Chapter3: Mechanisms
Section: Chapter Questions
Problem 135EQ
Related questions
Question
100%
Match the reagents to the reaction (place letter on reaction below)
Only 1 letter can apply to the reaction.
![A. LIAIH [R]
B. 1) NaBH [R]
2) H₂O*
C.
CH₂CH₂MgBr
D. 1) BH₂/THF
2) H₂O₂OH
SOCI₂
E.
F. H₂O*
G.
H.
Na₂Cr₂O, [0]
1) Peracetic Acid
2) H30*](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F76ee89b8-722d-4c27-b7b7-9a80c7775531%2Fc08bc877-91d0-49c1-95d9-23190f57f694%2F4zokrn_processed.jpeg&w=3840&q=75)
Transcribed Image Text:A. LIAIH [R]
B. 1) NaBH [R]
2) H₂O*
C.
CH₂CH₂MgBr
D. 1) BH₂/THF
2) H₂O₂OH
SOCI₂
E.
F. H₂O*
G.
H.
Na₂Cr₂O, [0]
1) Peracetic Acid
2) H30*
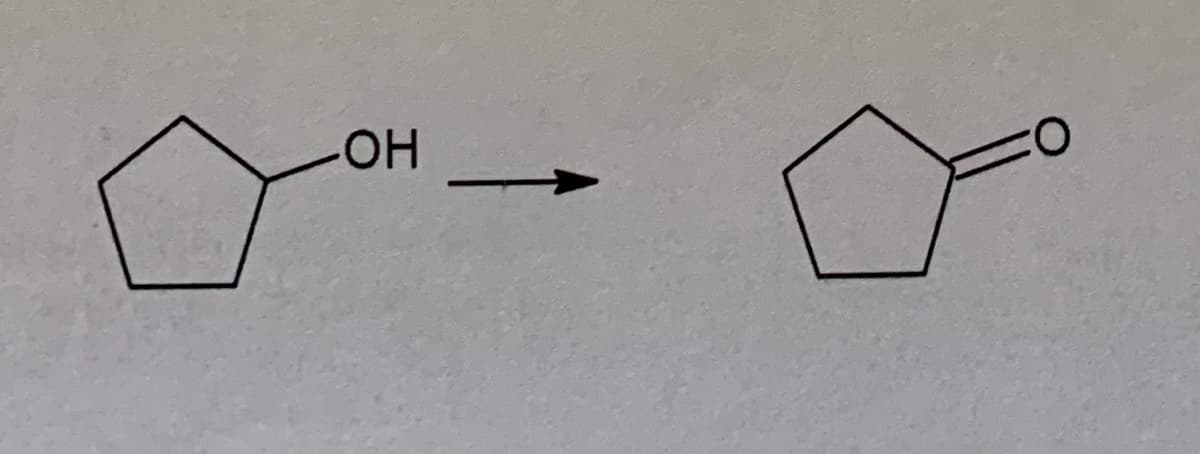
Transcribed Image Text:0:
НО
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

