Q: Identify the major product for each reaction
A: The nucleophilic substitution reaction of organolithium RLi with ester, the alcohol is formed as the...
Q: 4
A: The balanced chemical reaction is,
Q: eof Bayle's invw oenatan Turo in your dat D What is Boyle's law constant if a sealed rigid container...
A: Given: Volume= 500 mL=0.5 L Pressure= 128.1 kPa
Q: Before small batteries were available, carbide lamps were used for bicycle lights. Acetylene gas, C2...
A: The given reaction is, CaC2 + 2H2O → C2H2 + Ca(OH)2 The number of moles of calcium carbide present i...
Q: What is the molarity of an HCl solution if 43.6 mL of a 0.125 M KOH solution are needed to titrate a...
A: The above given equation is wrong : The correct equation for the reaction is : KOH +HCl------> H2...
Q: 2.
A: The relationship between the temperature in Kelvin and Fahrenheit is given as TK = (90 TF - 32) / 1....
Q: 5-Androstene-3,17-dione is isomerized to 4-androstene-3,17-dione by hydroxide ion. Propose a mechani...
A: Since the H between the C=O and C=C is acidic H due to resonance with C=O and C=C hence the H is tak...
Q: How many grams of solid calcium carbonate must decompose to produce solid calcium oxide and 526.0 mL...
A: The amount of solid calcium carbonate decomposed is to be calculated.
Q: For enzyme-catalyzed reactions that follow the mechanism E +S= E· S E·S= E+P a graph of the rate as ...
A: The enzyme catalysis reaction is shown as, The first step involves the reaction between enzyme and ...
Q: how many moles of F are needed to form 1.4 moles of XeF6?
A: The molecular formula of the compound is given as XeF6 hence we can say that the number of F atoms ...
Q: Dipalmitoylphosphatidylcholine is a surfactant found in the lining of the lungs. It prevents the lun...
A: Dipalmitoylphosphatidylcholine (DPPC )is a surfactant on the surface of the alveoli in the lungs. Th...
Q: Explain why the cyclopentadienide anion A gives only one signal in its 13C NMR spectrum.
A: Please find below the resonating structure of anion A.
Q: At 25°c ,the thermodynamic equilibrium constant for reaction 3C2H2(g) converts into C6H6(l) is K=1.0...
A: Since the change in Gibbs free energy of the reaction can be calculated by ΔGrxn = -RTln(K) where ...
Q: 2. For the equilibrium 2 SO2(g) + O2(g) ⇌ 2 SO3(g) ΔHo = –198 kJ.Predict the direction in which the ...
A: Le chatlier’s principle- This principle helps to determine effect of temperature on equilibrium of ...
Q: #17 on the picture (about alkalosis of a protein)
A:
Q: Calculate the energy, in joules, for a photon of 1024nm light
A: The energy of a photon can be expressed as follows-
Q: Find the pH at each of the following points for the titration of 25.0mL of 0.200M ethylamine with 0....
A: Since we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the question an...
Q: What is the molecular geometry of the following SeF4 SPF3 SOF4 ICl4-
A:
Q: Compounds containing triple bonds are also Diels–Alder dienophiles. With this in mind, draw the prod...
A:
Q: When an aqueous solution of 6.71 g of barium chloride was added to an aqueous solution of 7.77 g of ...
A: The reaction between barium chloride and potassium chromate is shown below, The given grams are con...
Q: unlv.instructure.com 2. Write the formula of the substance produced when a copper (II) ion is combin...
A: a) the formula of hydroxide ion is OH- Since Copper (II) ion will be Cu2+ hence to balance the 2+ ...
Q: Draw a short segment of nylon 44.
A: Nylon 44 polymer is formed by the reaction between butanediamine and 1,4-butandioic acid.
Q: Find the pH at each of the following points for the titration of 25.0mL of 0.200M ethylamine with 0....
A: Given: Titration of 25.0 mL of 0.200 M ethylamine with 0.150 M HCl.
Q: Identify one of the major products. Be sure to include all relevant sterochemistry using wedges and ...
A: Hey, since there are multiple questions posted, we will answer first question. If you want any speci...
Q: 1. What is the function of the cerebrospinal fluid? 2. What is a ganglion?
A: Since you did not mention which question, so we are answering 1st question for you:
Q: Chorismate mutase is an enzyme that promotes a pericyclic reaction by forcing the substrate to assum...
A: Chorismate mutase is an enzyme that promotes a pericyclic reaction by forcing the substrate to assum...
Q: SHOW ALL WORK FOR CREDIT!! A 18.0 gram sample of ice (H2O(s)) at 0 oC has 35.0 kJ of energy added to...
A:
Q: What is the temperature of 0.300 mole of neon in a 2.00 L vessel at 4.68 atm?
A: We will use Idea Gas Equation to solve this problem PV = nRT where. P = Pressure in atm V = Volume...
