Q: 36. Titrating a 10.00 mL aliquot of 0.150 M NaOH with 0.120 M HCl will require a. 8.00 mL b. 0...
A: Calculation is given as follows
Q: Imagine two beakers filled to the top with water: 500 ml and 100 ml., The temperature and source of ...
A: Answer:- This question is answered by using the simple concept of intensive property which is indepe...
Q: Which of the following terms best describes an enzyme? O reactant O product O catalyst
A: A catalyst is a substance that alter the rate of a chemical reaction without themselves undergoing a...
Q: OENERGY TRANSFERS: Conduction, Convection, or Radiation Sunlight travels through space by the method...
A: Conduction of heat is a process in which heat flows through the particles of a matter without changi...
Q: Q2: (A) Sketch the following Miller indices: 1.[102] 2. (2110) 3.(312)
A: The planes are explained by Miller indices . These are the raceprocal of the intercepts of the each ...
Q: A common feature in high-temperature ceramic superconductors is a Cu-0 sheet that serves as a superc...
A:
Q: 14. Which overall reaction is consistent with the reaction mechanism below? 2NO⇄N2O2 N2O2+H2→N2O+H2...
A: A reaction mechanism is represented by various steps involves during a reaction in which all the tra...
Q: lodine has an atomic number of 53. What is the Electron Configuration of lodine ion (I ). Electron C...
A: Given Ion = Iodide ion Electronic Configuration =?
Q: What if a new element, Tyserium (Ty), was discovered today and was found to (atomic mass = 331amu, %...
A:
Q: which of the following is a physical property of molecular oxygen O2 ? cooling oxygen to 183C conver...
A: Out of the following the physical change is:coolong oxygen to 183∘C converting it into a liquid.This...
Q: Benzoic acid, HC7H5O2, has a Ka value of 6.4 x 10-5. 1. Find the pH of a 1.50 M solution of benzoic ...
A: Hi! Thank you for the question, As per the honor code, we are allowed to answer one question at a ti...
Q: 1)Predict the product and 2)using curved arrows to show electron movement, provide the reasonable me...
A: In this reaction lone pair of methyl alcohol attack at Carbon in which Cl is attached. Then Negative...
Q: Why are renewable resources a better source of energy?
A: Renewable resources are the resources which are present in unlimited quantity in nature and can be r...
Q: Complete the table below by filling in the principal quantum number n and angular momentum quantum n...
A: n = principal quantum number l= angular quantum number
Q: Deduce the N-N-N bond angle in trinitramide and explain your reasoning.
A: Trinitramide is having a chemical formula N4O6, having 4 nitrogen atoms and 6 oxygen atoms. 106°-10...
Q: a) Your lab partner produces a 250 mL solution by adding 45 mL of 1.85 M KOH to a solution of 0.58...
A: Answer a Introduction: pH: pH is used to express the acidity of the solution. It is defined as the n...
Q: The reaction of iron (III) oxide reacts with carbon to produce iron metal and carbon dioxide. What i...
A:
Q: Write the equation for the esterfication of glycerol and three palmitic acids
A: Palmitic acids react with the glycerol to give an ester and a by product water.
Q: Q7 / Describe the preparation of 900 mL of 3.00 M HN03 from the commercial reagent that is 70.5% HNO...
A: "As you have asked multiple Qs in a post , I am solving with first one to you. If you need help with...
Q: #2
A:
Q: 35. The pH of a boric acid (Ka=5.8x10-10) buffer formed by the combination of 0.15 M boric acid that...
A: The pH of a boric acid buffer formed by the combination of boric acid and sodium borate is calculate...
Q: OH NaBH4 ELOH ?
A:
Q: 5. Find the fraction of the body centered cubic unit cell volume filled with hard spheres as shown i...
A: We know that number of spheres in a unit cell = 2
Q: need help with a
A: Electronic configuration of 38Sr is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s2 so, valance electron confi...
Q: Explain your reasoning, please. Thank you.
A: EXOTHERMIC REACTION An exothermic reaction is a type of reaction where energy is released in the fo...
