Q: H3CO OH 2 HCONH,
A: The reaction of ketone with formamide results in the formation of an imine. An aldehyde and ammonium…
Q: Draw a structural formula for the major organic product(s) of the reaction shown below. NH2 1. CH3…
A: Whenever there is an excess alkylation of amines then the products are quaternary ammonium salts.
Q: To 0.340 L of 0.150 M NH3 is added 0.150 L of 0.120 M MgCl₂ ▾ Part A How many grams of (NH4)2SO4…
A: Firstly, calculate the moles of every compound used.n(NH3)= (0.150 mol/L )(0 .340 L) = 0.051 moles…
Q: Draw the octahedral crystal field d orbital splitting diagrams for [Fe(OH2)6] 2+ and [Fe(CN)6]3-.…
A: Coordination compounds are formed by combining the transition metals with ligands. The degenerate…
Q: Please don't provide handwritten solution ....
A: The objective of this question is to calculate the pH of a buffer solution before and after the…
Q: Name each of the acids listed in the table below. chemical formula HCIO name HI ☐ H₂SO3 ☐ HCIO 4 ☐
A: For the binary acids, the name starts with the hydro. Then write the name of the anion with the…
Q: 5. Predict the product(s) of the following E2 reactions. DBN
A: This is an example of Hoffmann elimination reaction. In this reaction less substituted alkene is…
Q: Aromatic Substitution Reactions 1. Predict the major product(s) of the following reactions: CI…
A: Note: “Since you have posted a question with multiple sub parts, we will provide the solution only…
Q: When 85.0g of CH4 are mixed with 360. G of O2 the limiting reactany is
A: We need to determine limiting reagent of the reaction.
Q: 1. Determine the product(s) and propose a mechanism for the following reaction. OH MeOH H+ lottex (1
A: The objective of this question is to determine the product(s) and propose a mechanism for the given…
Q: (i) COOH (h) (1) 2 CH3CH₂Li (2) H3O+ H H3O+ ↑ C=PPh3 + 요. CH3 NILL H+, heat
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Consider a 0.586 M aqueous solution of barium hydroxide, Ba(OH)2Ba(OH)2 (aq). How many grams of…
A: The objective of the question is to calculate the amount of Ba(OH)2 in grams, the number of…
Q: Briefly explain the principle and application of potentiometry or pH meter or lon selective…
A: The method of measuring the voltage in a solution between two electrodes is called potentiometry.…
Q: Show how you would accomplish the following synthetic conversions. (a) 1-butene 1-bromobutane (b)…
A: The reactant can be converted into products by selecting the suitable reagents and reaction…
Q: . Part A Equilibrium and an Acid-Base Indicator - Equilibrium system: HA (aq) H* (aq) + A¯ (aq)…
A: Step 1 - Color of bromothymol blue in distilled water - GreenAnalysis:The acidic form of the…
Q: Help with question #17
A: The major product of the reaction is 3-methyleneheptane:Explanation:The given reaction is also known…
Q: single bonds. of the starting material to draw the major product(s). Use the single bond tool to…
A: The objective of the question is to find the structure of the major product for the given reaction…
Q: You have 50.0mL of 0.520M NH3. Kb = 1.8x10-5 You add 15.0mL of a 0.05M HCl solution. What is the pH…
A: The objective of this question is to calculate the pH of a solution after the addition of a certain…
Q: You have 50.0mL of 0.135M HCl. You add 10.0mL of 0.25 M NaOH What is the pH of the solution now?
A: The objective of this question is to calculate the pH of a solution after the addition of a base…
Q: Perform the following calculations, reporting the answers with the correct number of significant…
A: The question demands doing a mathematical calculation and stating the result in exponential format…
Q: Find the pH of 0.047 M trimethylammomium chloride solution given a pKa of 9.799 for the…
A: The objective of this question is to calculate the pH of a 0.047 M trimethylammonium chloride…
Q: Determine the mass, in grams, of 0.785 moles of Si (1 mol of Si has a mass of 28.09 g)
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: The structure below has an unusually large dipole. A skeletal structure with a cation is provided…
A: The solution is given belowExplanation:Aromaticity- The compounds are called aromatic if below…
Q: Draw the organic product(s) f 9.43 each reagent. a. H2SO4 b. NaH c. HCI + ZnCl2 d. HBr e. SOCI 2,…
A: The objective of this question is to predict the organic products formed when propanol (CH3CH2CH2OH)…
Q: PCI3 atomic metallic solid ionic solid molecular solid atomic nonmetallic solid
A: The objective of the question is to classify the type of solid that PCl3 (Phosphorus trichloride)…
Q: help ?
A: The reaction has drawn with proper arrows. Please find the attachment herewith.Explanation:
Q: For the following reaction, 6.70 grams of diphosphorus pentoxide are mixed with excess water. The…
A: Here the reaction is between diphosphorus pentoxide and water and the product is phosphoric acid.
Q: Do the reactions below proceed in good yield from left to right as shown? b) excess OH c) OH H₂O да…
A: The objective of the question is to predict the reactions that give good yield from the given…
Q: Please don't provide handwriting solutions....
A: The objective of this question is to determine whether the given reaction is exothermic or…
Q: Draw products and the mechanisms for each of the following:
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Show all possible electronic configurations for C (1s2 2s2 2p²) atom using term and symbol.
