Q: H3C. O HNO3 H₂SO4 NO₂ & NO₂ Q Search C
A:
Q: 18. Which statement(s) is/are true regarding chiral carbons? A. All chiral carbons must have a…
A: The chiral carbon atoms are carbon atoms that are attached to four different atoms or groups of…
Q: 4) Listen Provide the correct IUPAC name for this molecule H.C-CH2CH H₂C H₂C CH₂ CH₂ P
A:
Q: Choose the element that is found in all organic compounds Ohydrogen sulphur oxygen nitrogen O carbon
A: Organic compound are formed by generally carbon , hydrogen dueto high catenation property of carbon.
Q: A Strong Acid When HCl dissolves in water, it ionizes completely. HC COPY H₂O CT H₂O H₂O H₂O HF HF ✓…
A: Here some images are provided, let us explain the terms an images used in the given pictures.
Q: Answer the questions in the table below about the shape of the carbon dioxide (CO₂) molecule. How…
A: A lone pair is a pair of valence electrons that are not involved in a chemical bond between atoms.…
Q: ZI pH 4-5 N CH3CH2CH2C(O)CH2CH2CH3 Q 4
A: An enamine is an organic compound that contains both an amino group (-NH2) and a carbonyl group…
Q: 4. Mary drew the following structure for NO2¹- below. Do you agree or disagree with Mary's…
A:
Q: The correct name for the compound given 0=C-CH₂-CH₂ H -N -CH₂-CH₂-CH₂ N-propylpropanamide…
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Draw the major product of the substitution reaction shown below. Ignore any inorganic byproducts.
A: We have been given an organic reaction and we have been asked to draw the major product of given…
Q: rogen peroxide can decompose to water and oxygen by the following reaction: 2 H₂O₂(1)→2 H₂O(l) +…
A: Thermodynamic is the branch of chemistry that mainly deals with the heat transfer between system and…
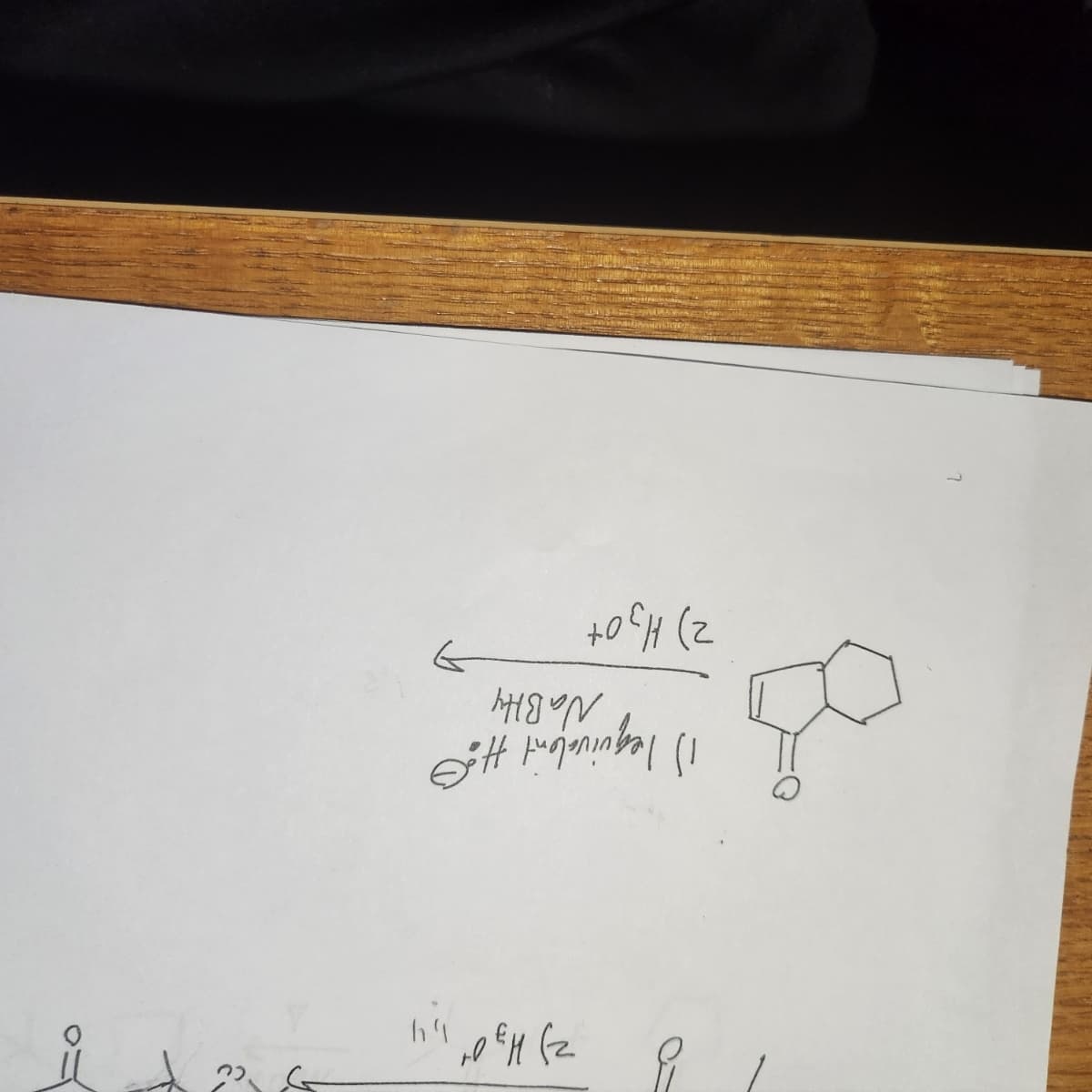
Q: 8. Identify the reagents needed to carry out the following conversion. 0 ? HC=CCH₂CH₂CH3 A.…
A:
Q: Identify the most stable radical structure.
A:
Q: The following information is given for manganese at 1 atm: T2095.00°C Tm 1244.00°C Specific heat…
A: Answer: Heat supplied to solid Manganese will be used in two steps: 1. To raise the temperature of…
