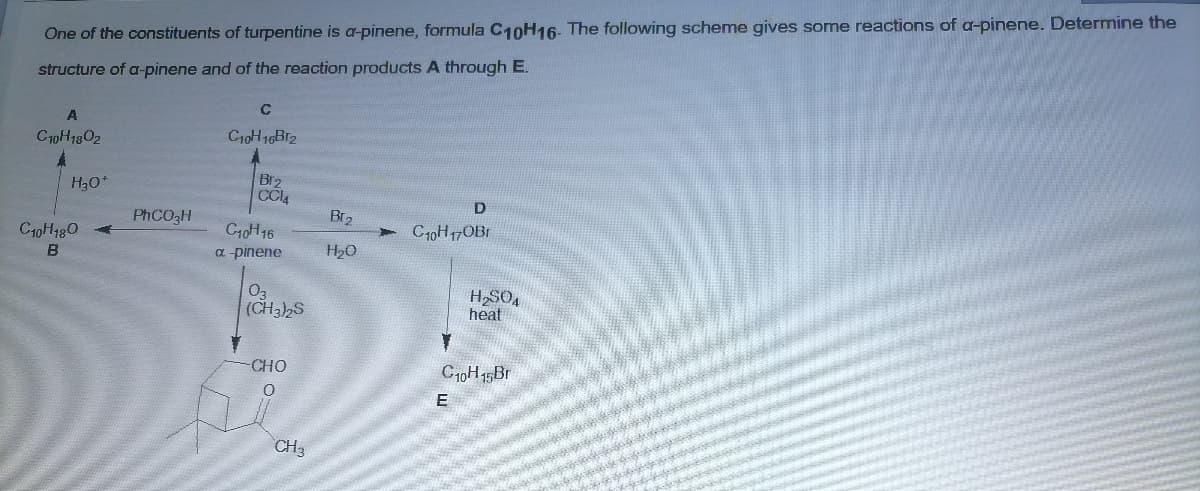
One of the constituents of turpentine is a-pinene, formula C10H16. The following scheme gives some reactions of a-pinene. Determine the structure of a-pinene and of the reaction products A through E. C C10H1302 C10H 16B12 Br2 CCl4 H30+ D PHCO3H Br2 C40H480 C10H16 C10H17OBI B a pinene H20 03 (CH3)2S heat CHO E CH
Q: Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to…
A:
Q: Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to…
A: Given target compound, Step 1 Nitration of benzene ring by the reaction of benzene with nitrating…
Q: Q5.) Two isomeric aromatic compounds X and Y have the following percentage composition by lmass! C =…
A:
Q: An unknown hydrocarbon G, whose formula is C16H26 contains two triple bonds. Ozonolysis followed by…
A: Ozonolysis: Ozonolysis is an organic reaction where the unsaturated bonds of alkenes, alkynes,…
Q: Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to…
A:
Q: A compound having the molecular formula C22H32O2 was isolated in 66% yield in the following…
A: Olefin metathesis 1-In olefin metathesis, two olefin compounds form a four-membered ring…
Q: An unknown hydrocarbon A with the formula C6H10 reacts with 1 molar equivalent of H2 over a…
A:
Q: An unknown hydrocarbon decolorizes bromine in CCL4, and it undergoes catalytic reduction to give…
A: Applying concept of organic synthesis.