A: The objective of this question is to predict the electronic configurations, as in the case of…
Q: < Solid Ca3(PO4)2 is placed into 10.0 L of water. When equilibrium is established, the concentration…
A: Equilibrium constant Kc for a reaction is defined as the product of the molar concentrations of the…
Q: g) Ammonium hydroxide + nitric acid → Molecular Total lonic Net lonic
A: Here, ammonium hydroxide is a base and nitric acid is an acid. Now, when an acid reacts with a base,…
Q: We have a weak aid with a Kg of 3.7. 10. The pH was found to be 3.66. What was the approximate…
A: equilibrium constant (Ka) = 3.7 10-7pH = 3.66 pH = -log[H+]
Q: You have an aliquot of 10 mL of 0.1022 mol/L phosphoric acid. The ka values for phosphoric acid are…
A: The objective of this question is to calculate the resultant pH of a solution after mixing…
Q: A O
A: The given molecule is a bridged bicyclic compound. The two rings are attached through a bridge.
Q: Please name the following compounds. Make sure you spell each name correctly, leave no spaces since…
A: 1) Cyclodeca-1,6-diyne2) (Z)-Cyclodec-1-en-6-yne3) (1Z,6Z)-Cyclodeca-1,6-diene4)…
Q: Your answer is incorrect A chemistry student weighs out 0.414 g of citric acid (H,CH,O,), a…
A:
Q: 4. For each of the following, determine if either a (a) hydride shift or (b) methyl shift has…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Help with question #13
A: 1-Phenylbutene (Z)Explanation:It is wittig reaction. In this reaction benzaldehyde reacts with ylide…
Q: What is the major product of the following reaction? О 1 eq. CH3CH2MgBr H3O+ OCH3 (CH3CH2)20 Step 2…
A: The objective of this question is to find the product formed from the given reaction.
Q: 2) Identify the reagents you would use in order to synthesize the compound below via an imine…
A: Answer shown in attachment provide in explanation box Explanation:
Q: You have 50.0mL of 0.116M HCl. You add 10.0mL of 0.25 M NaOH What is the pH of the solution now?
A: The objective of this question is to calculate the pH of a solution after the addition of a base…
Q: Could we have used other materials instead of copper and zinc plates ? If so, list 2 other materials…
A: The question is asking if there are alternative materials that could be used in place of copper and…
Q: A chemistry graduate student is given 250. mL of a 0.70M hydrocyanic acid (HCN) solution.…
A:
Q: You have an aliquot of 10 mL of 0.1060 mol/L phosphoric acid. The ka values for phosphoric acid are…
A: The objective of this question is to calculate the resultant pH of a solution after mixing…
Q: Gaseous ethane (CH3CH3) will react with gaseous oxygen (O2) to produce gaseous carbon dioxide (CO2)…
A: Given:Mass of ethane (CH3CH3) = 16 gMass of O2 = 117 gMolar mass of ethane (CH3CH3) = 30.07…
Q: Provide mechanism with arrows and charges H3CO NaOH H₂O مسم
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Arrange these substances in order of increasing boiling point: H2 H₂O LICI H₂S OH₂<H₂O<LiCl < H₂S…
A: The objective of the question is to arrange the given substances H2, H2O, LiCl, H2S in order of…
Q: -H CI-CI hv CI
A: The mechanism of the reaction takes place via radical formation and involves three steps:Step 1:…
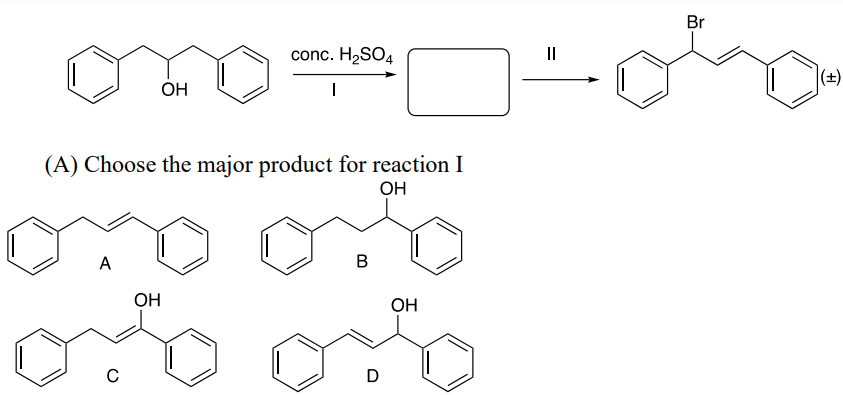
B. Choose the major product for reaction I
C. Choose the correct reagent for reaction II
A. HBr
B. Br2/hv
C. NBS/hv
D. HBr/ROOR
E. B or C
Step by step
Solved in 1 steps with 1 images

- Calculate △G° for the equilibrium reaction below. Given: CH3NH2(aq)+H2O(l)-->CH3NH3+(aq)+-OH(aq) Kb @25°C=4.4x10-4In cells, vitamin C exists largely as its conjugate base X. X is anantioxidant because radicals formed in oxidation processes abstract thelabeled H atom, forming a new radical that halts oxidation. Draw thestructure of the radical formed by H abstraction, and explain why this Hatom is most easily removed.Please help with this calculation of this, don't know to get the n(product) Page2. please help with it too Thanks in advance

