Q: A sample of metal has a mass of 17.85 g, and a volume of 7.99 mL. What is the density of this metal?...
A:
Q: Calculate the molecular weight of sulfur if 35.5 grams of sulfur dissolve in 100.0 grams of CS2 to p...
A: Given: Mass of sulfur=35.5g Mass of CS2 = 100g Boiling point of the solution is 49.48°C To find: The...
Q: During a certain reversible process, the surroundings undergo an entropy change, ∆Ssurr = -78 J>K...
A: As per the thermodynamic concept, the entropy of universe is the sum of entropy of system and entrop...
Q: Which one of the following statements about free radicals is not true? A. They are formed ...
A: Free radicals are very reactive species. Free radicals can be generated in many ways like chemical r...
Q: An object of mass 100 g suspended from the end of a rubber band has a vibrational frequency of 2.0 H...
A:
Q: Draw the molecular structure for an ether that is an isomer of 1-propanol.
A: 1-propanol is an alcohol consisting of three carbon atoms and ether is the compound containing -O-fu...
Q: A photochemical [3,5] sigmatropic rearrangement will occur through Select one: O a. Conrotatory Orbi...
A: Interpretation - To tell about that a photochemical [ 3,5 ] sigma tropic rearrangement will occur th...
Q: A certain reaction has the rate law: Rate = k [A]. The half-life of this reaction is 20 minutes. a) ...
A: It is given that the order of the reaction is the first-order reaction, as in the given expression o...
Q: Evaluate: 27.30 g + 12.755 g + 0.055 g = %3D Report answer to the correct number of decimal places a...
A: When addition operation is performed, the final answer have least number of digits after the decimal...
Q: The equilibrium constant K, for the reaction H2(g) + Br,(g) - 2HBr(g) is 2.18 x 10 at 730°C. Startin...
A:
Q: Draw the open structures of the molecules in the table below using the Lewis dot structure or VSEPR ...
A: First of all, we will discuss the molecules that are given in the question. The molecule OCS has a l...
Q: Complete the reactions given below
A: Addition of HBr to alkene is Electrophilic addition reaction ,Which follows Markovnikov's rule. Ma...
Q: Chemistry Question
A: Given, length in meters 1.81 x10-3 m To convert it into nanometres = ?
Q: h,i, can you solve this ? The benzene standard evaporation entropy is 96.4 J / K.mol and the standar...
A: Given: ∆Hvap = 33.9 kJ/mol = 33900 J/mol ∆Svap = 96.4 J/K.mol
Q: What is the first-order scintillation angle of the shimmering x-rays of a crystal surface with a dis...
A: Given that: Order of Scintillation (n)= 1 Distance between the two planes (d) = 0.204nm Wavelength o...
Q: In biomass digestion , the monomers and oligomers is converted to short chain volatile organic liqui...
A: A molecule that combine with other molecules to form a more complex molecule can be called as monome...
Q: Calculate the percent composition by mass of cach element in AIKOH),. Use at least three significant...
A: Following are the atomic masses of the atoms in Al(OH)3: atomic mass:Al = 27 g/molO = 16 g/molH = 1 ...
Q: Indicate which orbitals overlap to form the s bonds in each compound.a. BeBr2b. HgCl2c. ICN
A: According to valence bond theory a covalent bond is formed by the sharing of valence electrons of ha...
Q: Predict whether a precipitate will form if you mix 175.0 mL of a 0.0055 M KCl solution with 145.0 mL...
A: The solution of KCl reacts with solution of AgNO3 to form the precipitate AgCl and a solution of KNO...
Q: What happened during the Cambrian explosion? Dinosaurs went extinct. Many animals first appeared. Si...
A: Cambrian period refers to the initial geological period of the Palaeozoic Era as well as Phanerozoic...
Q: . What is the molecular basis for the observation that the overall charge on a protein becomes incre...
A: Proteins are the large polymeric compounds made up of monomeric units, namely amino acids. There are...
Q: One method for the quantitative determination of the concentration of constituents in a sample analy...