Q: 1. Calculate the molar mass (molecular weight) of the following compounds a. K,PO b. Al(NO₂),
A: The molar mass of any compound is the total sum of molar mass of all the atoms or elements present…
Q: (a) (c) CH3 HOO d) (b) Al(OH)3 + H₂SO4 CH3 102 CH3-CH-O-CCH2–CH–CH3 OH + + H₂O OH + H* H₂O H+ H₂SO4
A: We have to decide the types of reaction for the given reactions
Q: Enter the chemical formula of a binary molecular compound of hydrogen and a Group 7A element that…
A: The question is based on the concept of chemical compounds. we need to identify a compound of…
Q: An initial gas sample of NOBr decomposes according to the reaction below. The decomposition of NOBr…
A:
Q: 2. Given that there is transport of 4 moles of A and 2 moles of B towards a permeable membrane from…
A: Solution - The diffusion equation is a parabolic partial differential equation. it describes the…
Q: A chemist titrates 60.0 mL of a 0.5826 M potassium hydroxide (KOH) solution with 0.0606M HNO,…
A: Given : Volume of KOH = 60.0 ml Molar Concentration of KOH = 0.5826 M Molar Concentration of HNO3…
Q: please draw exactly how the answer should be represented. Use the following molecule as template for…
A: I have drawn this structure using Chemdraw which is given below:
Q: State, with justification, whether you expect nitric oxide (NO) to be a stronger or weaker…
A: I expect nitric oxide (NO) to be a stronger pi-acceptor ligand than carbon monoxide (CO). This is…
Q: a) Explain the general diagnostic principles using radiopharmaceuticals by which medical images may…
A: Solution - Radiopharmaceuticals - Radiopharmaceuticals are agents used to diagnose certain medical…
Q: EXAMPLE 2.14 For any gas, equation 2.48 can be written as (y-1)/y T (A) - 1/1 T₁ For an adiabatic,…
A:
Q: Write the net ionic equation for the following reaction, a. Lead Nitrate and Sodium Oxalate…
A: The net ionic equation for the given reactions are as follows:
Q: Calculate the molar solubility of SrC₂O4 in a solution that has a fixed H30 concentration of (Ksp…
A: Given Ksp of SrC2O4(s) = 5.0*10-8 For H2C2O4, Ka1 = 5.60*10-2 , and Ka2 = 5.42*10-5 .