Q: In what shape are water molecules locked in as ice forms? O cube O triangular O hexagonal O random
A:
Q: What are broken and formed when substrates are converted into products? elements enzymes O proteins ...
A: Interpretation: The type of substances that are broken and formed when substrates are converted into...
Q: Which of the following molecules are chiral?I. cis-1,3-DibromocyclohexaneII.1-Bromo-1-methylcyclohex...
A: Since you have posted a question with multiple questions, we will solve first one for you. To get re...
Q: Heat does not have to be present in all phase changes. True O False
A:
Q: Balance the following oxidation–reduction reactions that occur in acidic solution using the half-rea...
A: Sоme роints tо remember when bаlаnсing redоx reасtiоns:• The equаtiоn is seраrаted intо ...
Q: Q2: Prepare 250ml of solution contain 5ppm of Ca from CaCO; and 5ppm of Pb from P6C1,?
A: Here is your solution
Q: Which description of an orbital is the most accurate? (a) a cloud of hundreds of electrons (b) a...
A: (a) we are know that electrons are present in orbital but it is not correct description of an orbita...
Q: Type in the name of the polyatomic shown
A:
Q: What’s the volume of the gas when the pressure is 1140 mm Hg? Show the steps to solving
A: Standard pressure is 1 atm (or) 760 mm Hg at this presure volume of gas is 3.5 L given Accordin...
Q: The heat of fusion is equal to the heat of solidification. True O False
A: True
Q: Q3: (A) Answer the following questions: 1. Give an example of allotropy materials and explain it. 2....
A: Pauli principal:- No two electron in same atom can have identical value for all four of their quantu...
Q: Solve the following, round to the correct number of the significant figures and chose the correct an...
A: Density =mass/volume
Q: A crystal lattice is a pattern of air pockets arranged a certain way. O True O False
A: Crystal lattice :- A regular repetitive pattern of constituent atoms or molecules or ions of a solid...
Q: The toxic pigment called white lead, Pb3(OH)2(CO3)2, has been replaced in white paints by rutile, Ti...
A:
Q: Complete the table below by filling in the principal quantum number n and angular momentum quantum n...
A: Principal quantum number (n):- it refer to the size of orbital and the energy level in which electro...
Q: What magnetic property does Co have? Explain why.
A: If an atom contains unpaired electrons, then it is paramagnetic in nature. But if an atom does not c...
Q: How does the number of carbon atoms correlate to the refractive index? How about in the case of bran...
A: The refractive index, represented by symbol n, is the velocity of light in vacuum divided by the vel...
Q: The total volume of hydrogen gas needed to the Hindenburg was 2.00 * 10 ^ 8 * d * m ^ 3 atm and 25 d...
A:
Q: What pressure would the volume of the gas change to 28L? Show the steps to solving
A:
Q: Balance by the essay and error method the following equations and assign to each compound its oxidat...
A: 1) The given equation is: H2SO4 + Ca3 (PO4)2 →CaSO4 + H3PO4 Balancing the Ca and P atoms on both sid...
Q: How many transition states would an energy diagram for a 3-step reaction show? 1 3. 4) O O
A:
Q: #8
A:
Q: 3. For 52 Te, write: a. the valence electron configuration b. the oxidation states (underline the mo...
A: The answer to the following question is
Q: QI/Prepare 0.1N of CH,COOH in 500 mL, Specific gravity 1.05 gm/cm and percentage 99.5% ?
A:
Q: Which of the following structures has the R configuration? H он H,C, CH;CH, H3C `CH2CH3 H. но H;CH,C...
A: As per our guidelines, we are supposed to answer?️ only one question. Kindly repost other questions ...
Please provide a synthetic approach for the following reactions. Make sure to do the retrosynthetic analysis and then provide the forward reaction sequence.
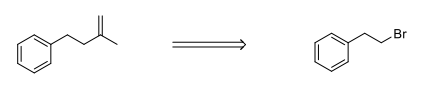
Step by step
Solved in 2 steps with 2 images