A: Given: The relative areas are 16.4, 45.2 and 30.2. The relative detector responses are 0.60,...
Q: Which member of each pair of metals is more likely to corrode (oxidize)? (a) Mg or Ca (b) Au or Hg (...
A: In corrosion process, the metal behaves as anode. so that, it will get oxidize. Metals with lower st...
Q: Please refer to photo. Can you balance this equation and outline the steps?
A:
Q: By what factor must the temperature be changed for the relative population difference (Nɑ - Nβ)/N to...
A: The factor by which the temperature for H1 nuclei is changed as the overall populace expanded by 5 v...
Q: Determine the number of atoms in 19.0 grams of sodium, Na. (The mass of one mole of sodium is 22.99 ...
A: First convert the mass of sodium into moles. From this moles Calculate the number of atoms by using ...
Q: Suppose that when you exercise, you consume 100 g of ghucose and that all the energy rekased as heat...
A: Enthalpy of combustion of glucose = -2805 KJ/molMolecular mass of glucose = 180g/molGiven mass = 100...
Q: What products are formed when attached acetal is hydrolyzed with aqueousacid?
A: The reaction of hydrolysis with aqueous acid is as follows:
Q: Chemistry Question
A: It can be solved using the conversion relations: a) 1μL = 10-3 mL b) 1μL = 10-8 hL c) 1μL = 10-3 cm3...
Q: Determine the number of atoms in 1.96 mL of mercury. The density of mercury is 13.5 g/mL
A: Given: Density of mercury = 13.5 g/mL Volume of mercury = 1.96 mL We know, Densit...
Q: You have 150.0 mL of a 0.693 M solution of Ce(NO3)a. What mass (in grams) of Ce(NO3)4 Would be requi...
A: Moles of Ce(NO3)4 = Molarity x volume in liters = 0.693 x (15...
Q: Part 1. Arrange the following alkane compounds according to the increasing of boiling points. Use th...
A: Given alkanes: 1. heptane 2. methane 3. octane 4. ethane 5. 2,2,3-trimethylbutane 6. butane 7. 2,2,3...
Q: Name each alkene and specify its configuration by the E,Z system
A: Name of the following compound : Identify the longest chain in the skeletal number it. Add the subs...
Q: Macromolecules can also be formed from cyclic monomers such as cyclic hydrocarbons, ethers, esters, ...
A: Polymerization could be a method through that an outsized range of compound molecule...
Q: Choose the stronger acid from the pair. Provide a brief explanation. H.
A:
Q: Name the following compound
A: To write the IUPAC name first identify the longest C- chain.
Q: 3. Give the IUPAC names CH2CH3 CH = C-CH-CH3
A: To avoid confusion in the names of the organic compound, IUPAC formed some set of rules. The names f...
Q: How much heat (in kilocalories) is evolved or absorbed in the reaction of 1.00 g of Na with H2O? Is ...
A:
Q: Calculate the boiling temperature, freezing temperature and osmotic pressure at 20C of the mixture ...
A: There is no substance with name sakarosis If we are assuming that it is sucrose than sucrose can be...
Q: Briefly explain (i) Thompson Experiment OR (ii) Rutherford Experiment emphasizing the discovery(ies)...
A: Many models were proposed to determine the atomic structure of the element. Thomson and Rutherford's...
Q: Chemistry Question
A: Graduated cylinders, beakers, volumetric pipets, burets, and volumetric flasks are five kinds of gla...
Q: Which of the following two protons on each molecule is more acidic? Explain in terms of conjuga stab...
A: The first compound is 4-Mercaptocyclohexan-1-ol. The electronegativity of oxygen (3.5) is more than...
Q: What is the oxidation state of an element, like Cu or F2? O-1 02
A:
Q: Find the product by writing down the mechanism of the reactions given below.
A: The organic reactions are carried out with the help of various reagents. Each reaction is carried ou...
Q: At 350 K, the rate of decomposition of a gaseous compound was 10.01 Pa s−1 when 10.0 per cent had re...
A: The decomposition reaction involves the breaking of larger molecules into smaller fragments. Example...
