он OEt cinnamaldehyde cinnamic acid ethyl cinnamate 1. Dissolve in ether 2. Add aq. NaOH aqueous organic Add HCI organic aqueous solid compound(s)
он OEt cinnamaldehyde cinnamic acid ethyl cinnamate 1. Dissolve in ether 2. Add aq. NaOH aqueous organic Add HCI organic aqueous solid compound(s)
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section: Chapter Questions
Problem 22.51P
Related questions
Question
5
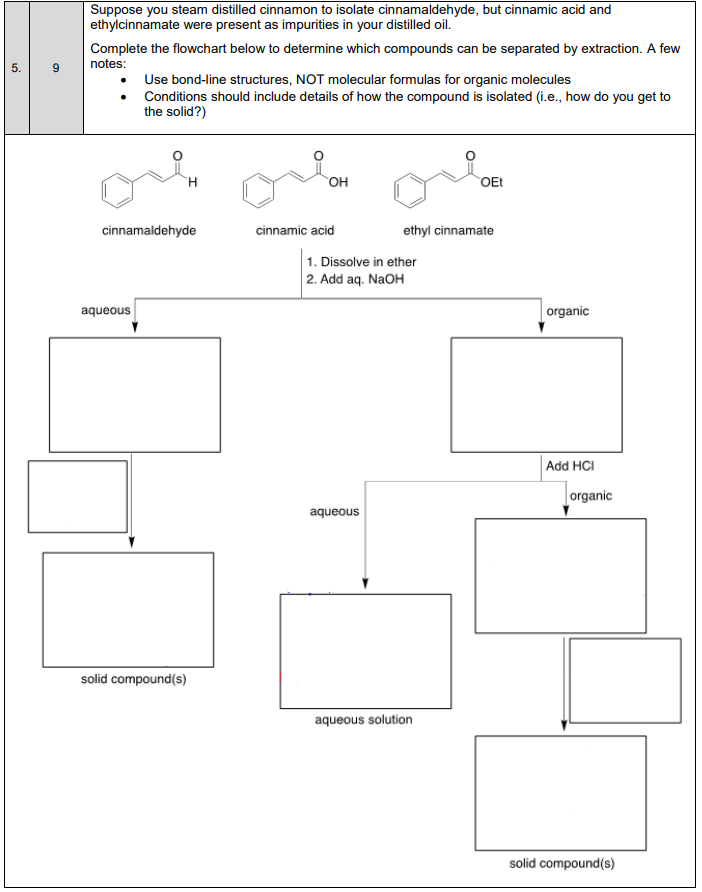
Transcribed Image Text:Suppose you steam distilled cinnamon to isolate cinnamaldehyde, but cinnamic acid and
ethylcinnamate were present as impurities in your distilled oil.
Complete the flowchart below to determine which compounds can be separated by extraction. A few
notes:
Use bond-line structures, NOT molecular formulas for organic molecules
Conditions should include details of how the compound is isolated (i.e., how do you get to
the solid?)
OH
OEt
cinnamaldehyde
cinnamic acid
ethyl cinnamate
1. Dissolve in ether
2. Add aq. NaOH
aqueous
organic
Add HCI
organic
aqueous
solid compound(s)
aqueous solution
solid compound(s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning