Q: A solution is prepared at 25 °C that is initially 0.48 M in propanoic acid (HC₂H,CO₂), a weak acid…
A: Concentration of propanoic acid ([HC2H5CO2] = 0.48 M Concentration of potassium propanoate…
Q: Suppose you perform an experiment tracking the formation of a product over time, resulting in the…
A: We have a graph representing the formation of product over time. We will discuss about the different…
Q: Provide the mechanism of the transformations below. 1. TFA (in red box) is trifluoroacetic acid.…
A: The given transformation is an example of the deprotection reaction of amine catalysed by trifluoro…
Q: presented by M For the reaction shown, draw the transient product of one equivalent of reagent…
A: The given reaction is an example of reduction reaction of alkyne. First reduction of alkyne results…
Q: SnBu3 Br starting materials product transmetallation intermediate Pd(PPH3)4 Ph3P-Pdº-PPh3 Bu-Sn-Br…
A: This is a C-C coupling reaction, known as stille coupling. Here two types of starting materials are…
Q: TsOH, H2O 0 ОН
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: A certain enzymatic reaction is found to be zero ordaith respect to the substrate being converted.…
A: Order of reaction = zero Rate constant (k) = 540 Ms-1 Initial concentration of substrate (C) =…
Q: 55 >> Macmillan Learning CI Draw the alkyne formed when 1,1-dichloro-3-methylbutane is treated with…
A: We know sodamide acts as a base and favours the removal of Cl .
Q: Use the References to access important values if needed for this question. A gas is compressed and…
A: Given, The work on the gas from surroundings = 181 J The absorbed heat by the gas = 235 J.
Q: Propose a synthesis HO 1 goer OEt
A: The given transformation can be done by reaction of starting material with several grignards…
Q: K Indicate a complete name for the following compound: α- D-Allose eTextbook and Media Hint HO CH₂OH…
A: There are three main types of carbohydrates: monosaccharides, disaccharides, and polysaccharides.…
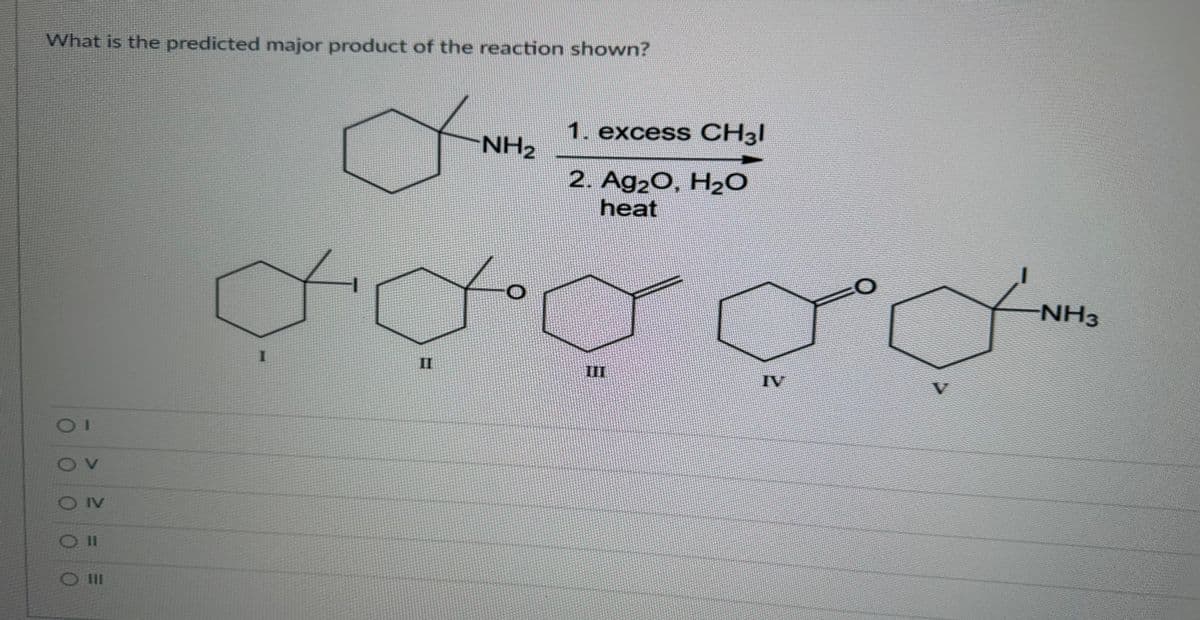
Q: What is the predicted product of the reaction shown? ον ο ο ο ο ο >= 2 O IV Oll Br 1. NaN3 2. H₂, Pd…
A:
Q: A sealed vessel containing 2.00 L of gas at 160 atm is measured to be 47.0 °C. The pressure is then…
A: Consider the given information as follows; Initial volume V1 = 2.00 L Initial pressure P1 = 160 atm…
Q: Name the cycloalkene that would react with chlorine to form the following. CH3. CH3 Name of…
A: Question detail
Q: Predict the major product for the following reaction. CN heat Modify the given structure of the…
A: The first reaction is hydrolysis of nitrile which gives carboxylic acid as a product. The second…
Q: Which of the following best describes the effect of an alkoxyl group substituent on electrophilic…
A: When all the properties of a molecule cannot be explained by a single structure , we draw more than…
Q: Determine the missing pieces in the following reaction. How many liters of gas would be produced…
A:
Q: Part A What is the vapor pressure of SiCl, in mmHg at 23.0 °C? The vapor pressure of SiCL, is 100…
A: Information
Q: C 1.2 Na, NH3 2. D₂, Pd 3
A:
Q: For the dehydrohalogenation (E2) reaction, draw the major organic product(s), inclu CI H H KOH…
A:
Q: 1) Provide an arow-pushing mechanism for the reaction shown below. NH3, [H₂SO4] NaBH3CN NH₂
A: The question is based on the concept of organic reactions. we need to write mechanism for given…
Q: Which has the greater number of particles, 3.011x1023 N2 molecules or 14.01 g N2 molecules? a.…
A:
Q: 2.16) Using the table of fatty acids, draw the SKELETAL structure of an unsaturated triglyceride.…
A: A question based on biologically important chemicals. The skeletal structure of an unsaturated fatty…
Q: Which of the following statements are true about the cathode during the operation of a voltaic…
A:
Q: When a base accepts a proton (H+) from another species, the species that is created is the:…
A:
Q: Which of the following characteristics of perchlorate (CIO4) best explains why perchloric acid…
A:
Q: Explain the difference between an indicator endpoint for a titration analysis and the true…
A: Titration is a laboratory technique used to determine the concentration of a substance (the analyte)…
Q: How many ml of 0.643 Molar HCl are needed to react completely with 72.3 ml. of 0.302 M NaOH…