Q: Give the bond polarity for each type of bond in the molecule for ch3ch2oh?
A: The polarity of a bond depends upon the electronegativity difference between the atoms involved in b...
Q: For the reaction 2Co 3+ (aq)+2Cl − (aq)→2Co 2+ (aq)+Cl 2 (g). E ∘ =0.483 V what is the cell potentia...
A: Given information: Standard cell potential of the cell = 0.483 V Temperature = 25 ℃ Concentration of...
Q: A solution contains 1.0 x 10-5 M Ag+ and 2.0 x 10-6 M PO43- Will silver phosphate precipitate? Expla...
A: The solubility product constant (Ksp) represents the solubility of products at equilibrium for solid...
Q: Which polymer is more apt to contain abnormal head-to-head linkages: poly(vinyl chloride) or polysty...
A: Polystyrene is less apt to contain head to head linkages. It contains head to tail linkages. Poly v...
Q: Sarin is a nerve gas that was previously used as a chemical weapon. Considering the physical propert...
A: The substance exists as solid or liquid or gas as per the below condition 1) If the temperature is l...
Q: If the density of solid tungsten is 19.3 g/cm319.3 g/cm3, what is the packing efficiency of WW if it...
A: Given, The packing efficiency is calculated by the formula shown below.
Q: What is the cell potential of a concentration cell made by coupling a Zn2+(0.0100 M)/Zn half-cell to...
A:
Q: How many moles of HCl are contained in 16.66 mL of 0.775 M HCl solution? 0.0465 mol ...
A: Conversion of mL into L:
Q: For each reaction, identify the carbonyl AND the alkyl halide starting materials that form the produ...
A:
Q: At 40o C Kw = 2.9 x 10–14. What are the [H+], pH and pOH for water at40o C?
A: The ionic product of water Kw is temperature-dependent i.e. it changes with temperature. Therefore, ...
Q: 24. Methylamine, CH3NH4 is a weak base that ionizes in solution as shown by the following equation. ...
A:
Q: What would be the C-terminal amino acid if the codon at the 3′-end of the mRNA in 5′-AAA-GUU-GGC-UAC...
A: UUU UUC they belomg to Phe a. If the first base was changed to A GUC becomes AUC AUC responsible to ...
Q: Mn crystallizes in the same type of cubic unit cell as Cu. Assuming that the radius of Mn is 5.6% la...
A: Mn crysllizes in the same type of cubic cell as Cu The density of copper = 8.96 g/cm3 From the value...
Q: Balance the following reaction in acidic solution: Cr2O72- (aq) + I- (aq) ⟶Cr3+ (aq) +I2 (s)
A:
Q: Calculate the energy, in kJ, for a mole of photons if 1024nm light
A: Energy of a photon is given by E = hc/λ where E = energy in J h = Planks constant = 6.626 X 10-34 Js...
Q: 1) What is Boyle's law? What must be held constant in order for the Boyle's law relationship to work...
A:
Q: Infrared radiation from young stars can pass through the heavy dust clouds surrounding them, allowin...
A:
Q: Provide a plausible synthetic pathway for the following organic transformation starting with acetic ...
A: The alkyl derivatives of Li and Cu acting as useful reagents in many reactions are known as Gilman r...
Q: Cyclohex-2-enone has two protons on its carbon–carbon double bond (labeled Ha and Hb) and two proton...
A: Given compound:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients ...
A: The given reaction is, Hg2+ + Mn2+ → Hg + MnO4- The anode half-reaction is, Mn2+ → MnO4- The cathode...
Q: provid Lewis structure, molecular geometry and hybridization for. CH4. BH3. PF5. XeF2. SF6. NO^- 2....
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
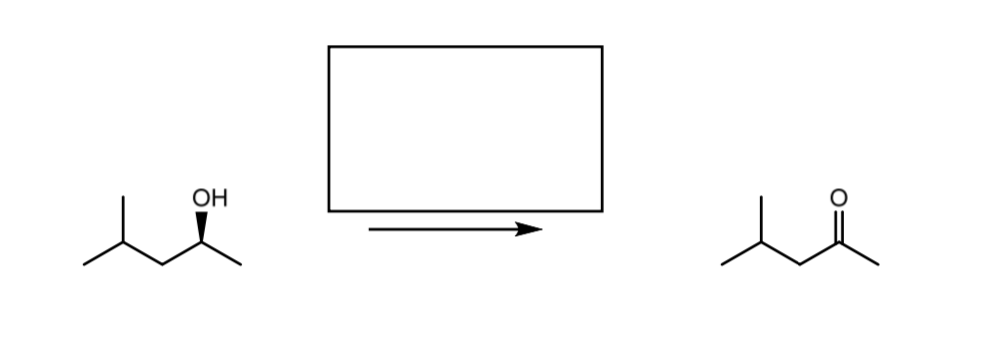
I am trying to find what reagent will cause this reaction to take place.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