Q: How many moles of Aluminum are needed to react with 15 moles of Hydrochloric Acid. If you…
A:
Q: 22.12 (SYN) Show how to carry out each of the following transformations. (a) (c) تنى:b نت تحمل…
A:
Q: . which species is not an electrolyte? HBr, LiCl, Ne, KOH, NaNO3 2. which compound has the highest…
A: Since, Electrolyte are those substance which can be dissolve in water then completed dissociated…
Q: An electric range burner weighing 602.0 grams is turned off after reaching a temperature of 464.7°C,…
A:
Q: The chemical formula of ibuprofen is C22H₂N₂O. Ibuprofen is an over-the-counter drug for treating…
A: a. we have to calculate moles of 43.9 g ibuprofen
Q: . When H2SO4 is neutralized by NaOH in aq solution, the net ionic equation is ____. 2. Combining…
A: Since, Balanced reaction means that both side number of atom present in equal number. Thus, For…
Q: Calculate the energy (in J) required to change the temperature of 15.0 g of ice at -25.0 oC to water…
A: Write the expression to relate the amount of heat required to change the temperature of 1kg mass by…
Q: Three samples of three different gases are listed in the table below. All the samples contain…
A: The ideal gas equation gives the relation between pressure, volume, temperature, and the number of…
Q: Draw the structural or line bond formulas of the following compounds. 2,2 dichloropropane…
A: Structural formula represents the atoms along with bonds while bond line formulaa represents only…
Q: If you find that [CoCl42-(aq)] = 0.783 M, [Co(H2O)62+(aq)] = 0.569 M, and [Cl-(aq)] = 0.495 M at 6…
A:
Q: Calculate the solubility of Fe(OH)2 in water at 25 °C. You'll find Kp data in the ALEKS Data tab.…
A:
Q: 4. Draw the three organic product(s) of the following reaction. Why might this not be a very useful…
A: Product of following reaction are as follow.
Q: Reagents available « NINH, b. H₂, Lindlar's catalyst c. 2 Na, NH3 (liq) d. H₂, Pd/C e. OsO4, H₂O₂ 1.…
A: The given problem is based on the functional group interconversions in organic chemistry. Synthesize…
Q: A B 9.38 Which diagram represents what happens when HCN dissolves in water? Explain your choice. A B…
A: The question is based on the concept of atomic models. we need to identify the correct diagram for…
Q: Identity the conjugation acid-base pairs in the following reactions. An acid donates a proton to…
A: Since, An acid donates a proton to become aconjugate base. A base accepts a proton to form a…
Q: Identify the strongest intermolecular force in each of the following compounds. Give a reason for…
A: Intermolecular forces are the attractive or repulsive interactions between molecules that determine…
Q: From the graph, determine the solubility of KNO3 at 35° C. 2. From the graph, determine the…
A: To find out the solubility of KNO3 at 35°C by using the graph of gram of KNO3 per 100 g of water…
Q: true or false? - 3 The enthalpy changes for the reaction CO₂(g) + 3H₂(g) CH₂OH(g) + H₂O(g) exceeds…
A: The enthalpy change of a reaction is given by the difference between sum of enthalpies of products…
Q: What is the Chemical equation for nickel iron alkaline and nickel cadmium battery
A: A question based on battery cells. Electrochemical equations representing overall cell reactions of…
Q: Raffinose is a trisaccharide found in whole grains. What is the correct description of the…
A: The linkage of two monosaccharides through oxygen is known as glycosidic linkage.
Q: Draw a Newman projection for the molecule below from the perspective indicated. Which of the groups…
A: The term "gauche interaction" refers to a type of steric hindrance that occurs between two…
Q: Assume that 28.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a…
A: Acid and base react with each other to form salt and water. The nature of salt depends on the nature…
Q: A chemist must dilute 90.2 mL of 6.73 M aqueous silver perchlorate (AgCIO4) solution until the…
A:
Q: (a) Which of the following octahedral complexes are chiral: cis-[CoCl₂(en)₂], [Cr(ox)3]³¯¯,…
A: If a molecule or complex has centre of inversion or plane of symmetry then the molecule is optically…
Step by step
Solved in 2 steps with 2 images