Q: Dinitrogen monoxide can be prepared by heating ammonium nitrate, which decomposes into dinitrogen mo...
A: At STP , Volume of 1 mol gas = 22.4 L From this relation we can can convert the volume of gas into ...
Q: What is the relationship between the dipole moment and bond moment? Explain it with examples, how is...
A:
Q: Predict the masses and the structures of the most abundant fragments observed in the mass spectra of...
A: The masses and the structures of the most abundant fragments observed in the mass spectra of the cyc...
What product is formed when attached compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4, H2O?
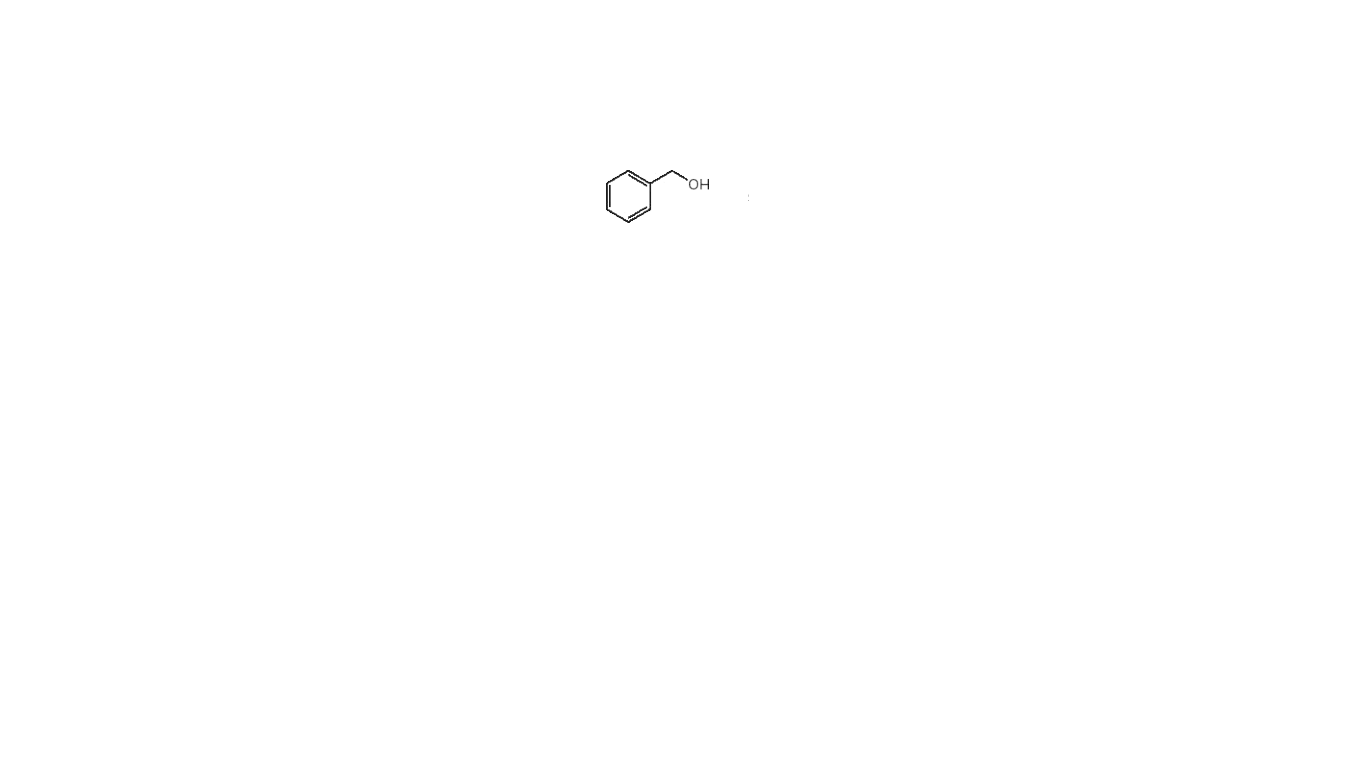
Step by step
Solved in 2 steps with 2 images