A: Sodium hydroxide reacts with hydrochloric acid, to form sodium chloride and water. The equation for…
Q: The ratio of C-14 to C-12 in a piece of wood from an archeological site was found to be only 0.380…
A: we have to calculate the age of the wood
Q: 7. Determine the Ka of each of the following. a. A 0.00475 M solution of acid HY has a pH of 2.68.…
A:
Q: How many liters of a 0.209 M KI solution is needed to completely react with 2.43 g of Cu(NO₃)₂…
A: The reaction is balanced that is the number of each type of atom on the reactant side is same as…
Q: What’s the molarity of a KCl (Molar mass = 74.55 g/mol) solution containing 22.0 grams of KCl in…
A:
Q: Given the balanced equation: 3 Mg + Fe2O3 --> 2 Fe + 3 MgO Molar masses (g/mol): Mg = 24.31; Fe2O3 =…
A: Mass of Mg consumed = 55.00 g In next question, mass of mg 17.25 g and mass of Fe2O3 17.25 g…
Q: Given the balanced equation: 2 Na + ZnI2 --> 2 NaI + Zn Molar masses (g/mol): Na = 22.99; ZnI2 =…
A: The balanced equation is 2 Na + ZnI2--> 2NaI + Zn Moles of ZnI2=0.895 mol Mass of Na=64.16 g…
Q: Identify the expected major product of the following reaction. OV OIV ош T ОН 11 ОН ОН 1.…
A: A reagent is a substance that is used in a chemical reaction to cause a chemical transformation.…
Q: What is the shorthand notation that represents the following galvanic cell reaction? Sc(s)+…
A: Recall the galvanic cell reaction, Sc s + AlNO33 aq → ScNO33…
Q: Which one is more acidic than the other? Explain using one or more than one of the following terms:…
A: Acidity is defined as the ability of a molecule to release protons. if a compound easily donate…
Q: In the laboratory you dissolve 15.4 g of zinc sulfate in a volumetric flask and add water to a total…
A: Consider the given information as follows; Mass of zinc sulfate ZnSO4 = 15.4 g Volume of the…
Q: The chemical environment of protons can be deduced from their chemical shifts. What are the relative…
A: 1H-NMR spectroscopy is mainly used for to complete identification of the structure of unknown…
Q: 1. Cl₂, FeCl3 2. HNO3, H₂SO4 4. NaOH 5. 0 H₂SO4, heat OH 6. Zn(Hg), HCI
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: Which do you predict to have a higher boiling point and why? 1. CH3OH 2. CH3CH₂CH₂OH Because 3. more…
A: we have to tell which has larger boiling point and why
Q: FIS Ho- HO
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: Compare the solubility of lead chromate in each of the following aqueous solution 0.10 M Pb(CH3COO)2…
A:
Q: Determine the molecular and empirical formulas for the substance shown in the ball-and-stick model…
A: The molecular formula and empirical formula are two ways of representing the chemical formula of a…
Q: Consider the following speed distributions for different gases at the same temperature. sessus 0 O₂…
A: The distribution of the speed of gas molecules at various temperatures can be understood by the…
Q: Identify the correct dipole for the N-H bond in the Lewis structure of ammonia shown using the…
A: we have to interprete the position of charges
Q: Calculate the number of moles Na2C2O4 used in each of the three samples. Calculate the concentration…
A:
Q: Which of the following are valid transitions that produce lines in the emission spectrum of cadmium?…
A: For emission spectrum electron jumps from higher energy orbital to lower energy orbital. Here for…
Q: When heated to a temperature of 321 °C, formic acid, which has the molecular formula of HCOOH, will…
A: Using integrated rate equation for first order reaction. We can figure out the time required to…
Q: [References] Use the References to access important values if needed for this question. What is the…
A:
m
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Predict the main product of the reaction in Figure 10. * A B C D The main product of the reaction cannot be predicted.Predict the product, draw the mechanism, and plot the reaction coordinate diagram for theE1 reaction.The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?
- Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →Do the reactions below proceed in good yield from left to right as shown? Please fill in Yes or NoConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:
- M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :Which of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?