Q: 2 C,H9Lİ [C;H6 j* (Li*); 2 C4H10 | -
A: Ans
Q: Chemistry Provide the product structure for the reaction of acetylene with the following sequence of…
A: (1)->NaNH2 (2)->CH3CH2Br (3)->H2/Lindlar catalyst (4)->BH3 - THF (5)->H2O2,NaOH
Q: Upon ozonolysis, Compound X produces two compounds: Compound Y and Compound Z. Compound Y can also…
A:
Q: Benzene, C6H6 undergoes substitution reaction with concentrated nitric acid, HNO3 to produce…
A: In organic chemistry, inter conversion of organic molecule takes place from one form to another form…
Q: For following substituted benzenes: [1] C6H5Br; [2] C6H5CN; [3] C6H5OCOCH3: On balance, does the…
A: Electrophilic substitution reactions are most common reactions of aromatic compounds. In these…
Q: 5. An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: The given unknown hydrocarbon A with formula C6H12reacts with 1 molar equivalent of H2 over a Pd…
Q: Q5.) Two isomeric aromatic compounds X and Y have the following percentage composition by mass: C =…
A: Molecular formula from percentage composition Convert mass of each atom into moles Find the…
Q: - The compound (X) is formed from catalytic hydration of ethene. The compound (X) react with the…
A:
Q: Nucleophilic substitution happens on compounds having nucleophilic groups as leaving groups. The…
A: The compounds given are,
Q: Adipic acid is an important bifunctional molecule that can be Но, used as a linker between proteins…
A:
Q: 1) NaH 2) CH3CH2CH2B. РСС, THF NazCr2O7
A:
Q: Compound A has the formula C9H19Cl. B is a C9H19Br compound.A and B undergo base-promoted E2…
A: According to the question, A and B are haloalkanes that on reaction with a base form an alkene C as…
Q: O: C14H1206 maleic anhydride in excess
A: The reaction in which conjugated diene react with dienophile is known as Diels alder reaction.
Q: Give the structure including the stereochemistry of the product formed in the following reaction.…
A: organometallic chemistry.
Q: Stearolic acid, C18H32O2, yields stearic acid on catalytic hydrogenation and undergoes oxidative…
A: , From the given data, the structure of stearic acid is , R
Q: Compound A (C 7H 11Br) is treated with magnesium in ether to give B (C 7H 11MgBr), which reacts…
A: Organic reactions:
Q: The compound BD3 is a deuterated form of borane. Predict the product formed when 1-methylcyclohexene…
A: Given compound: 1-methylcyclohexene
Q: Provide the structure of the reactants necessary to prepare the following compound using the Wittig…
A: The structure of the reactant needed to prepare the given product using wittig reaction = ?
Q: An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A:
Q: Compound X (structure shown below) has a molecular formula C5H10 and reacts with H2/Pt to give…
A:
Q: Hydrocarbon X has the formula C6H12. X reacts with one molar equivalent of hydrogen in the presence…
A: Given compound X has molecular formula, C6H12
Q: Complete the following reaction by supplying the product. If more than one product is predicted give…
A: The electrons rich species which carry either negative charge or lone pair of electrons are…
Q: Compound A, C10H180, undergoes a reaction with concentrated sulfuric acid under heat, to yield a…
A: Organic reaction mechanisms.
Q: A dibromide loses only one bromine when it reacts with sodium hydroxide. The dibromide forms toluene…
A: Given that, A dibromide loses only one bromine when it reacts with sodium hydroxide. The dibromide…
Q: The following product is expected from the reaction of 3-heptyne with sodium metal in liquid…
A:
Q: 7. Pick two of the following compounds, and provide a synthetic scheme to produce the following…
A:
Q: b) An alkyl halide W has the molecular formula C4H,Br. One of the isomers, X can rotate…
A: Given : W has the molecular formula C4H9Br. X can rotate plane-polarised light. Y is a primary…
Q: An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: Reduction of alkene gives alkane.
Q: There are 20 commonly occurring amino acids from which all proteins are derived, although many other…
A: The structures are
Q: Compound name is Toluene . Name the structures of all possible chemical (Electrophilic aromatic…
A: All the reaction schemes are shown below including name and structure of all the products. The…
Q: Compound AA has a molecular formula of C3H6O and gives a positive
A: The given molecular formula of the compound is C3H6O. If it's give positive test with tollens…
Q: Two moles of organic compound ‘A’ on treatment with a strong base gives two compounds ‘B’ and ‘C’.…
A: It is given that 2 moles of a compound A are undergoing a reaction with a strong base, which gives…
Q: Isoerythrogenic acid, C18H26O2, is an acetylic fatty acid that turns a vivid blue on exposure to UV…
A: Based on the data given above, the line structure is shown below- It is also known as isanic acid.
