °C. Acidic…
A:
Q: Analyze the IR for the molecular formula C8H8O3 (vanillin). List only the relevant absorbancies (it…
A: Question is based on the concept of IR spectroscopy. we need to analyse the spectra.
Q: Draw the detailed mechanism and the product for the following reaction. Be sure to include…
A: Addition of Br2 to alkene produces trans-1,2-dibromoalkane.
Q: For the equilibrium system below, which of the following would result in an increase in the quantity…
A: Le’chatelier principle : According to this principle , when any change is applied on the reaction ,…
Q: The following skeletal oxidation-reduction reaction occurs under acidic conditions. Write the…
A: Question is based on redox reactions. we need to balance the given reactions under acidic…
Q: Question 9 Provide the systematic name of the compound shown below. CH3 07 OCH3 CH3 Ethyl…
A: The question is based on the concept of IUPAC naming. we need to write IUPAC name of given compound…
Q: A sample of gas has a mass of 1.110 g. Its volume is 117 mL at a temperature of 85 °C and a pressure…
A:
Q: (a) Calculate the value of Kc for the reaction: PCl5 (g) PCl3 (g) + Cl2 (g)…
A:
Q: nuc: 상 nuc: slower or faster
A: This is a type of nucleophilic addition reaction
Q: 1. On the basis of crystal structure, compute the theoretical density (cm³) of sodium chloride. Ana…
A:
Q: For the synthesis of veronalin, synthesize starting from diethyl malonate and using other necessary…
A: We have find out the synthesis of Veronalin.
Q: Calculate the value of Kc for the reaction: PCl5 (g) PCl3 (g) + Cl2 (g)…
A:
Q: pH = 8.03 Express your answer using two significant figures. [H3O+]= Submit Part D [H3O+]= VE ΑΣΦ pH…
A: The question is based on the concept of pH of the solution. it is defined as a negative logarithm…
Q: Identify the best product for the following reaction. CH₂CH₂ AICI; CH₂ CH₂ CH3
A: The given reaction is an example of lewis acid mediated acylation of benzene to form an acylated…
Q: Which one of the following complexes could be resolved into a pair of optical isomers? (ox = C2O42-)…
A: Optical isomers are two compounds which contain the same number and kinds of atoms, and bonds but…
Q: Which of the shown Fischer projections corresponds to open form of the pyranose chair shown? ОН I…
A: The aldohexoses and ketohexoses can undergo a change in their structure from linear to cyclic form…
Q: What is K for a reaction if AGº =86.0 kJ/mol at 25.0 °C? (R = 8.314 J/mol K) ●
A: ΔG° = 86.0 kJ/mol = 86.0×103 J/mol Temperature (T) = 25°C = (25+273) K = 298 K Gas constant (R) =…
Q: 5. A stock buffer solution is made by mixing 1 L of 0.5 M HF and 1 L of 0.5 M NaF. You take a 400 mL…
A:
Q: Consider the following reaction at 298K. Hg2+ (aq) + 2 Cr2+ (aq) Hg (1) + 2 Cr³+ (aq) Which of the…
A:
Q: Consider the following equilibrium: N₂O₂(g)-2NO₂(g) AG=5.4 kJ Now suppose a reaction vessel is…
A: Recall the given reaction, N2O4 g ↔ 2NO2 g…
Q: Propose an efficient synthesis for the following transformation The transformation above can be…
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: Calculate the cell potential for the galvanic cell in which the reaction Cd(s)+Cu2+(aq)↽−−⇀…