Q: An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: H2/of hydrogenate alkene into alkane. OsO4 adds two hydroxy group on the double bond via syn…
Q: Compounds X has the formala CyH15CI: Y is CH15Br. X undergoes base-promoted E2 elimination to give a…
A: Both product X and Y are formed by E2 elimination reaction. X and Y are isomeric. X undergoes SN2…
Q: For the compound p-anisidne 2. Name and draw the structures of all possible chemical (Electrophilic…
A: We have to write the all reactions
Q: Myristoleic acid, C14H26O2, yields myristic acid on catalytic hydrogenation and undergoes oxidative…
A:
Q: Describe the ozonolysis of alkenes one mole of a hydrocarbon(A) reacts with one mole of beomine…
A: one mole of a hydrocarbon (A) reacts with one mole of bromine. So, the dibromo compound will have…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- In which of the statements given below, the event is not an oxidation reaction? A. Product formation as a result of the reaction of benzoine with nitric acid B. Formation of the product as a result of the reaction of 2-propene at KMnO4 / Basic ambient temperature C. Formation of the product as a result of the reaction of 2-propene in KMnO4 / Basic medium cold D. CH3CH2OH + KMnO4 → CH3COOH E. Reaction of an alkyl halide with ammoniaOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Describe the ozonolysis of alkenes one mole of a hydrocarbon(A) reacts with one mole of beomine giving a dibromo compound C5H10Br2.Substance A on treatment with cold dilute kMnO4 solution forms a compound C5C12O2(C5H12O2) on ozonolysis A,gives equimolar quantities of propanone and ethanol.Deduce the structure of substance A.
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Stearolic acid, C18H32O2, yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of stearolic acid?Compound P (C2H4) which is an alkene undergoes reaction with HCl to produce compound Q (C2H5Cl). Reaction of compound Q with benzene in the presence of AlCl3 as catalyst produces compound R. Then, nitration of compound R in the presence ofnH2SO4 produces two compounds, S and T. But when compound R is reacted with a hot acidified solution of alkaline KMnO4 gives compound U. Deduce the structure of compounds P to U.
- the organic compound 2-heptanone, belonging to the ketone family, is responsible for the strong penetrating odor in Roquefort cheeses. Starting from acetylene as the starting reagent, propose a synthesis line with the reaction mechanisms involved for the synthetic obtaining of 2-heptanone and use it as a food additive in analogous cheeses.Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)2,4,6-Trinitrotoluene (TNT)(i) Name, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the presence of H2SO4 and provide a clear rationale as to why this is the major product of the reaction. (ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.
- Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.) 1-Bromo-3-nitrobenzeneCan you please check my work on the following ochem reaction scheme and let me know if it is correct or what is wrong... the question was: Consider 3,4-dimethylpiperidine being subjected to the following: Step 1: CH3I (excess); Step 2: NaOH, heat Step 3: CH3I (excess); Step 4: NaOH, heat Provide the bond line structures for the major organic product obtained in each step and discuss the regiochemistry for Step 2.To synthesize trans-cinnamic acid from benzaldehyde (PhCHO) and acetic anhydride (Ac2O) under basic, refluxing conditions to exemplify the Perkin condensation. 1. Draw the complete chemical reaction to be carried out. 2. Provide a complete arrow-pushing mechanism for the synthesis of trans-cinnamic acid. 3. Briefly describe the role of water, Na2CO3, and HCl in the isolation of the product. Draw the structure of the product at basic pH (after Na2CO3 addition) and at acidic pH (after HCl addition).