A:
Q: What is the angular momentum quantum number for the electrons with the highest energy in a zirconium…
A: The angular momentum quantum number for the electrons with the highest energy in zirconium atom…
Q: Which one is the product of the following reaction? Br CH₂Cl AICI 3 4 A.m-Bromochlorobenzene B.o-…
A: The given reaction is an example of Fridlecraft alkylation reaction and position of incoming…
Q: For each pair of substances pick the one that you expect to have the higher standard molar entropy…
A: According to thermodynamics, entropy is amount of disorder or randomness in a system. More the…
Q: What is the difference between the strength of an acid or base and its concentration
A: An acid is a species which donates Proton while a base accepts proton. an acid after donating…
Q: OH Br Br -ОН
A: This is a type of substitution reaction. We have find out the answer. Note -as per our company…
Q: rite the Ksp expression for the sparingly soluble compound calcium sulfate, CaSO4. either the…
A: CaSO4 is sparingly soluble salt and when dissolves in water gives Ca2+ and SO42- ion. Ksp also…
Q: Given the following chromatogram of opiates found in urine, determine the separation resolution…
A: Chromatography is an analytical technique which is used to determine the components of a mixture by…
Q: Consider the following system at equilibrium: C₂H₂(g) + Br₂(g) + 44 kJ C₂H₂Br₂(g) Which of the…
A: The question is based on the concept of Le-chatlier principle. It States that when a system at…
Q: A flask at room temperature contains equal numbers of di-nitrogen molecules and krypton atoms. (a)…
A: The question is based on the concept of gaseous state . we need to identify the gas having higher…
Q: A 275-mL flask contains pure helium at a pressure of 0.989 atm. A second flask with a volume of…
A: Given, First flask, Volume of Helium ( He) gas = 275 mL. Partial pressure = 0.989 atm. Second…
Q: A voltaic cell is based on the reaction between Ag+(aq) and Ni(s), producing Ag(s) and Ni2+(aq).…
A: Voltaic cell producing Ag(s) and Ni2+(aq) from the reaction of Ag+(aq) and Ni(s) Anode reaction :…
Q: major organic product? XO Et "OEt 0 A NaOH/H₂0 H30°, 4.
A: Esters undergo basic hydrolysis to give carboxylic acids. If a stable carboanion can be formed by…
Q: When solid aluminum hydroxide is in equilibrium with its ions, the ratio of aluminum ion to…
A: Given that aluminium hydroxide solution is in equilibrium with its ions . Formula for aluminium…
Q: A medical technician is working with the four samples of radionuclides listed in the table below.…
A: We have to arrange the given radionuclide according to initial radioactivity and calculate the time…
Q: Write the half-cell reaction for the following: a. Pt(s), Cl2 (g), HCl (aq), → HBr (aq), Br2 (l), Pt…
A: Left hand side is written the anode reaction and right hand side is written the cathode reaction.…
Q: Describe the overlap between orbitals in the cyanide ion (CN-).
A: CN- is made up of carbon and nitrogen and are bonded with triple bond . To determine hybridisation…
Q: First order rate constant(s) for the above is(are): I A) K₁ B) K₁+k₁ C) k₂ D) K₁ + K₂ E) K₁+k₂
A: The rate of an elementary chemical reaction is directly proportional to the products of the…
Q: Determine whether each of the following molecules is a hemiacetal, acetal, or neither and select the…
A: When same carbon has one -OH and one -OR group is called as hemiacetal.
Q: 0) What is the pH of a solution made by mixing 10.00 mL of 0.25 M acetic acid with 10.00 mL of 0.25…
A: Molarity of acetic acid solution= 0.25 M Volume of acetic acid solution= 10.00 mL Molarity of KOH…
Q: 1. The formula for the conjugate acid of ClO4- is . 2. The formula for the conjugate acid of…
A: Conjugate acid : According to the Bronsted- Lowery principle , conjugate acid contain one more H…
Q: What is supernatent? clear liquid above the precipitate. solution of liquid and precipitate colored…
A: The question is based on the concept of analytical chemistry. we need to define supernatant.…
Q: Subject; che
A: 1H-NMR is only used for the hydrogens. In the spectrum of 1H-NMR, we have to predict the different…
Q: In each of the following radioactive decay processes, supply the missing particle. 40 K→ 40 Ar +? a.…
A: It is based on the concept of radioactivity. Here we are required to find the missing particle in…
Q: Consider the reaction 2CO₂(g) +5H₂(g) → C₂H₂(g) + 4H₂O(g) for which AH = 46.50 kJ and AS = -124.8…
A: Answer: According to 2nd law of thermodynamics for every irreversible process or spontaneous entropy…
Q: The Δ?vap of a certain compound is 14.17 kJ·mol−1 and its Δ?vap is 93.89 J·mol−1·K−1. What is the…
A: The entropy change accompanying the change in phase from liquid phase to vapor phase is called the…
Practicing but don't understand how to draw
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Draw alcohol product. DRaw the different major organic producst of this reaction. IGnore inoragnic byproducts. Please answer fast i give you upvote.What are the starting materials you need for reaction 1 for the included molecule? Find a commercial source and report the names and CAS-numbers of the compounds. I also included the retrosynthesis for the molecule and reaction 1 so the answer is based off those.please draw out the complete mechanism for this reaction. I upvote responses
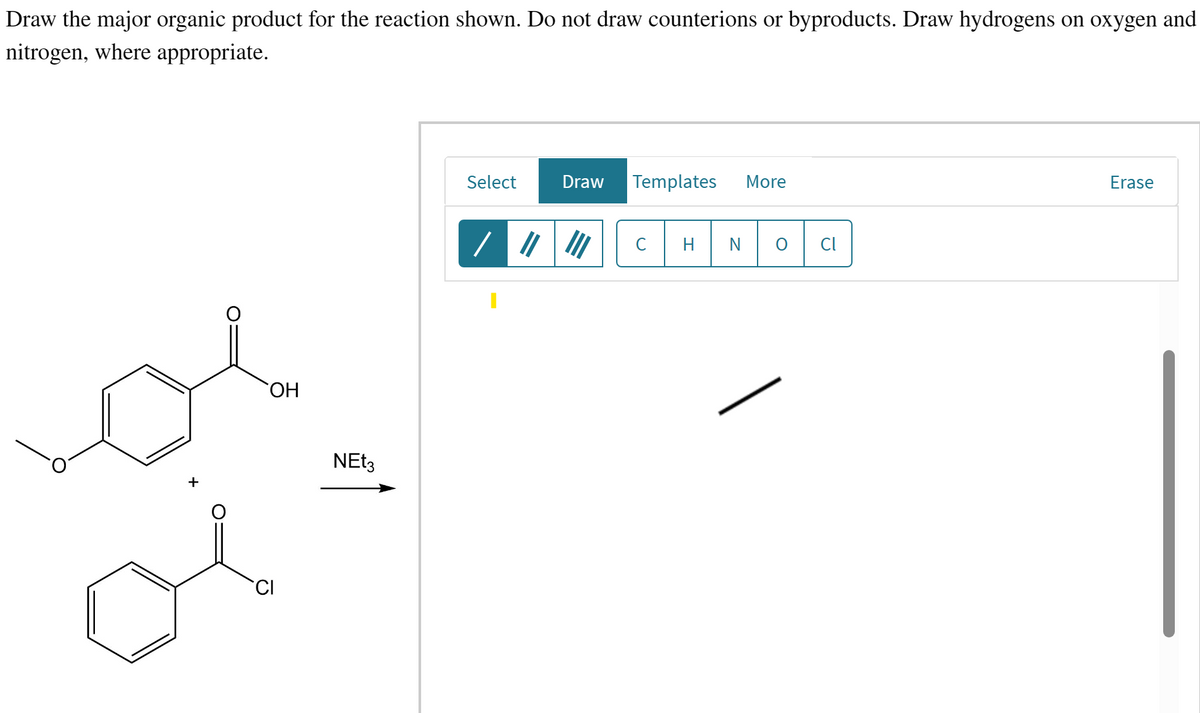
- Draw the major organic product for the reaction shown. Do not draw counterions or byproducts. Draw hydrogens on oxygen or nitrogen, where appropriate.Shown below is a retrosynthesis reaction. Identify the missing reagents. What is the forward reaction or synthesis of the reaction? Identify the reagents for the forward reaction.Show the detailed reaction mechanism symmetrical aldol condensation of propanal to 2-methyl-2-pentenal for meprobamate. (show movement of electrons, arrows, reagents, conditions, main product and biproducts etc.)
- Propuse a mecanism for the following reactionDraw the major organic product or products for this reaction. Be sure to include stereochemistry where appropriate. Assume it takes place in the most appropriate solvent. Br: Please answer fast I give you upvote.Draw the missing products and/or reagents in the following multistep synthesis. Ignore any inorganic byproducts.

